Abstract
Background
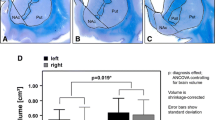
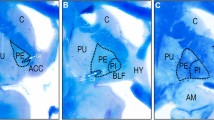
The Nucleus accumbens (Acc) is the main structure of the ventral striatum. It acts as a motor-limbic interface, being involved in emotional and psychomotor functions, frequently disturbed in neuropsychiatric disorders such as obsessive compulsive disorder and addiction. Most of the studies concerning the Acc were made in animals and those performed in humans are contradictory. Nevertheless, it has become a target for stereotactic deep brain stimulation for some of those diseases, when refractory to medical treatment. Previous studies performed by our group have established the localization, limits and dimensions of the human Acc and its stereotactic coordinates. Now it is our purpose to perform the Acc anatomical three-dimensional (3D) reconstruction in order to clarify its shape and topography and to render this nucleus a safer target for stereotactic procedures.
Methods
Anatomical coronal slicing of ten Acc from human brains was performed, perpendicular to the anterior commissure-posterior commissure line and to the midline; then the Acc contours were traced and its dimensions and 3D stereotactic coordinates measured, on each slice. Finally a 3D computerized model was created.
Results
The human Acc was identified as a distinct brain structure, with clear-cut limits on its posterior half. It lies parallel to the midline, descends caudally, and progresses from a globose to a flattened and dorsolateral concave shape. Its main expression is subcomissural.
Conclusion
This study defined more accurately the 3D anatomy of the human Acc, providing new tools for stereotactic procedures.






Similar content being viewed by others
References
Alho E, Grinberg L, Heinsen H, Fonoff E (2011) Review of Printed and Electronic Stereotactic Atlases of the Human Brain. In: Peres J Neuroimaging for Clinicians - Combining Research and Practice, InTech, pp 145–172
Aouizerate B, Cuny E, Bardinet E, Yelnik J, Martin-Guehl C, Rotge JY, Rougier A, Bioulac B, Tignol J, Mallet L, Burbaud P, Guehl D (2009) Distinct striatal targets in treating obsessive-compulsive disorder and major depression. J Neurosurg 111:775–779
Ballmaier M, Toga AW, Siddarth P, Blanton RE, Levitt JG, Lee M, Caplan R (2004) Thought disorder and nucleus accumbens in childhood: a structural MRI study. Psychiatry Res 130:43–55
Benabid AL, Pollak P, Louveau A, Henry S, de Rougemont J (1987) Combined (thalamotomy and stimulation) stereotactic surgery of the VIM thalamic nucleus for bilateral Parkinson disease. Appl Neurophysiol 50:344–346
Bewernick BH, Hurlemann R, Matusch A, Kayser S, Grubert C, Hadrysiewicz B, Axmacher N, Lemke M, Cooper-Mahkorn D, Cohen M, Brockmann H, Lenartz D, Sturm V, Schlaepfer T (2010) Nucleus accumbens deep brain stimulation decreases ratings of depression and anxiety in treatment-resistant depression. Biol Psychiatry 67:110–116
Blomstedt P, Sjöberg RL, Hansson M, Bodlund O, Hariz MI (2011) Deep brain stimulation in the treatment of depression. Acta Psychiatr Scand 123:4–11
Boileau I, Assaad JM, Pihl RO, Benkelfat C, Leyton M, Diksic M, Tremblay RE, Dagher A (2003) Alcohol promotes dopamine release in the human nucleus accumbens. Synapse 49:226–231
Brabec J, Krásený J, Petrovický P (2003) Volumetry of striatum and pallidum in man - anatomy, cytoarchitecture, connections, MRI and aging. Sb Lek 104:13–65
Carelli RM (2002) Nucleus accumbens cell firing during goal-directed behaviors for cocaine vs. “natural” reinforcement. Physiol Behav 76:379–387
Coffey RJ (2009) Printed stereotactic atlases, review. In: Lozano AM, Gildenberg PL, Tasker RR (eds) Textbook of stereotactic and functional neurosurgery. Springer, Berlin Heidelberg, pp 347–372
de Koning PP, Figee M, van den Munckhof P, Schuurman P, Denys D (2011) Current status of deep brain stimulation for obsessive-compulsive disorder: a clinical review of different targets. Curr Psychiatry Rep 13:274–282
Denys D, Mantione M, Figee M, van den Munckhof P, Koerselman F, Westenberg H, Bosch A, Schuurman R (2010) Deep brain stimulation of the nucleus accumbens for treatment-refractory obsessive-compulsive disorder. Arch Gen Psychiatry 67:1061–1068
Di Chiara G (2002) Nucleus accumbens shell and core dopamine: differential role in behaviour and addiction. Behav Brain Res 137:75–114
Fernandéz-Espejo E (2000) How does the nucleus accumbens tick? Rev Neurol 30:845–849
Fischer AH, Jacobson KA, Rose J, Zeller R (2008) Cryosectioning tissues. Cold Spring Harb Protoc. doi:10.1101/pdb.prot4991
Franzini A, Messina G, Gambini O, Muffatti R, Scarone S, Cordella R, Broggi G (2010) Deep-brain stimulation of the nucleus accumbens in obsessive compulsive disorder: clinical, surgical and electrophysiological considerations in two consecutive patients. Neurol Sci 31:353–359
Greenberg BD, Gabriels LA, Malone DA, Rezai AR, Friehs GM, Okun MS, Shapira NA, Foote KD, Cosyns PR, Kubu CS, Malloy PF, Salloway SP, Giftakis JE, Rise MT, Machado AG, Baker KB, Stypulkowski PH, Goodman WK, Rasmussen SA, Nuttin BJ (2008) Deep brain stimulation of the ventral internal capsule/ventral striatum for obsessive-compulsive disorder: worldwide experience. Mol Psychiatry 15:64–79
Greenberg BD, Malone DA, Friehs GM, Rezai AR, Kubu CS, Malloy PF, Salloway SP, Okun MS, Goodman WK, Rasmussen SA (2006) Three-year outcomes in deep brain stimulation for highly resistant obsessive-compulsive disorder. Neuropsychopharmacology 31:2384–93
Groeneweger HJ, Wright CI, Beuer AVJ (1996) The nucleus accumbens: gateway for limbic structures to reach the motor system? Prog Brain Res 107:485–511
Haber SN, Mc Farland NR (1999) The concept of the ventral striatum in nonhuman primates. Ann N Y Acad Sci 877:33–48
Heimer L (2000) Basal forebrain in the context of schizophrenia. Brain Res Rev 31:205–235
Heimer L (2003) A new anatomical framework for neuropsychiatric disorders and drug abuse. Am J Psychiatry 160:1726–1739
Heimer L (2003) The legacy of the silver methods and the new anatomy of the basal forebrain: implications for neuropsychiatry and drug abuse. Scand J Psychol 44:189–201
Heimer L, Harlan RE, Alheid GF, de Olmos J (1997) Substantia innominata: a notion which impedes clinical-anatomical correlations in neuropsychiatric disorders. Neuroscience 76:957–1006
Heinze HJ, Heldmann M, Voges J, Hinrichs H, Marco-Pallares J, Hopf JM, Müller UJ, Galazky I, Sturm V, Bogerts B, Münte TF (2009) Counteracting incentive sensitization in severe alcohol dependence using deep brain stimulation of the nucleus accumbens: clinical and basic science aspects. Front Hum Neurosci 3:1–11
Henderson MB, Green AI, Bradford PS, Chau DT, Roberts DW, Leiter JC (2010) Deep brain stimulation of the nucleus accumbens reduces alcohol intake in alcohol-preferring rats. Neurosurg Focus 29(E12):1–7
Holt D, Graybiel A, Saper C (1997) Neurochemical architecture of the human striatum. J Comp Neurol 384:1–25
Kuhn J, Brauer R, Pohl S, Lenartz D, Huff W, Kim EH, Klosterkoeffer J, Sturm V (2009) Observations on unaided smoking cessation after deep brain stimulation of the nucleus accumbens. Eur Addict Res 15:196–201
Kuhn J, Lenartz D, Mai JK, Huff W, Lee SH, Koulousakis A, Kloslerkoetter J, Sturm V (2007) Deep brain stimulation of the nucleus acumbens and the internal capsule in therapeutically refractory Tourette-syndrome. J Neurol 254:963–965
Lauer M, Heinsen H (1996) Cytoarchitectonics of the human nucleus accumbens. J Hirnforsch 37:243–254
Lauer M, Senitz D, Beckmann H (2001) Increased volume of the nucleus accumbens in schizophrenia. J Neural Transm 108:645–660
Mai JK, Assheuer J, Paxinos G (2004) Atlas of the human brain. Elsevier Academic Press, San Diego
Malone D, Dougherty D, Rezai A, Carpenter L, Friehs G, Eskandar E, Rauch S, Rasmussen S, Machado A, Kubu C, Tyrka A, Price L, Stypulkowski P, Giftakis J, Rise M, Malloy P, Salloway S, Greenberg B (2009) Deep brain stimulation of the ventral capsule/ventral striatum for treatment-resistant depression. Biol Psychiatry 65:267–275
Mavridis I, Anagnostopoulou S (2009) The human nucleus accumbens as a target for deep brain stimulation: anatomic study of electrode’s target point and stereotactic coordinates. Minim Invasive Neurosurg 52:212–215
Mavridis I, Anagnostopoulou S (2011) The safest electrode trajectory for deep brain stimulation of the human nucleus accumbens: a stereotactic anatomic study. Minim Invasive Neurosurg 54:16–20
Mavridis I, Boriatsis E, Anagnostopoulous S (2011) Anatomy of the human nucleus accumbens: a combined morphometric study. Surg Radiol Anat 33:405–414
Mavridis I, Boviatsis E, Anagnostopoulou S (2011) Stereotactic anatomy of the human nucleus accumbens: from applied mathematics to microsurgical accuracy. Surg Radiol Anat 33:583–94
Meredith GE, Pattiselanno A, Groenewegen HJ, Haber SN (1996) Shell amd core in the monkey and human nucleus accumbens identified with antibodies to calbindin D28K. J Comp Neurol 365:628–639
Mogenson GJ, Jones DL, Yim CY (1980) From motivation to action: functional interface between the limbic system and the motor system. Prog Neurobiol 14:69–97
Nestler EJ, Malenka RC (2004) The addicted brain. Sci Am 50–57
Neto LL, Oliveira E, Correia F, Ferreira AG (2008) The human nucleus accumbens: Where is it? A stereotactic, anatomical and magnetic resonance imaging study. Neuromodulation 11:13–22
Neuner I, Podoll K, Lenartz D, Sturm V, Schneider F (2009) Deep brain stimulation in the nucleus accumbens for intractable Tourette’s syndrome: follow-up report of 36 months. Biol Psychiatry 65:e5–e6
Nuttin B, Cosyns P, Demeulemeester H, Gybels J, Meyerson B (1999) Electrical stimulation in anterior limbs of internal capsules in patients with obsessive-compulsive disorder. Lancet 354:1526
Prensa L, Richard S, Parent A (2003) Chemical anatomy of the human ventral striatum and adjacent basal forebrain structures. J Comp Neurol 460:345–367
Saxena S, Rauch SL (2000) Functional neuroimaging and the neuroanatomy of obsessive-compulsive disorder. Psychiatr Clin N Am 23:563–586
Schaltenbrand G, Bailey P (1959) Introduction to stereotaxis with an atlas of the human brain. Georg Thieme Verlag, Stuttgart
Schlaepfer T, Cohen M, Frick C, Kosel M, Brodesser D, Axmacher N, Joe A, Kreft M, Lenartz D, Sturm V (2008) Deep brain stimulation to reward circuitry alleviates anhedonia in refractory major depression. Neuropsychopharmacology 33:368–377
Schulz G, Crooijmans HJA, Germann M, Scheffler K, Müller-Gerbl M, Müller B (2011) Three-dimensional strain fields in human brain resulting from formalin fixation. J Neurosci Methods 202:17–27
Sturm V, Lenartz D, Koulousakis A, Treuer H, Herholz K, Klein JC, Klosterkotter J (2003) The nucleus accumbens: a target for deep brain stimulation in obsessive-compulsive and anxiety disorders. J Chem Neuroanat 26:293–299
Talairach J, Tournoux P (1988) Co-planar stereotaxic atlas of the human brain. Georg Thieme Verlag, Stuttgart
Torres CV, Lozano AM (2008) Estimulación cerebral profunda para el tratamiento de la depresión resistente. Rev Neurol 47:477–482
Valencia-Alfonso C, Luigjes J, Smolders R, Cohen M, Levar N, Mazaheri A, van den Munckhof P, Schuurman R, van den Brink W, Denys D (2012) Effective deep brain stimulation in heroin addiction: a case report with complementary intracranial electroencephalogram. Biol Psychiatry 71:e35–e37
van Dijk A, Mason O, Klompmakers AA, Feenstra MGP, Denys D (2011) Unilateral deep brain stimulation in the nucleus accumbens core does not affect local monoamine release. J Neurosci Methods 202:113–118
Viswanathan A, Jimenez-Shaded J, Carvallo J, Jankovic J (2012) Deep brain stimulation for Tourette syndrome: target selection. Stereotact Funct Neurosurg 90:213–224
Voorn P, Brady LS, Schotte A, Berendse HW, Richfield EK (1994) Evidence for two neurochemical divisions in the human nucleus accumbens. Eur J Neurosci 6:1913–1916
Ward HE, Hwynn N, Okun MS (2010) Update on deep brain stimulation for neuropsychiatric disorders. Neurobiol Dis 38:346–353
Yelnik J, Bardinet E, Dormont D, Malandain G, Ourselin S, Tandé D, Karachi C, Ayache N, Cornu P, Agid Y (2007) A three-dimensional, histological and deformable atlas of the human basal ganglia. I. Atlas construction based on immunohistochemical and MRI data. NeuroImage 34:618–638
Zaborszky L, Hoemke L, Mohlberg H, Schleicher A, Amunts K, Zilles K (2008) Stereotactic probabilistic maps of the magnocellular cell groups in human basal forebrain. NeuroImage 42:1127–1141
Zahm DS (1999) Functional-anatomical implications of the nucleus accumbens core and shell subterritories. Ann N Y Acad Sci 877:113–128
Zhou H, Xu J, Jiang J (2011) Deep brain stimulation of nucleus accumbens on heroin-seeking behaviors: a case report. Biol Psychiatry 69:e41–e42
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comments
The nucleus accumbens definitely plays a pivotal role in the “reward” circuitry. Operations in this circuit are based on actions of two essential neurotransmitters: dopamine, which promotes desire, and serotonin, whose effects include satiety and inhibition. Furthermore, many animal studies have shown that all drugs increase the production of dopamine in the nucleus accumbens, while reducing that of serotonin. Deep bain stimulation (DBS) of the nucleus accumbens has been, therefore, investigated for the treatment of several psychiatric conditions including obsessive–compulsive disorder, treatment-resistant depression and, more recently, to modulate substance-induced dysfunction and to promote reversal of addictive behavior. This study, from the experienced group of Lisbon, will be a fundamental reference for DBS of the nucleus accumbens, a treatment that is increasingly offered to patients for psychiatric conditions. The limitation of this study is the same as that of traditional stereotactic atlases based on cadaver specimen, namely the potential spatial errors due to “post-mortem” structural changes and fixation and histological sectioning. Nonetheless, by using multiple specimens the authors offset this source of inaccuracy. The best stereotactic target coordinates within the nucleus accumbens, as delineated in this study, will come in the next future by integrating intraoperative neuroelectrophysiology, pre- and intraoperative neuroimaging, and postoperative neurological assessment.
Alfredo Conti
Messina, ITALY
Electronic supplementary material
Below is the link to the electronic supplementary material.
(MPG 7270 kb)
(MPG 3290 kb)
(MPG 7748 kb)
Rights and permissions
About this article
Cite this article
Lucas-Neto, L., Neto, D., Oliveira, E. et al. Three dimensional anatomy of the human nucleus accumbens. Acta Neurochir 155, 2389–2398 (2013). https://doi.org/10.1007/s00701-013-1820-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-013-1820-z