Abstract
Background
Largely discussed during the past decade, motor cortex reorganization in brain tumor surgery has been investigated only by few studies. We therefore aimed to investigate cortical motor representation after resection of perirolandic WHO grade II and III gliomas using navigated transcranial magnetic stimulation (nTMS).
Methods
Five patients were examined before neurosurgery and after a follow-up period of 17.7 ± 6.8 months. As a control, five healthy age-matched subjects were equally studied by nTMS in two sessions spaced 12.6 (range 2–35) days apart. Resting motor thresholds (RMT), hotspots and centers of gravity (CoG) were identified for the first dorsal interosseous (FDI), abductor pollicis brevis (APB), extensor digitorum (EXT), tibialis anterior (TA) and abductor hallucis (AH) muscles. Euclidian distances, coefficients of variance and intraclass correlation coefficients (ICC) were calculated.
Results
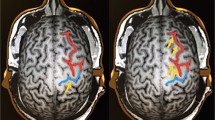
Healthy subjects showed moderate to excellent reliability measurement of RMT (ICC = 0.69–0.94). Average displacement of CoGs across sessions was 0.68 ± 0.34 cm in the dominant and 0.76 ± 0.38 cm in the non-dominant hemisphere; hotspots moved 0.87 ± 0.51 cm and 0.83 ± 0.45 cm, respectively.
In one patient these parameters differed significantly from the control group (p < 0.05 for both CoGs and hotspots). Overall, all patients’ CoGs moved 1.12 ± 0.93 cm, and hotspots were 1.06 ± 0.7 cm apart. In both patients and healthy subjects, movement of assessed parameters was more important along the X- than the Y-axis.
Conclusions
nTMS allows evaluating cortical reorganization after brain tumor surgery. It may contribute to the understanding of neurofunctional dynamics, thus influencing therapeutic strategy.

Similar content being viewed by others
References
Balslev D, Braet W, McAllister C, Miall RC (2007) Inter-individual variability in optimal current direction for transcranial magnetic stimulation of the motor cortex. J Neurosci Methods 162:309–313
Brown TG SC (1912) On the instability of a cortical point. Proc R Soc London Ser B 85
Byrnes ML, Thickbroom GW, Phillips BA, Mastaglia FL (2001) Long-term changes in motor cortical organisation after recovery from subcortical stroke. Brain Res 889:278–287
Cacchio A, Cimini N, Alosi P, Santilli V, Marrelli A (2009) Reliability of transcranial magnetic stimulation-related measurements of tibialis anterior muscle in healthy subjects. Clin Neurophysiol 120:414–419
Cacchio A, Paoloni M, Cimini N, Mangone M, Liris G, Aloisi P, Santilli V, Marrelli A (2011) Reliability of TMS-related measures of tibialis anterior muscle in patients with chronic stroke and healthy subjects. J Neurol Sci 303:90–94
Carroll TJ, Riek S, Carson RG (2001) Reliability of the input-output properties of the cortico-spinal pathway obtained from transcranial magnetic and electrical stimulation. J Neurosci Methods 112:193–202
Desmurget M, Bonnetblanc F, Duffau H (2007) Contrasting acute and slow-growing lesions: a new door to brain plasticity. Brain 130:898–914
Devanne H, Lavoie BA, Capaday C (1997) Input-output properties and gain changes in the human corticospinal pathway. Exp Brain Res 114:329–338
Duffau H (2001) Acute functional reorganisation of the human motor cortex during resection of central lesions: a study using intraoperative brain mapping. J Neurol Neurosurg Psychiatry 70:506–513
Duffau H (2005) Lessons from brain mapping in surgery for low-grade glioma: insights into associations between tumour and brain plasticity. Lancet Neurol 4:476–486
Duffau H (2009) A personal consecutive series of surgically treated 51 cases of insular WHO Grade II glioma: advances and limitations. J Neurosurg 110:696–708
Duffau H, Capelle L, Denvil D, Sichez N, Gatignol P, Lopes M, Mitchell MC, Sichez JP, Van Effenterre R (2003) Functional recovery after surgical resection of low grade gliomas in eloquent brain: hypothesis of brain compensation. J Neurol Neurosurg Psychiatry 74:901–907
Duffau H, Denvil D, Capelle L (2002) Long term reshaping of language, sensory, and motor maps after glioma resection: a new parameter to integrate in the surgical strategy. J Neurol Neurosurg Psychiatry 72:511–516
Fandino J, Kollias SS, Wieser HG, Valavanis A, Yonekawa Y (1999) Intraoperative validation of functional magnetic resonance imaging and cortical reorganization patterns in patients with brain tumors involving the primary motor cortex. J Neurosurg 91:238–250
Forster MT, Hattingen E, Senft C, Gasser T, Seifert V, Szelenyi A (2011) Navigated transcranial magnetic stimulation and functional magnetic resonance imaging—advanced adjuncts in preoperative planning for central region tumors. Neurosurgery 68:1317–1325
Gil-Robles S, Duffau H (2010) Surgical management of World Health Organization grade II gliomas in eloquent areas: the necessity of preserving a margin around functional structures. Neurosurg Focus 28:E8
Julkunen P, Saisanen L, Danner N, Niskanen E, Hukkanen T, Mervaala E, Kononen M (2009) Comparison of navigated and non-navigated transcranial magnetic stimulation for motor cortex mapping, motor threshold and motor evoked potentials. Neuroimage 44:790–795
Liepert J, Miltner WH, Bauder H, Sommer M, Dettmers C, Taub E, Weiller C (1998) Motor cortex plasticity during constraint-induced movement therapy in stroke patients. Neurosci Lett 250:5–8
Livingston SC, Ingersoll CD (2008) Intra-rater reliability of a transcranial magnetic stimulation technique to obtain motor evoked potentials. Int J Neurosci 118:239–256
Malcolm MP, Triggs WJ, Light KE, Shechtman O, Khandekar G, Gonzalez Rothi LJ (2006) Reliability of motor cortex transcranial magnetic stimulation in four muscle representations. Clin Neurophysiol 117:1037–1046
McGraw KOWS (1996) Forming inferences about some intraclass correlation coefficients. Psychol Methods 1:30–46
Meyer PT, Sturz L, Schreckenberger M, Spetzger U, Meyer GF, Setani KS, Sabri O, Buell U (2003) Preoperative mapping of cortical language areas in adult brain tumour patients using PET and individual non-normalised SPM analyses. Eur J Nucl Med Mol Imaging 30:951–960
Mills KR, Boniface SJ, Schubert M (1992) Magnetic brain stimulation with a double coil: the importance of coil orientation. Electroencephalogr Clin Neurophysiol 85:17–21
Mills KR, Nithi KA (1997) Corticomotor threshold to magnetic stimulation: normal values and repeatability. Muscle Nerve 20:570–576
Mortifee P, Stewart H, Schulzer M, Eisen A (1994) Reliability of transcranial magnetic stimulation for mapping the human motor cortex. Electroencephalogr Clin Neurophysiol 93:131–137
Niskanen E, Julkunen P, Saisanen L, Vanninen R, Karjalainen P, Kononen M (2010) Group-level variations in motor representation areas of thenar and anterior tibial muscles: navigated transcranial magnetic stimulation study. Hum Brain Mapp 31:1272–1280
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113
Portney LWM (2000) Foundation of clinical research: applications to practice. Prentice-Hall, Upper Saddle River, NJ
Robles SG, Gatignol P, Lehericy S, Duffau H (2008) Long-term brain plasticity allowing a multistage surgical approach to World Health Organization Grade II gliomas in eloquent areas. J Neurosurg 109:615–624
Rossini PM, Calautti C, Pauri F, Baron JC (2003) Post-stroke plastic reorganisation in the adult brain. Lancet neurology 2:493–502
Sanai N, Berger MS (2008) Glioma extent of resection and its impact on patient outcome. Neurosurgery 62:753–764, discussion 264–756
Schiffbauer H, Ferrari P, Rowley HA, Berger MS, Roberts TP (2001) Functional activity within brain tumors: a magnetic source imaging study. Neurosurgery 49:1313–1320, discussion 1320–1311
Solinas C, Lee YC, Reutens DC (2008) Effect of levetiracetam on cortical excitability: a transcranial magnetic stimulation study. Eur J Neurol 15:501–505
Tang LM, Chen ST, Hsu WC, Leung WM (1996) A longitudinal study of multimodal evoked potentials in patients following radiotherapy for nasopharyngeal carcinoma. Neurology 47:521–525
Taniguchi M, Kato A, Ninomiya H, Hirata M, Cheyne D, Robinson SE, Maruno M, Saitoh Y, Kishima H, Yoshimine T (2004) Cerebral motor control in patients with gliomas around the central sulcus studied with spatially filtered magnetoencephalography. J Neurol Neurosurg Psychiatry 75:466–471
Thickbroom GW, Sammut R, Mastaglia FL (1998) Magnetic stimulation mapping of motor cortex: factors contributing to map area. Electroencephalogr Clin Neurophysiol 109:79–84
Thiel A, Herholz K, Koyuncu A, Ghaemi M, Kracht LW, Habedank B, Heiss WD (2001) Plasticity of language networks in patients with brain tumors: a positron emission tomography activation study. Ann Neurol 50:620–629
Uy J, Ridding MC, Miles TS (2002) Stability of maps of human motor cortex made with transcranial magnetic stimulation. Brain Topogr 14:293–297
Wassermann EM, McShane LM, Hallett M, Cohen LG (1992) Noninvasive mapping of muscle representations in human motor cortex. Electroencephalogr Clin Neurophysiol 85:1–8
Wolf SL, Butler AJ, Campana GI, Parris TA, Struys DM, Weinstein SR, Weiss P (2004) Intra-subject reliability of parameters contributing to maps generated by transcranial magnetic stimulation in able-bodied adults. Clin Neurophysiol 115:1740–1747
Wunderlich G, Knorr U, Herzog H, Kiwit JC, Freund HJ, Seitz RJ (1998) Precentral glioma location determines the displacement of cortical hand representation. Neurosurgery 42:18–26, discussion 26–17
Ziemann U, Lonnecker S, Steinhoff BJ, Paulus W (1996) Effects of antiepileptic drugs on motor cortex excitability in humans: a transcranial magnetic stimulation study. Ann Neurol 40:367–378
Acknowledgement
The authors thank Marina Heibel for her assistance with figure preparation.
Disclosure
Dr. Forster and Dr. Szelényi have served as speakers for Nexstim. Financing of the study was completely provided by institutional grants from the Department of Neurosurgery. The authors have no personal or institutional interest in any of the devices, materials or methods described in this article.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Forster, MT., Senft, C., Hattingen, E. et al. Motor cortex evaluation by nTMS after surgery of central region tumors: a feasibility study. Acta Neurochir 154, 1351–1359 (2012). https://doi.org/10.1007/s00701-012-1403-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-012-1403-4