Abstract
Purpose
Vascular endothelial growth factor (VEGF) has been reported to enhance vascular permeability and angiogenesis in the abdominal wall, thereby contributing to peritoneal dissemination with malignant ascites. We conducted this experimental study to find out if bevacizumab, a humanized monoclonal antibody against VEGF, had a suppressive effect on peritoneal dissemination from gastric cancer, in an experimental nude mouse model of peritoneal metastasis.
Methods
Each mouse was treated with a single intraperitoneal (i.p.) injection of bevacizumab. Five mice were killed, and we measured their body weight, the mean number of tumor nodules, and the volume of ascites. We also extracted retroperitoneal tissues for histological examination, to count the frequency of mitosis, and to calculate the mitotic index. Another five mice were monitored until death, and their mean survival duration was calculated.
Results
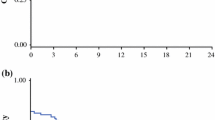
The volume of ascites and the mitotic index were significantly lower in the therapy group than in the nontherapy group (P = 0.042 and P < 0.01, respectively). The survival curve of the therapy group was significantly higher than that of the nontherapy group (P = 0.005).
Conclusion
Bevacizumab may suppress peritoneal dissemination from gastric cancer.
Similar content being viewed by others
References
Sakuramoto S, Sasako M, Yamaguchi T, Kinoshita T, Fujii M, Nashimoto A, et al. Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. N Engl J Med 2007;357:1810–1820.
Ikeguchi M, Miyake T, Matsunaga T, Yamamoto M, Fukumoto Y, Yamada Y, et al. Recent results of therapy for scirrhous gastric cancer. Surg Today 2009;39:290–294.
Yonemura Y, Endou Y, Yamaguchi T. Mechanisms of the formation of the peritoneal dissemination in gastric cancer. Int J Oncol 1996;8:795–802.
Yonemura Y. Peritoneal dissemination. Tokyo: Health Publishers; 1996.
Liotta LA. Tumor invasion and metastases-role of the extracellular matrix. Cancer Res 1986;46:1–7.
Zebrowski BK, Liu W, Ramirez K, Akagi Y, Mills GB, Ellis LM. Markedly elevated levels of vascular endothelial growth factor in malignant ascites. Ann Surg Oncol 1999;6:373–378.
Dvorak HF, Brown LF, Detmar M, Dvorak AM. Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am J Pathol 1995;50:1209–1239.
Belotti D, Paganoni P, Manenti L, Garofalo A, Marchini S, Taraboletti G, et al. Matrix metalloproteinases (MMP9 and MMP2) induce the release of vascular endothelial growth factor (VEGF) by ovarian carcinoma cells: implications for ascites formation. Cancer Res 2003;63:5224–5229.
Aoyagi K, Kouhuji K, Yano S, Miyagi M, Imaizumi T, Takeda J, et al. VEGF significance in peritoneal recurrence from gastric cancer. Gastric Cancer 2005;8:155–163.
Miyagi M, Aoyagi K, Kato S, Shirouzu K. The TIMP-1 gene transferred through adenovirus mediation shows a suppressive effect on peritoneal metastases from gastric cancer. Int J Clin Oncol 2007;12:17–24.
Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey Y, Dvorak HF. Tumor cells secret a vascular permeability factor that promotes accumulation of ascites fluid. Science 1983;219:983–985.
Ferrara N, Henzel WJ. Pituitary follicular cells secrete a novel heparin-binding growth factor specific for vascular endothelial cells. Biochem Biophys Res Commun 1989;161:851–858.
Pepper MS, Ferrara N, Orci L, Montesano R. Vascular endothelial growth factor (VEGF) induces plasminogen activators and plasminogen activator inhibitor-1 in microvascular endothelial cells. Biochem Biophys Res Commun 1991;181:902–906.
Uenomori EN, Ferrara N, Bauer EA, Amento EP. Vascular endothelial growth factor induces interstitial collagenase expression in human endothelial cells. J Cell Physiol 1992;153:357–362.
Shaheen RM, Davis DW, Liu W, Zebrowski BK, Wilson MR, Bucana CD, et al. Antiangiogenic therapy targeting the tyrosine kinase receptor for vascular endothelial growth factor receptor inhibits the growth of colon cancer liver metastasis and induces tumor and endothelial cell apoptosis. Cancer Res 1999;59:5412–5416.
Gerber HP, McMurtrey A, Kowalski J, Yan M, Keyt BA, Dixit V, et al. Vascular endothelial growth factor regulates endothelial cell survival through the phosphatidylinositol 3′-kinase/Akt signal transduction pathway: requirement for Klk-1/FDR activation. J Biol Chem 1998;273:30336–30343.
Lissoni P, Malugani F, Bonfanti A, Bucovec R, Secondino S, Brivio F, et al. Abnormally enhanced blood concentrations of vascular growth factors (VEGF) in metastatic cancer patients and their relation to circulating dendritic cells, IL-12 and endothelin-1. J Biol Regul Homeost Agents 2001;15:140–144.
Ellis LM, Hicklin DJ. Pathways mediating resistance to vascular endothelial growth factor-targeted therapy. Clin Cancer Res 2008;14:6371–6375.
Yoneda J, Kuniyasu H, Crispens MA, Price JE, Bucana CD, Fidler IJ. Expression of angiogenesis-related genes and progression of human ovarian carcinomas in nude mice. J Natl Cancer Inst 1998;90:447–454.
Kondo Y, Arii S, Mori A, Furutani M, Chiba T, Imamura M. Enhancement of angiogenesis, tumor growth, and metastasis by transfection of vascular endothelial growth factor into Lo Vo human colon cancer cell line. Clin Cancer Res 2000;6:622–630.
Mori A, Arii S, Furutani M, Mizumoto M, Uchida S, Furuyama H, et al. Soluble Flt-1 gene therapy for peritoneal metastases using HVJ-cationic liposomes. Gene Ther 2000;7:1027–1033.
Nagy JA, Masse EM, Herzberg KT, Meyers MS, Yeo KT, Yeo TK, et al. Pathogenesis of ascites tumor growth: vascular permeability factor, vascular hyperpermeability, and ascites fluid accumulation. Cancer Res 1995;55:360–368.
Kraft A, Weindel K, Ochs A, Marth C, Zmija J, Schumacher P, et al. Vascular endothelial growth factor in the sera and effusion of patients with malignant and nonmalignant disease. Cancer 1999;85:178–187.
Mesiano S, Ferrara N, Jaffe RB. Role of vascular growth factor in ovarian cancer: inhibition of ascites formation by immunoneutralization. Am J Pathol 1998;153:1249–1256.
Wildiers H, Guetens G, De Boeck G, Verbeken E, Landuyt B, Landuyt W, et al. Effect of antivascular endothelial growth factor treatment on the intratumoral uptake of CPT-11. Br J Cancer 2003;88:1979–1986.
Ferrara N, Hillan KJ, Gerber HP, Novotny W. Discovery and development of bevacizumab, an anti-VEGF antibody for treating cancer. Nat Rev Drug Discov 2004;3:391–400.
Jain RK, Tong RT, Munn LL. Effect of vascular normalization by antiangiogenic therapy on interstitial hypertension, peritumor edema, and lymphatic metastasis: insights from a mathematical model. Cancer Res 2007;67:2729–2735.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Imaizumi, T., Aoyagi, K., Miyagi, M. et al. Suppressive effect of bevacizumab on peritoneal dissemination from gastric cancer in a peritoneal metastasis model. Surg Today 40, 851–857 (2010). https://doi.org/10.1007/s00595-009-4154-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-009-4154-y