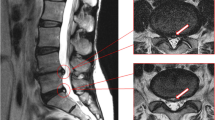
Abstract
Purpose
Low back pain (LBP) is one of the most prevalent health condition worldwide and responsible for the most years lived with disability, yet the etiology is often unknown. Magnetic resonance imaging (MRI) is frequently used for treatment decision even though it is often inconclusive. There are many different image features that could relate to low back pain. Conversely, multiple etiologies do relate to spinal degeneration but do not actually cause the perceived pain. This narrative review provides an overview of all possible relevant features visible on MRI images and determines their relation to LBP.
Methods
We conducted a separate literature search per image feature. All included studies were scored using the GRADE guidelines. Based on the reported results per feature an evidence agreement (EA) score was provided, enabling us to compare the collected evidence of separate image features. The various relations between MRI features and their associated pain mechanisms were evaluated to provide a list of features that are related to LBP.
Results
All searches combined generated a total of 4472 hits of which 31 articles were included. Features were divided into five different categories:’discogenic’, ‘neuropathic’,’osseous’, ‘facetogenic’, and’paraspinal’, and discussed separately.
Conclusion
Our research suggests that type I Modic changes, disc degeneration, endplate defects, disc herniation, spinal canal stenosis, nerve compression, and muscle fat infiltration have the highest probability to be related to LBP. These can be used to improve clinical decision-making for patients with LBP based on MRI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Low back pain (LBP) is responsible for more years lived with disability than any other health condition worldwide [1, 2]. Current literature describes a prevalence ranging between 1.4% and 20% depending on which definition of LBP is used [3]. In the Netherlands, approximately 44% of the population experiences at least one episode of LBP in their lifetime with one in five reporting persistent back pain lasting longer than three months, defined as chronic low back pain (CLBP) [4]. LBP often results in substantial limitations in functional activities and is responsible for high healthcare and socioeconomic costs [5, 6]. In the vast majority of patients with LBP (85–90%) [7] the etiology is unknown, and it is challenging for medical specialists to identify patients who would benefit from either surgical or non-surgical interventions.
Although magnetic resonance imaging (MRI) of the lumbar spine is frequently performed in patients with LBP, appropriate use and interpretation in patients with LBP remains controversial [8, 9]. There are many different image features visible on MRI that could relate to LBP. Conversely, multiple etiologies do relate to spinal degeneration but do not actually cause the perceived pain[10]. Potential causes of LBP and the corresponding image features can be divided into five categories:’discogenic’, ‘neuropathic’,’osseous’, ‘facetogenic’, and’paraspinal’ [11]. The currently available literature is mainly focused on only one of these image feature categories, and thus each study or review addresses only part of the possible etiologies. An overview with a broader scope in which all possible lumbar MRI features are related to LBP is lacking. Radiologists, spinal surgeons, and other clinicians could benefit from such an overview in their reporting and decision-making. The multitude of small-scale studies on single image features makes it currently difficult to oversee which study results are reliable and which features are of clinical relevance and supported by evidence.
The purpose of this review was to evaluate a wide range of image features and the available evidence for a relation with LBP across five feature categories (i.e.’discogenic’, ‘neuropathic’,’osseous’, ‘facetogenic’, and’paraspinal’) using separate literature searches. In light of the large number of image features and corresponding literature, the focus of this review was to include large-scale, high evidence studies. This review provides a comprehensive overview and discussion of relevant LBP image features.
Methods
This study was designed as a narrative review and follows the quality assessment guidelines of the Scale for the Assessment of Narrative Review Articles (SANRA), a brief critical appraisal tool for the assessment of non-systematic articles [12]. To provide this overview the project team, consisting of orthopedic spine surgeons, musculoskeletal radiologists, and a methodologist created an initial list of image features by reviewing relevant literature. An image feature was defined as a pathology or degenerative process visible on lumbar spine MRI images that possibly relate to LBP. The resulting 29 image features (Table 1) were categorized as’discogenic’, ‘neuropathic’,’osseous’, ‘facetogenic’, or’paraspinal’ [11].
Search strategy
A separate literature search was performed for each image feature in MEDLINE. The queries consisted of search terms related to magnetic resonance and LBP, which were used in all queries, and search terms related to a specific image feature (Appendix, Table 4). The feature-specific search terms were constructed from keywords and synonyms related to that specific feature.
Study selection
To ensure that only relevant publications were included, studies were screened for eligibility on predefined selection criteria. First, all search results were filtered using three inclusion criteria: (1) published in or after the year 2000 to ensure that subjects were scanned with modern MRI systems, (2) written in English, and (3) studies involving human subjects, which excluded all animal studies. Publications that met these criteria underwent title and abstract screening. Publications were included for full-text screening when the research aim was to directly assess the relation between one or multiple image features and any form of LBP. Although reviews were not included, publications that were referenced in these reviews were considered for inclusion. Full-text publications were excluded based on two criteria. First, studies with fewer than 100 subjects were excluded to limit our review to larger population-based studies that provide more robust evidence than smaller observational studies. Second, studies that reported on MRI scans acquired with a field strength of less than 1.5 T were excluded to ensure that there were no large differences in image quality and to match clinical practice where 1.5 T and 3 T are most common. Relevant findings from the remaining studies were entered into a data extraction table.
Data extraction
Data extracted included the year of publication, study design, type and duration of low back pain (current LBP (no period specified), CLBP, LBP for a specified period, disabling LBP, sciatica, radiculopathy, and all types of LBP), number of included subjects, main finding per separate image feature (e.g., the odds ratio and the related p-value), and a GRADE score (1–4). Data extraction was performed by one reviewer (JG) and checked by a second (MH).
Best evidence synthesis
The GRADE scoring system was used to assess the certainty of the evidence of each study [13]. GRADE consists of four categories to indicate the quality of evidence: high (GRADE 1), moderate (GRADE 2), low (GRADE 3), and very low (GRADE 4). The study design determines the initial quality of evidence. Randomized trials start in the high category (GRADE 1) and observational studies start in the low category (GRADE 3). The initial GRADE score decreases when finding risk of bias, imprecision, inconsistency, indirectness, or publication bias and increased when finding a large magnitude of effect and when all residual confounding would decrease the magnitude of effect.
We classified an association of an image feature with LBP as insufficiently evident when either no studies or only one GRADE 3 study met the inclusion criteria. When at least one GRADE 2 study or more than two GRADE 3 studies met the criteria, we considered the association sufficiently evident. Subsequently, when no studies or only one GRADE 3 study was included, it was labeled as “insufficient amount of evidence”. Best evidence synthesis was performed by two reviewers (JG and MH).
The agreement between different studies reporting on the same image feature was summarized by calculating the average evidence agreement (EA) per feature. The EA was defined as the percentage of studies that did find a relation between a feature and LBP compared to the studies that did not. The results of each study were categorized as either positive association, no association, or mixed results. The results of a study were labeled as mixed results when were inconclusive, such as different findings for different types of LBP. The average EA per image feature was calculated by dividing the number of studies with a positive association by the total number of studies. Studies with mixed results were counted as half a positive study. Consequently, when all studies of a specific image feature showed mixed results the EA was 50%. Based on the EA, all image features were divided into three categories to give an overall summary of the collected evidence: positive association (EA ≥ 67%), mixed results (33% < EA < 67%), and no association (EA ≤ 33%). No association meant that the included studies were inconclusive, and therefore no association was found.
Results
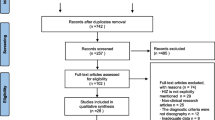
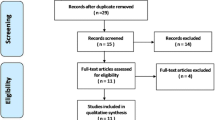
All searches combined generated a total of 4472 hits, including duplicates. After title and abstract screening, 251 were assessed in full text. A total of 31 studies met the selection criteria and were included in this review (Fig. 1). In Table 2 an overview of study characteristics is shown. Twelve studies were rated with a GRADE score of two while the remaining 19 studies had a GRADE score of three.
An overview of the best evidence synthesis per image feature is shown in Table 3. Eleven out of the 29 image features showed a positive association (EA ≥ 66%; Modic changes in general [67%], Modic changes type I [100%], disc narrowing [67%], endplate defects [67%], Pfirrmann grade [67%], disc herniation [100%], disc extrusion [100%], ligamentum flavum hypertrophy [100%], central spinal canal stenosis [70%], nerve compression [100%], and muscle fat infiltration [67%]). Two image features appeared to have mixed results (EA between 33–66%; spondylolisthesis [40%], and paraspinal muscle cross-sectional area [50%]). Seven out of the 29 image features showed no association (EA ≤ 33%; Modic changes type II [33%], Modic changes type III [0%], disc bulging [0%], high intensity zone [31%], disc protrusion [0%], foramen stenosis [33%], and facet fluid sign [25%]).
For nine image features the evidence was inconclusive (Table 3). For these features, an insufficient amount of evidence was found meaning either no studies were found that met our selection criteria or only one study with GRADE 3 was included. These features were black discs, synovial cysts, epidural fat, facet tropism, facet arthrosis, facet hypertrophy, osteomyelitis, and vertebral body infarction.
Discussion
The purpose of this review was to create an overview of the available evidence on the relation between certain MRI image features and low back pain (LBP). The use of MRI in clinical decision making for patients with LBP is debatable. Especially in the older population, an MRI scan of the lumbar spine is almost certain to show degenerative changes of the spine. There is an abundance of small cross-sectional studies that evaluate a small set of image features, while large population-based studies of high methodological quality are scarce. A complex multifactorial disorder such as LBP demands high-quality research which addresses potential causes of LBP and their relation to MRI image features. In this narrative review, the relevant features were carefully reviewed and compared to the currently available literature. Of the 29 considered features, categorized as discogenic, neurogenic, osseous, facetogenic, and paraspinal, 11 features showed a positive association, 2 showed mixed results, 7 showed no association with LBP, and 9 had inconclusive evidence. The relations between features and their associated pain mechanisms were evaluated to provide an overview of features that have the highest probability to be related to LBP. All features are discussed separately per feature category.
Discogenic features
One of the major sources of discogenic pain is disc degeneration, which is a complex process [11]. In a meta-analysis performed by Brinjikji et al. [45], the authors found a strong association between disc degeneration and LBP in the adult population 50 years of age or younger. Disc degeneration is commonly classified with the Pfirrmann grading system [46]. In this review three studies on Pfirrmann grading showed an EA of 67%, resulting in a positive association with LBP. The single paper in which no relation was found between Pfirrmann grading and LBP, initially did show a significant relation [21]. However, when adjusted for Modic changes and disc protrusion/hernia, this relation ceases to be significant, illustrating the complex relationship between disc degeneration and LBP. Since the Pfirrmann grade is determined by evaluating the disc height and signal intensity of the nucleus pulposus it can be expected that these features also have a high EA. For disc height as an individual image feature, we found an EA of 67% and for low signal intensity of the nucleus pulposus (black disc), we found limited evidence for a relationship with LBP. This might raise the question of whether only disc height would be sufficient in evaluating disc degeneration. However, the presented evidence in this study on black discs was insufficient, leaving this question unanswered.
Disc bulging is related to disc degeneration and reduced disc height, yet this feature showed an EA of 0%. Only two studies were included on disc bulging, both stating no relation between this image feature and LBP [18, 19]. However, in the meta-analyses of Brinjikji et al. the results contradict this finding [45]. The authors included three studies on disc bulging, using different inclusion and exclusion criteria, and concluded that a strong association exists between disc bulging and LBP [47,48,49]. However, in two of these studies, only adolescent patients were included [48, 49]. It is known that adolescents have a significantly lower prevalence of disc bulging compared to adult populations [50].
Annular fissures, often reported as high intensity zones (HIZ) in the annulus, are related to both disc degeneration and disc bulging as well [51, 52]. Eight studies on annular fissures and HIZ were included with an overall EA of 31%. The recently published systematic review by Teraguchi et al. (1541 included patients) and the narrative review by Cheung et al., both stated there is no uniform consensus in the present literature, which is in line with the results we present [51, 53]. However, annular fissures can be difficult to identify in MR images, especially when the image quality is low. Berger-Roscher et al. showed that MR imaging systems with varying field strengths demonstrated differences in their ability to visualize annular fissures and concluded that clinically used systems often do not visualize annular fissures well[54]. This limitation of MR imaging systems currently used in clinical practice may be the cause of inconsistent results in different studies. Further research with high resolution MR images is necessary to determine whether a relationship between annular fissures and LBP exists.
Neurogenic features
Neurogenic features are mostly associated with different types of neuropathic pain such as radiculopathy or sciatica. Even though our search strategy specifically targeted research that included LBP, studies that reported neuropathic pain were also included. The EA was based on all included studies, regardless of the reported type of pain. Of all 31 included studies, one [28] focused on neuropathic pain only, and eight [15, 16, 18, 21, 22, 37, 42, 43] studies focused on both neuropathic pain and LBP.
In total six studies were included on either disc herniation, disc extrusion, and disc protrusion (EA 100%, 100%, and 0% respectively) or a combination of the three. The difference between disc extrusion and protrusion is explained by the mechanism of pain. Both often cause neuropathic pain due to nerve compression [11]. Disc extrusion is distinguished from protrusion by the damage or tear of the annulus. This in itself triggers an immune response and inflammation which can induce additional LBP [11]. The pain mechanisms of all other neurogenic image features are either mechanical pressure of tissue nerve roots or nerve stimulation by proinflammatory cytokines and neurotransmitters [11]. Of these features only spinal canal stenosis (EA of 70%), nerve compression (EA of 100%) and ligamentum flavum hypertrophy (EA of 100%) had an EA higher than 67%.
Surprisingly, foraminal stenosis showed no association with LBP (33%) even though the same pain mechanisms can be at play as with spinal canal stenosis and nerve compression. Pain symptoms may have a different distribution depending on the type and location of neurovascular compression [55]. Since foraminal stenosis causes compression of specific nerves, symptoms are usually similar to radiculopathy [11]. In total three studies on foraminal stenosis were included in the current review. Two studies showed no association with LBP and evaluated LBP specifically without looking at any types of neuropathic pain [19, 25]. The third included study, written by Janardhana et al. (2010), evaluated LBP as well as neuropathic pain and showed a positive association with pain in general [18]. This illustrates the difference between LBP and neuropathic pain and might explain the different findings within neurogenic image features.
Osseous features
Osseous features can be divided into two different categories. Firstly, features related to pathologies within the bone that cause pain, such as Modic changes and endplate defects. Secondly, structural abnormalities causing instability or spinal compression such as spondylolisthesis or spondylolysis.
Modic changes are often reported and used in clinical decision making, yet their etiology is not well understood [11]. Our findings show that both Modic changes in general (67%) and type I changes (100%) are related to LBP. However, type II (33%) and III (0%) changes showed no association with LBP. With type I changes the general agreement is that pain is induced by an inflammatory reaction, but the cause of inflammation is still debated [11, 56, 57]. This relation with LBP is mostly acknowledged [58], which does not apply to type II and III Modic changes [11]. This is also supported by the findings of this review. Similarly, Brinkjikji et al. reported a significant association between Modic type I and LBP while Modic changes as a whole (Modic 1–3) did not have an association with LBP [45]. In contrast, Herlin et al. stated that the associations between Modic changes and LBP-related outcomes are inconsistent in current literature [59].
Endplate defects include Schmorl nodes, fractures, avulsions/erosions, and calcifications which all have been considered to be clinically associated with LBP [57]. Also, endplate defects identified using discography prove to be associated with LBP [60]. This corroborates with the EA of 75% for the association between endplate defects and LBP found in our study. These findings are in line with a recently published systematic review, which provides moderate quality evidence of an association between structural endplate defects and LBP [61]. More specifically, consistent evidence was shown for an association between erosion, sclerosis and Schmorl nodes, and LBP [61]. Moreover, Schmorl nodes are known to be associated with Modic changes and other degenerative features [57].
Spondylolisthesis is widely used in clinical decision making. However, in our study we included five studies showing a combined EA of 40% for LBP. Two of these studies showed no association with LBP specifically [19, 20], whereas the remaining three studies showed either a positive association or mixed results while including both LBP and neuropathic pain [15, 17, 22]. Both Ishimoto et al. and de Schepper et al. found no association with LBP and a positive association with radiating pain [15, 17]. These results are supported by the review of Gagnet et al. in which radiculopathy was mentioned as the most important symptom of spondylolisthesis [62]. The mixed result found in our study is therefore likely due to our search strategy, which was predominantly focused on LBP. Spondylolysis, which is often associated with spondylolisthesis, showed insufficient evidence. While often asymptomatic, patients with symptoms often suffer from LBP [62, 63]. The diagnosis of spondylolysis is rarely performed using MRI images since cortical bone is hypointense in both T1 and T2 weighted images. Therefore, fracture of the vertebral arch is mainly assessed using X-ray or CT images, which might explain the inconclusive evidence found in this review.
Facetogenic features
An estimated 15% of all LBP patients are caused by facetogenic pain [64, 65]. Pain is often caused by inflammation of the zygapophysial joint, which can directly cause pain [64]. This inflammation also causes swelling which possibly induces neurovascular compression [64]. In this review a total of three studies were included [19, 25, 34], which resulted in insufficient evidence on all facetogenic features except for facet fluid sign.
One paper showed no association between LBP and facet fluid sign [34] and one showed mixed results [25]. This feature shows an accumulation of fluid within the facet joint and is often associated with vertebral instability and spondylolisthesis [66]. However, the study performed by Shinto et al. reports no significant relation between facet effusion and L4-spondylolisthesis, which contradicts its relation with vertebral instability [34]. The authors also found that the prevalence of facet effusion did not increase with age, questioning its relation with degenerative changes.
Similar to spondylolysis, facetogenic pain is often associated with neuropathic pain, which was not specifically targeted by our search strategy. Furthermore, cortical bone, and therefore the zygapophysial joint, are better visualized in CT images.
Paraspinal features
The paraspinal features reviewed in this study focused on the paraspinal muscles and consisted of muscle fat infiltration (FI) (EA of 67%) and muscle cross-sectional area (CSA) (EA 50%). FI is related to muscle activity whereas CSA relates to muscle strength. The paraspinal muscles stabilize the spine and therefore protect spinal structures from potentially damaging stresses [67]. It is still debated whether paraspinal muscle dysfunction could be a primary cause of LBP.
In total four studies were included in this review [28, 32, 35, 41] of which three had mixed results since they included different muscles in their research, i.e. Multifidii, Erector spinae, psoas, and Quadratus lumborum. When separating the results per muscle group the multifidus muscle showed a positive association (67%) between FI and LBP, and mixed results (50%) between CSA and LBP. Furthermore, the erector spinae only showed a positive association (100%) between FI and LBP while all other muscles showed no association with either feature. This is supported by the review by Ranger et al. that showed mixed results as well, with only a strong relation between multifidus CSA and LBP [67]. All other muscles showed either conflicting evidence or no association between the two image features and LBP.
Nevertheless, the discussion remains on whether pain is generated by a certain muscle morphology or vice versa. Suri et al. systematically reviewed the relation between paraspinal muscle characteristics and future LBP. The authors found an association with both multifidus CSA and erector spinae FI, and future LBP while all other muscle characteristics had limited evidence or no association with LBP [68]. On the other hand, Cooley et al. found that neurocompressive disorders seem to alter muscle morphology at or below the affected level [69]. They conclude that muscle characteristics change due to present radiculopathy instead of the other way around. These contradicting results illustrate the lack of evidence on a causal relation between these paraspinal image features and LBP.
Overall findings
The purpose of this review was to create an overview of the available evidence on the relation between certain MRI image features and LBP. Ideally, this results in a list of features that are strongly associated with LBP, which will benefit radiological reporting and clinical decision making. Due to the intricate and conflicting nature of this research field, caution is warranted when drawing conclusions from the literature. In this narrative review, the relevant features were carefully reviewed and compared to the currently available literature. The various relations between MRI features and their associated pain mechanisms were evaluated to provide a list of features that have the highest probability to be related to LBP. This list comprises of type I Modic changes, disc degeneration, endplate defects, disc herniation, spinal canal stenosis, nerve compression, and muscle fat infiltration. Furthermore, this review reveals the gaps in the current literature and points out features for which robust evidence is lacking and for which large-scale studies are needed.
Limitations
The use of MRI in patients with LBP remains a controversial discussion and a narrative review with such a broad scope inherently has limitations. The first limitation is the distinction between different types of pain. There is a wide variety in types and duration of LBP as reported by the included studies as shown in Table 2. This study is focused on LBP, making it difficult to draw concise conclusions. Particularly, neurogenic features are mostly associated with neuropathic (radiating) pain, which was not specifically targeted in the search strategy. As many of the studies did not make a distinction between LBP and radiating pain, we chose to include all types of reported pain to create a clear overview of the present literature.
Second, the quality of evidence was assessed using the GRADE score, as part of the evidence synthesis [13]. All included studies were initially scored as GRADE 3 since no studies with an experimental design were included. This score was only upgraded to a GRADE 2 when either a large effect was shown or a large patient group was included. The way this scoring system was implemented therefore does not accurately assess the quality of evidence since it is only affected by effect size. Consequently, no weighing of features was performed in this review. However, it was still used to determine whether a sufficient amount of evidence was included to draw any conclusions.
Third, an evidence agreement of 67% or higher was considered as sufficient evidence for a positive association between LBP and an image feature. Even though this percentage might seem low, it still depicts the trend in current literature. Also, only 31 studies were included of the combined 4472 hits found with the different searches. This large number of hits also includes all the duplicates which was inevitable with our chosen method. Nevertheless, only a small number of studies met our inclusion criteria, which were deliberately defined such that studies with small sample sizes were excluded. This enabled this review to be more concise, keep the number of included papers at a reasonable level, and only focus on papers with a higher level of evidence. Our findings also display the limited amount of studies on MRI and LBP with more than 100 subjects included, indicating the need for larger studies in this field.
Lastly, as a criterion for this narrative review we included studies published after 2000. Consequently, relevant studies published before the year 2000 might have been excluded. The methods and technology used in medical imaging have improved significantly in the past 20 years, which could affect the accuracy and reliability of older studies, and with that the results in this review. However, excluding studies in which strong relationships between certain image features and LBP have been established before 2000 might have resulted in false negative results in our review. Comparison of the results with existing literature regardless of publication date mitigates this limitation by ensuring that relevant studies published before the year 2000 were discussed.
Conclusion
This study resulted in a comprehensive overview of which diagnostic lumbar MRI image features have the highest probability of a strong relationship with LBP. These features include type I Modic changes, disc degeneration, endplate defects, disc herniation, spinal canal stenosis, nerve compression, and muscle fat infiltration. Also, several features which are generally associated with LBP show no apparent relationship with LBP. This overview supports interpreting lumbar MRI scans of LBP patients and shows which features are related to the perceived pain. The findings of this review can be used to improve clinical decision-making for patients with LBP based on MRI images.
References
Hoy D, March L, Brooks P et al (2014) The global burden of low back pain: estimates from the global burden of disease 2010 study. Ann Rheum Dis 73:968–974. https://doi.org/10.1136/annrheumdis-2013-204428
Wu A, March L, Zheng X et al (2020) Global low back pain prevalence and years lived with disability from 1990 to 2017: estimates from the global burden of disease study 2017. Ann Transl Med 8:299–299. https://doi.org/10.21037/atm.2020.02.175
Fatoye F, Gebrye T, Odeyemi I (2019) Real-world incidence and prevalence of low back pain using routinely collected data. Rheumatol Int 39:619–626. https://doi.org/10.1007/s00296-019-04273-0
Picavet HSJ, Schouten JSAG (2003) Musculoskeletal pain in the Netherlands: prevalences, consequences and risk groups, the DMC3-study. Pain 102:167–178. https://doi.org/10.1016/s0304-3959(02)00372-x
Lambeek LC, van Tulder MW, Swinkels ICS et al (2007) The trend in total cost of back pain in the netherlands in the period 2002 to 2007. Spine 36:1050–1058. https://doi.org/10.1097/BRS.0b013e3181e70488
van Tulder M (2011) Health technology assessment (HTA) increasingly important in spine research. Eur Spine J 20:999–1000. https://doi.org/10.1007/s00586-011-1885-4
Haldeman S, Kopansky-Giles D, Hurwitz EL et al (2012) Advancements in the management of spine disorders. Best Pract Res Clin Rheumatol 26:263–280. https://doi.org/10.1016/j.berh.2012.03.006
Sheehan NJ (2010) Magnetic resonance imaging for low back pain: indications and limitations. Ann Rheum Dis 69:7–11. https://doi.org/10.1136/ard.2009.110973
Rao D, Scuderi G, Scuderi C et al (2018) The use of imaging in management of patients with low back pain. J Clin Imaging Sci 8:30. https://doi.org/10.4103/jcis.JCIS_16_18
Boden S, Davis D, Dina T et al (1990) Abnormal magnetic-resonance scans of the lumbar spine in asymptomatic subjects. a prospective investigation. J Bone Jt Surg 72:403–408
Ota Y, Connolly M, Srinivasan A et al (2020) Mechanisms and origins of spinal pain: from molecules to anatomy, with diagnostic clues and imaging findings. Radiographics 40:1163–1181. https://doi.org/10.1148/rg.2020190185
Baethge C, Goldbeck-Wood S, Mertens S (2019) SANRA—a scale for the quality assessment of narrative review articles. Res Integr Peer Rev 4:5. https://doi.org/10.1186/s41073-019-0064-8
Balshem H, Helfand M, Schünemann HJ et al (2011) GRADE guidelines: 3. Rating the quality of evidence. J Clin Epidemiol 64:401–406. https://doi.org/10.1016/j.jclinepi.2010.07.015
Çevik S, Yılmaz H (2020) Evaluation of the relationship between clinical symptoms and modic changes. Cureus. https://doi.org/10.7759/cureus.6970
de Schepper EI, Koes BW, Veldhuizen EF et al (2016) Prevalence of spinal pathology in patients presenting for lumbar MRI as referred from general practice. Fam Pract 33:51–56. https://doi.org/10.1093/fampra/cmv097
Haig AJ, Geisser ME, Tong HC et al (2007) Electromyographic and magnetic resonance imaging to predict lumbar stenosis, low-back pain, and no back symptoms. JBJS 9:358–366
Ishimoto Y, Yoshimura N, Muraki S et al (2017) Association of lumbar spondylolisthesis with low back pain and symptomatic lumbar spinal stenosis in a population-based cohort: the Wakayama spine study. Spine 42:E666–E671. https://doi.org/10.1097/BRS.0000000000001960
Janardhana AP, Rao S, Kamath A (2010) Correlation between clinical features and magnetic resonance imaging findings in lumbar disc prolapse. Indian J Orthop 44:263–269
Jarvik JJ, Hollingworth W, Heagerty P et al (2001) The longitudinal assessment of imaging and disability of the back (LAIDBack) study: baseline data. Spine 26:1158–1166. https://doi.org/10.1097/00007632-200105150-00014
Kasch R, Truthmann J, Hancock MJ et al (2021) Association of lumbar MRI findings with current and future back pain in a population-based cohort study. Spine 47:201–211. https://doi.org/10.1097/BRS.0000000000004198
Kovacs FM, Arana E, Royuela A et al (2014) Disc degeneration and chronic low back pain: an association which becomes nonsignificant when endplate changes and disc contour are taken into account. Neuroradiology 56:25–33. https://doi.org/10.1007/s00234-013-1294-y
Lai MKL, Cheung PWH, Samartzis D et al (2021) Clinical implications of lumbar developmental spinal stenosis on back pain, radicular leg pain, and disability. Bone Jt J 103-B:131–140. https://doi.org/10.1302/0301-620X.103B1.BJJ-2020-1186.R2
Määttä JH, Wadge S, MacGregor A et al (2015) ISSLS prize winner: vertebral endplate (modic) change is an independent risk factor for episodes of severe and disabling low back pain. Spine 40:1187–1193. https://doi.org/10.1097/BRS.0000000000000937
Määttä JH, Karppinen J, Paananen M et al (2016) Refined phenotyping of modic changes: imaging biomarkers of prolonged severe low back pain and disability. Med Baltim 95:e3495. https://doi.org/10.1097/MD.0000000000003495
Mainka T, Lemburg SP, Heyer CM et al (2013) Association between clinical signs assessed by manual segmental examination and findings of the lumbar facet joints on magnetic resonance scans in subjects with and without current low back pain: A prospective, single-blind study☆. Pain 154:1886–1895. https://doi.org/10.1016/j.pain.2013.06.018
Mera Y, Teraguchi M, Hashizume H et al (2020) Association between types of Modic changes in the lumbar region and low back pain in a large cohort: the Wakayama spine study. Eur Spine J. https://doi.org/10.1007/s00586-020-06618-x
Middendorp M, Vogl TJ, Kollias K et al (2017) Association between intervertebral disc degeneration and the oswestry disability index. J Back Musculoskelet Rehabil 30:819–823. https://doi.org/10.3233/BMR-150516
Min JH, Choi HS, Ihl Rhee W, Lee JI (2013) Association between radiculopathy and lumbar multifidus atrophy in magnetic resonance imaging. J Back Musculoskelet Rehabil 26:175–181. https://doi.org/10.3233/BMR-130365
Mok FPS, Samartzis D, Karppinen J et al (2016) Modic changes of the lumbar spine: prevalence, risk factors, and association with disc degeneration and low back pain in a large-scale population-based cohort. Spine J 16:32–41. https://doi.org/10.1016/j.spinee.2015.09.060
Munir S, Freidin MB, Rade M et al (2018) Endplate defect is heritable, associated with low back pain and triggers intervertebral disc degeneration: a longitudinal study from TwinsUK. Spine 43:1496–1501. https://doi.org/10.1097/BRS.0000000000002721
Ogon I, Takashima H, Morita T et al (2020) Relevance between Schmorl’s node and lumbar intervertebral disc degeneration quantified with magnetic resonance imaging T2 mapping in chronic low back pain. Asian Spine J 14:621–628. https://doi.org/10.31616/asj.2019.0231
Sasaki T, Yoshimura N, Hashizume H et al (2017) MRI-defined paraspinal muscle morphology in Japanese population: the Wakayama spine study. PLOS ONE 12:e0187765. https://doi.org/10.1371/journal.pone.0187765
Saukkonen J, Määttä J, Oura P et al (2020) Association between modic changes and low back pain in middle age: a Northern Finland birth cohort study. Spine 45:1360–1367. https://doi.org/10.1097/BRS.0000000000003529
Shinto K, Minamide A, Hashizume H et al (2019) Prevalence of facet effusion and its relationship with lumbar spondylolisthesis and low back pain: the Wakayama spine study. J Pain Res 12:3521–3528. https://doi.org/10.2147/JPR.S227153
Sions JM, Elliott JM, Pohlig RT, Hicks GE (2017) Trunk muscle characteristics of the multifidi, erector spinae, psoas, and quadratus lumborum in older adults with and without chronic low back pain. J Orthop Sports Phys Ther 47:173–179. https://doi.org/10.2519/jospt.2017.7002
Takatalo J, Karppinen J, Niinimäki J et al (2012) Association of modic changes, schmorlʼs nodes, spondylolytic defects, high-intensity zone lesions, disc herniations, and radial tears with low back symptom severity among young finnish adults. Spine 37:1231–1239. https://doi.org/10.1097/BRS.0b013e3182443855
Takeuchi M, Nagamachi A, Adachi K et al (2018) Prevalence of high-intensity zones in the lumbar spine according to age and their correlation with other degenerative findings on magnetic resonance imaging. Spine Surg Relat Res 2:299–303. https://doi.org/10.22603/ssrr.2017-0071
Teraguchi M, Yoshimura N, Hashizume H et al (2015) The association of combination of disc degeneration, end plate signal change, and Schmorl node with low back pain in a large population study: the Wakayama spine study. Spine J 15:622–628. https://doi.org/10.1016/j.spinee.2014.11.012
Teraguchi M, Cheung JPY, Karppinen J et al (2020) Lumbar high-intensity zones on MRI: imaging biomarkers for severe, prolonged low back pain and sciatica in a population-based cohort. Spine J 20:1025–1034. https://doi.org/10.1016/j.spinee.2020.02.015
Videman T, Battié MC, Gibbons LE et al (2003) Associations between back pain history and lumbar MRI findings. Spine 28:582–588. https://doi.org/10.1097/01.BRS.0000049905.44466.73
Wan Q, Lin C, Li X et al (2015) MRI assessment of paraspinal muscles in patients with acute and chronic unilateral low back pain. Br J Radiol 88:20140546. https://doi.org/10.1259/bjr.20140546
Wang Z-X, Hu Y-G (2012) High-intensity zone (HIZ) of lumbar intervertebral disc on T2-weighted magnetic resonance images: spatial distribution, and correlation of distribution with low back pain (LBP). Eur Spine J 21:1311–1315. https://doi.org/10.1007/s00586-012-2240-0
Wang Z-X, Hu Y-G (2018) Factors associated with lumbar disc high-intensity zone (HIZ) on T2-weighted magnetic resonance image: a retrospective study of 3185 discs in 637 patients. J Orthop Surg 13:307. https://doi.org/10.1186/s13018-018-1010-z
Zehra U, Cheung JPY, Bow C et al (2019) Multidimensional vertebral endplate defects are associated with disc degeneration, modic changes, facet joint abnormalities, and pain. J Orthop Res 37:1080–1089. https://doi.org/10.1002/jor.24195
Brinjikji W, Diehn FE, Jarvik JG et al (2015) MRI findings of disc degeneration are more prevalent in adults with low back pain than in asymptomatic controls: a systematic review and meta-analysis. Am J Neuroradiol 36:2394–2399. https://doi.org/10.3174/ajnr.A4498
Pfirrmann CWA, Metzdorf A, Zanetti M et al (2001) Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine 26:1873–1878. https://doi.org/10.1097/00007632-200109010-00011
Jensen MC, Brant-Zawadzki MN, Obuchowski N et al (1994) Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med 331:69–73. https://doi.org/10.1056/NEJM199407143310201
Al-saeed O, Al-Jarallah K, Raeess M et al (2012) Magnetic resonance imaging of the lumbar spine in young Arabs with low back pain. Asian Spine J 6:249. https://doi.org/10.4184/asj.2012.6.4.249
Visuri T, Ulaska J, Eskelin M, Pulkkinen P (2005) Narrowing of lumbar spinal canal predicts chronic low back pain more accurately than intervertebral disc degeneration: a magnetic resonance imaging study in young finnish male conscripts. Mil Med 170:926–930. https://doi.org/10.7205/MILMED.170.11.926
Brinjikji W, Luetmer PH, Comstock B et al (2015) Systematic literature review of imaging features of spinal degeneration in asymptomatic populations. Am J Neuroradiol 36:811–816. https://doi.org/10.3174/ajnr.A4173
Cheung JPY, Luk KDK (2019) The relevance of high-intensity zones in degenerative disc disease. Int Orthop 43:861–867. https://doi.org/10.1007/s00264-018-4260-9
Jha SC, Takata Y, Abe M et al (2017) High intensity zone in lumbar spine and its correlation with disc degeneration. J Med Invest 64:39–42. https://doi.org/10.2152/jmi.64.39
Teraguchi M, Yim R, Cheung JP-Y, Samartzis D (2018) The association of high-intensity zones on MRI and low back pain: a systematic review. Scoliosis Spinal Disord 13:22. https://doi.org/10.1186/s13013-018-0168-9
Berger-Roscher N, Galbusera F, Rasche V, Wilke H-J (2015) Intervertebral disc lesions: visualisation with ultra-high field MRI at 11.7 T. Eur Spine J 24:2488–2495. https://doi.org/10.1007/s00586-015-4146-0
Allegri M, Montella S, Salici F et al (2016) Mechanisms of low back pain: a guide for diagnosis and therapy. F1000Research 5:8105
Crockett MT, Kelly BS, van Baarsel S, Kavanagh EC (2017) Modic type 1 vertebral endplate changes: injury, inflammation, or infection? Am J Roentgenol 209:167–170. https://doi.org/10.2214/AJR.16.17403
Lotz JC, Fields AJ, Liebenberg EC (2013) The role of the vertebral end plate in low back pain. Glob Spine J 3:153–163. https://doi.org/10.1055/s-0033-1347298
Rahme R, Moussa R (2008) The modic vertebral endplate and marrow changes: pathologic significance and relation to low back pain and segmental instability of the lumbar spine. Am J Neuroradiol 29:838–842. https://doi.org/10.3174/ajnr.A0925
Herlin C, Kjaer P, Espeland A et al (2018) Modic changes—their associations with low back pain and activity limitation: a systematic literature review and meta-analysis. PLOS ONE 13:e0200677. https://doi.org/10.1371/journal.pone.0200677
Wang Y, Videman T, Battié MC (2012) ISSLS prize winner: lumbar vertebral endplate lesions. Spine 37:1490–1496. https://doi.org/10.1097/BRS.0b013e3182608ac4
Lawan A, Crites Videman J, Battié MC (2021) The association between vertebral endplate structural defects and back pain: a systematic review and meta-analysis. Eur Spine J 30:2531–2548. https://doi.org/10.1007/s00586-021-06865-6
Gagnet P, Kern K, Andrews K et al (2018) Spondylolysis and spondylolisthesis: a review of the literature. J Orthop 15:404–407. https://doi.org/10.1016/j.jor.2018.03.008
Kalichman L, Kim DH, Li L et al (2009) Spondylolysis and spondylolisthesis: prevalence and association with low back pain in the adult community-based population. Spine 34:199–205. https://doi.org/10.1097/BRS.0b013e31818edcfd
Van Kleef M, Vanelderen P, Cohen SP et al (2010) 12. pain originating from the lumbar facet joints: lumbar facet pain. Pain Pract 10:459–469. https://doi.org/10.1111/j.1533-2500.2010.00393.x
Cohen SP, Raja SN (2007) Pathogenesis, diagnosis, and treatment of lumbar zygapophysial (Facet) joint pain. Anesthesiology 106:591–614. https://doi.org/10.1097/00000542-200703000-00024
Lattig F, Fekete TF, Grob D et al (2012) Lumbar facet joint effusion in MRI: a sign of instability in degenerative spondylolisthesis? Eur Spine J 21:276–281. https://doi.org/10.1007/s00586-011-1993-1
Ranger TA, Cicuttini FM, Jensen TS et al (2017) Are the size and composition of the paraspinal muscles associated with low back pain? A systematic review. Spine J 17:1729–1748. https://doi.org/10.1016/j.spinee.2017.07.002
Suri P, Fry AL, Gellhorn AC (2015) Do muscle characteristics on lumbar spine magnetic resonance imaging or computed tomography predict future low back pain, physical function, or performance? a systematic review. PM&R 7:1269–1281. https://doi.org/10.1016/j.pmrj.2015.04.016
Cooley JR, Walker BF, Ardakani ME et al (2018) Relationships between paraspinal muscle morphology and neurocompressive conditions of the lumbar spine: a systematic review with meta-analysis. BMC Musculoskelet Disord 19:351. https://doi.org/10.1186/s12891-018-2266-5
Acknowledgements
This study was funded by Radboud AI for Health (ICAI).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
van der Graaf, J.W., Kroeze, R.J., Buckens, C.F.M. et al. MRI image features with an evident relation to low back pain: a narrative review. Eur Spine J 32, 1830–1841 (2023). https://doi.org/10.1007/s00586-023-07602-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-023-07602-x