Abstract
Background
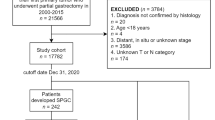
Colorectal endoscopic resection (C-ER) is spreading due to the increase of colorectal cancer (CRC) in Japan. Gastric cancer (GC) sometimes occurs after C-ER. We aimed to analyze the status of GC after C-ER using large-scale data.
Methods
We retrospectively used commercially anonymized health insurance claims data of 5.71 million patients from 2005 to 2018, and extracted 62,392 patients ≥ 50 years old who received C-ER. The incidence and risk factors of GC were analyzed. Additionally, subjects were divided into ≥ 2 cm group and < 2 cm group and risks of GC were analyzed.
Results
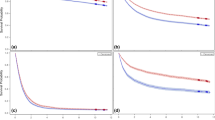
The median age (range) was 58 (50–75) years and the overall rate of GC was 0.68% (423/62,392). Multivariate analysis showed that significant risk factors for GC [odds rates (OR), 95% confidence interval (CI)] were colorectal lesion size ≥ 2 cm (1.75, 1.24–2.47, p = 0.002), age ≥ 65 y.o. (1.65, 1.31–2.07, p < 0.001), male (2.35, 1. 76–3.13, p < 0.001), diabetes mellitus (1.40, 1.02–1.92, p = 0.035), liver disease (1.54, 1.06–2.24, p = 0.025), Helicobacter pylori infection (2.10, 1.65–2.67, p < 0.001), chronic atrophic gastritis (1.58, 1.14–2.18, p = 0.006), and CRC (1.72, 1.10–2.68, p = 0.017). The rate of GC in the ≥ 2 cm was significantly higher than that in < 2 cm groups (1.17% and 0.65%, p < 0.001). According to the number of significant risk factors, the rates of GC and the hazard ratios of GC (95%CI) were 0.64% and 3.64 (2.20–6.02) and 1.95% and 11.17 (6.57–19.00) for patient with 1–2 and ≥ 3 risk factors, compared with patients without risk factors.
Conclusions
Using large-scale data, risk factors for GC, including colorecal lesions ≥ 2 cm after C-ER could be investigated.


Similar content being viewed by others
Data availability
The data analyzed in this study are available from JMDC Co., Ltd. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors upon reasonable request with the permission of JMDC Co., Ltd.
References
Cancer statistics [Internet]. Cancer Information Service, National Cancer Center (National Cancer Registry, Ministry of Health, Labour and Welfare). [Cited 2022 December 26]. Available from: https://ganjoho.jp/reg_stat/statistics/data/dl/index.html.
Cancer [Internet]. World Health Organization. [Cited 2022 July 14]. Available from: International Agency for Research on Cancer. Available from: https://www.who.int/news-room/fact-sheets/detail/cancer.
Zauber AG, Winawer SJ, O’Brien MJ, et al. Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med. 2012;366:687–96.
Tanaka S, Kashida H, Saito Y, et al. Japan gastroenterological endoscopy society guidelines for colorectal endoscopic submucosal dissection/endoscopic mucosal resection. Dig Endosc. 2020;32:219–39.
Atkin W, Wooldrage K, Brenner A, et al. Adenoma surveillance and colorectal cancer incidence: a retrospective, multicentre, cohort study. Lancet Oncol. 2017;18:823–34.
Saito Y, Oka S, Kawamura T, et al. Colonoscopy screening and surveillance guidelines. Dig Endosc. 2021;33:486–519.
Yoshida N, Naito Y, Siah KT, et al. High incidence of metachronous advanced adenoma and cancer after endoscopic resection of colon polyps ≥ 20 mm in size. Dig Endosc. 2016;28:194–202.
Lim SB, Jeong SY, Choi HS, et al. Synchronous gastric cancer in primary sporadic colorectal cancer patients in Korea. Int J Colorectal Dis. 2008;23:61–5.
Fukase K, Kato M, Kikuchi S, et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomized controlled trial. Lancet. 2008;372:392–7.
Thrift AP, Nguyen TH. Gastric cancer epidemiology. Gastrointest Endosc Clin N Am. 2021;31:425–39.
Mori G, Nakajima T, Asada K, et al. Incidence of and risk factors for metachronous gastric cancer after endoscopic resection and successful Helicobacter pylori eradication: results of a large-scale, multicenter cohort study in Japan. Gastric Cancer. 2016;19:911–8.
Ueno T, Muto M, Oya H, et al. Multiple primary cancer: an experience at the cancer institute hospital with special reference to colorectal cancer. Int J Clin Oncol. 2003;8:162–7.
Nagai K, Tanaka T, Kodaira N, et al. Data resource profile: JMDC claims database sourced from health insurance societies. J Gen Fam Med. 2021;22:118–27.
Yoshida N, Ishikawa H, Otani T, et al. Aspirin-mediated prevention of colorectal adenomas recurrence is affected by blood biochemistry and nutritional intake. Cancer Prev Res (Phila). 2022;15:837–46.
Wright FL, Green J, Canoy D, et al. Vascular disease in women: comparison of diagnoses in hospital episode statistics and general practice records in England. BMC Med Res Methodol. 2012;12:161.
Junqueira RM, Duarte EC. Hospitalizations due to ambulatory care-sensitive conditions in the Federal District, Brazil, 2008. Rev Saude Publica. 2012;46:761–8.
Thygesen S, Christiansen CF, Charistensen S, et al. The predictive value of ICD-10 diagnostic coding used to assess Charlson comorbidity index conditions in the population-based Danish national registry of patients. BMC Med Res Methodol. 2011;11:83.
van den Bussche H, Schön G, Kolonko T, et al. Patterns of ambulatory medical care utilization in elderly patients with special reference to chronic diseases and multimorbidity—results from a claims data based observational study in Germany. BMC Geriatr. 2011;11:54.
Charlson ME, Pompei P, Ales KL, et al. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–83.
Vogt A, Schmid S, Heinimann K, et al. Multiple primary tumours: challenges and approaches, a review. ESMO Open. 2017;2: e000172.
Noura S, Ohue M, Seki Y, et al. Second primary cancer in patients with colorectal cancer after a curative resection. Dig Surg. 2009;26:400–5.
Odani S, Tabuchi T, Nakata K, et al. Incidence and relative risk of metachronous second primary cancers for 16 cancer sites, Osaka, Japan, 2000–2015: population-based analysis. Cancer Med. 2022;11:507–19.
Guo J, Liu C, Pan J, et al. Relationship between diabetes and risk of gastric cancer: a systematic review and meta-analysis of cohort studies. Diabetes Res Clin Pract. 2022;187: 109866.
Ami R, Hatta W, Iijima K, et al. Factors associated with metachronous gastric cancer development after endoscopic submucosal dissection for early gastric cancer. J Clin Gastroenterol. 2017;51:494–9.
Shichijo S, Hirata Y, Niikura R, et al. Association between gastric cancer and the Kyoto classification of gastritis. J Gastroenterol Hepatol. 2017;32:1581–6.
Uemura N, Okamoto S, Yamamoto S, et al. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784–9.
Tak DH, Moon HS, Kang SH, et al. Prevalence and risk Factors of gastric adenoma and gastric cancer in colorectal cancer patients. Gastroenteol Res Pract. 2016;2016:2469521.
Zheng G, Sundquist K, Sundquist J, et al. Second primary cancers after gastric cancer, and gastric cancer as second primary cancer. Clin Epidemiol. 2021;13:515–25.
Lee SY, Yoshida N, Dohi O, et al. Differences in prevalence of lymphovascular invasion among early gastric cancers between Korea and Japan. Gut Liver. 2017;11:383–91.
Aldridge AJ, Simson JN. Histological assessment of colorectal adenomas by size. Are polyps less than 10 mm in size clinically important? Eur J Surg. 2001;167:777–81.
Park DI, Park SH, Yoo TW, et al. The prevalence of colorectal neoplasia in patients with gastric cancer: a Korean association for the study of intestinal disease (KASID) Study. J Clin Gastroenterol. 2010;44:102–5.
Joo MK, Park JJ, Lee WW, et al. Differences in the prevalence of colorectal polyps in patients undergoing endoscopic removal of gastric adenoma or early gastric cancer and in healthy individuals. Endoscopy. 2010;42:114–20.
Kim SY, Jung SW, Hyun JJ, et al. Is colonoscopic screening necessary for patients with gastric adenoma or cancer? Dig Dis Sci. 2013;58:3263–9.
Oh SY, Park DI, Yoo TW, et al. Is gastric cancer a new indication for surveillance colonoscopy? Colon cancer is increased in gastric cancer patients. Korean J Gastroenterol. 2006;47:191–7.
Zuo Y, Jing Z, Bie M, et al. Association between Helicobacter pylori infection and the risk of colorectal cancer: a systematic review and meta-analysis. Medicine (Baltim). 2020;99: e21832.
Selgrad M, Bornschein J, Kandulski A, et al. Helicobacter pylori but not gastrin is associated with the development of colonic neoplasms. Int J Cancer. 2014;135:1127–31.
Smith AM, Watson SA. Review article: gastrin and colorectal cancer. Aliment Pharmacol Ther. 2000;14:1231–47.
Kopchick JJ, Basu R, Berryman DE, et al. Covert actions of growth hormone: fibrosis, cardiovascular diseases and cancer. Nat Rev Endocrinol. 2022;18:558–73.
Colorectal cancer [Internet]. World Health Organization [Cited 2023 August 7]: Available from: https://www.who.int/news-room/fact-sheets/detail/colorectal-cancer.
Arnold M, Abnet CC, Neale RE, et al. Global burden of 5 major types of gastrointestinal cancer. Gastroenterology. 2020;159:335-49.e15.
Usui Y, Taniyama Y, Endo M, et al. Helicobacter pylori, homologous-recombination genes, and gastric cancer. N Engl J Med. 2023;388:1181–90.
Blair VR, McLeod M, Carneiro F, et al. Hereditary diffuse gastric cancer: updated clinical practice guidelines. Lancet Oncol. 2020;21:e386–97.
Lee GH, Malietzis G, Askari A, et al. Is right-sided colon cancer different to left-sided colorectal cancer? A systematic review. Eur J Surg Oncol. 2015;41:300–8.
Keum N, Giovannucci E. Global burden of colorectal cancer: emerging trends, risk factors and prevention strategies. Nat Rev Gastroenterol Hepatol. 2019;16:713–32.
Acknowledgements
The authors would like to thank all members of the Department of Molecular Gastroenterology and Hepatology, Kyoto Prefectural University of Medicine, for their help with this study.
Funding
Nothing.
Author information
Authors and Affiliations
Contributions
NY and HI designed this study, AM-M, YK, and YM performed statistical analysis, MM, YT, RH, OD, and YI had comprehensive comments about the results. All authors reviewed a paper and agreed with the contents.
Corresponding author
Ethics declarations
Conflict of interest
Naohisa Yoshida and Osamu Dohi received a research grant from Fujifilm Co. The other authors declare no conflicts of interest in association with the present study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yoshida, N., Maeda-Minami, A., Ishikawa, H. et al. Analysis of the development of gastric cancer after resecting colorectal lesions using large-scale health insurance claims data. J Gastroenterol 58, 1105–1113 (2023). https://doi.org/10.1007/s00535-023-02035-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-023-02035-1