Abstract
Background
Even after successful Helicobacter pylori eradication, primary or metachronous gastric cancers are sometimes discovered. The endoscopic features of these cancers may be modified by controlling inflammation. Characteristic findings for such lesions in terms of narrow-band imaging with magnifying endoscopy (NBI-ME) and histopathology need to be clarified to allow accurate diagnosis.
Methods
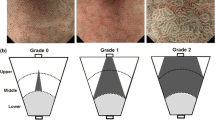
Distinctive NBI-ME characteristics were examined retrospectively in intramucosal or minimally submucosal and differentiated-type adenocarcinomas from a successful eradication group (42 patients, 50 lesions) and a non-eradicated control group (44 patients, 50 lesions) matched in age and sex. A “gastritis-like” appearance under NBI-ME was characterized by uniform papillae and/or tubular pits with a whitish border, regular or faint microvessels and unclear demarcation, resembling the adjacent noncancerous mucosa. Histological differentiation at the luminal surface of the cancer was evaluated according to Ki-67 immunoreactivity restricted at the middle or lower portion of the tubules. NBI-ME alteration was prospectively confirmed in 29 patients (30 lesions) after eradication therapy.
Results
The frequency of a “gastritis-like” appearance was 44 % (22/50) for the eradication group, which was significantly higher than the 4 % (2/50) for the control group (p < 0.001). In the eradication group, the “gastritis-like” appearance was significantly correlated with histological surface differentiation (p < 0.001). In the prospective study, NBI-ME showed changes to heterogeneous papillary microstructures in 43 % (10/23) of the lesions after successful eradication at short-term follow-up.
Conclusions
Identification of surface maturation under NBI-ME offers a promising approach for accurate diagnosis of early gastric cancers after successful eradication.




Similar content being viewed by others
References
Take S, Mizuno M, Ishiki K, Nagahara Y, Yoshida T, Yokota K, et al. The effect of eradicating Helicobacter pylori on the development of gastric cancer in patients with peptic ulcer disease. Am J Gastroenterol. 2005;100:1037–42.
Wu CY, Kuo KN, Wu MS, Chen YJ, Wang CB, Lin JT. Early Helicobacter pylori eradication decreases risk of gastric cancer in patients with peptic ulcer disease. Gastroenterology. 2009;137:1641–8.
Mabe K, Takahashi M, Oizumi H, Tsukuma H, Shibata A, Fukase K, et al. Does Helicobacter pylori eradication therapy for peptic ulcer prevent gastric cancer? World J Gastroenterol. 2009;15:4290–7.
Fukase K, Kato M, Kikuchi S, Inoue K, Uemura N, Okamoto S, Japan Gast Study Group, et al. Effect of eradication of Helicobacter pylori on incidence of metachronous gastric carcinoma after endoscopic resection of early gastric cancer: an open-label, randomised controlled trial. Lancet. 2008;372:392–7.
Kamada T, Hata J, Sugiu K, Kusunoki H, Ito M, Tanaka S, et al. Clinical features of gastric cancer discovered after successful eradication of Helicobacter pylori: results from a 9-year prospective follow-up study in Japan. Aliment Pharmacol Ther. 2005;21:1121–6.
de Vries AC, Kuipers EJ, Rauws EA. Helicobacter pylori eradication and gastric cancer: when is the horse out of the barn? Am J Gastroenterol. 2009;104:1342–5.
Yamamoto K, Kato M, Takahashi M, Haneda M, Shinada K, Nishida U, et al. Clinicopathological analysis of early-stage gastric cancers detected after successful eradication of Helicobacter pylori. Helicobacter. 2011;16:210–6.
Matsuo T, Ito M, Tatsugami M, Boda T, Takata S, Tanaka S, et al. Gastric cancer development after Helicobacter pylori eradication therapy: a new form of gastric neoplasia. Digestion. 2012;85:61–7.
Yao K, Anagnostopoulos GK, Ragunath K. Magnifying endoscopy for diagnosing and delineating early gastric cancer. Endoscopy. 2009;41:462–7.
Dinis-Ribeiro M, Areia M, de Vries AC, Marcos-Pinto R, Monteiro-Soares M, O’Connor A, MAPS Participants, et al. Management of precancerous conditions and lesions in the stomach (MAPS): guideline from the European Society of Gastrointestinal Endoscopy (ESGE), European Helicobacter Study Group (EHSG), European Society of Pathology (ESP), and the Sociedade Portuguesa de Endoscopia Digestiva (SPED). Endoscopy. 2012;44:74–94.
Nagahama T, Yao K, Maki S, Yasaka M, Takaki Y, Matsui T, et al. Usefulness of magnifying endoscopy with narrow-band imaging for determining the horizontal extent of early gastric cancer when there is an unclear margin by chromoendoscopy (with video). Gastrointest Endosc. 2011;74:1259–67.
Kimura K, Takemoto T. An endoscopic recognition of the atrophic border and its significance in chronic gastritis. Endoscopy. 1969;3:87–97.
Japanese Gastric Cancer Association. Japanese Classification of Gastric Carcinoma, 3rd English Edition. Gastric Cancer. 2011;14:101–12.
Lauwers GY, Carneiro F, Graham DY, Curado MP, Franceschi S, Montgomery E, et al. Gastric carcinoma. In: Bosman FT, Carneiro F, Hruban RH, Theise ND, editors. WHO Classification of tumours of the digestive system. 4th ed. Lyon: IARC Press; 2010. p. 48–58.
Kobayashi M, Takeuchi M, Ajioka Y, Hashimoto S, Sato A, Narisawa R, et al. Mucin phenotype and narrow-band imaging with magnifying endoscopy for differentiated-type mucosal gastric cancer. J Gastroenterol. 2011;46:1064–70.
Kaise M, Kato M, Urashima M, Arai Y, Kaneyama H, Kanzazawa Y, et al. Magnifying endoscopy combined with narrow-band imaging for differential diagnosis of superficial depressed gastric lesions. Endoscopy. 2009;41:310–5.
Yagi K, Nozawa Y, Endou S, Nakamura A. Diagnosis of early gastric cancer by magnifying endoscopy with NBI from viewpoint of histological imaging: mucosal patterning in terms of white zone visibility and its relationship to histology. Diagn Ther Endosc. 2012;. doi:10.1155/2012/954809.
Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161–81.
Kobayashi M, Watanabe H, Ajioka Y, Honma T, Asakura H. Effect of K-ras mutation on morphogenesis of colorectal adenomas and early cancers: relationship to distribution of proliferating cells. Hum Pathol. 1996;27:1042–9.
Nozawa Y, Nishikura K, Ajioka Y, Aoyagi Y. Relationship between alpha-methylacyl-coenzyme: a racemase expression and mucin phenotype in gastric cancer. Hum Pathol. 2012;43:878–87.
Kato M, Kaise M, Yonezawa J, Toyoizumi H, Yoshimura N, Yoshida Y, et al. Magnifying endoscopy with narrow-band imaging achieves superior accuracy in the differential diagnosis of superficial gastric lesions identified with white-light endoscopy: a prospective study. Gastrointest Endosc. 2010;72:523–9.
Ezoe Y, Muto M, Uedo N, Doyama H, Yao K, Oda I, et al. Magnifying narrowband imaging is more accurate than conventional white-light imaging in diagnosis of gastric mucosal cancer. Gastroenterology. 2011;141:2017–25.
Kobayashi M, Narisawa R, Sato Y, Takeuchi M, Aoyagi Y. Self-limiting risk of metachronous gastric cancers after endoscopic resection. Dig Endosc. 2010;22:169–73.
Endoh Y, Tamura G, Watanabe H, Ajioka Y, Motoyama T. The common 18-base pair deletion at codons 418–423 of the E-cadherin gene in differentiated-type adenocarcinomas and intramucosal precancerous lesions of the stomach with the features of gastric foveolar epithelium. J Pathol. 1999;189:201–6.
Endoh Y, Tamura G, Motoyama T, Ajioka Y, Watanabe H. Well-differentiated adenocarcinoma mimicking complete-type intestinal metaplasia in the stomach. Hum Pathol. 1999;30:826–32.
Noffsinger AE, Miller MA, Cusi MV, Fenoglio-Preiser CM. The pattern of cell proliferation in neoplastic and nonneoplastic lesions of ulcerative colitis. Cancer. 1996;78:2307–12.
Mikami T, Yoshida T, Akino F, Motoori T, Yajima M, Okayasu I. Apoptosis regulation differs between ulcerative colitis–associated and sporadic colonic tumors. Am J Clin Pathol. 2003;119:723–30.
Lynch DA, Mapstone NP, Clarke AMT, Sobala GM, Jackson P, Morrison L, et al. Cell proliferation in Helicobacter pylori associated gastritis and the effect of eradication therapy. Gut. 1995;36:346–50.
Cahill RJ, Kilgallen C, Beattie S, Hamilton H, O’Morain C. Gastric epithelial cell kinetics in the progression from normal mucosa to gastric carcinoma. Gut. 1996;38:177–81.
Scotiniotis IA, Rokkas T, Furth EE, Rigas B, Shiff SJ. Altered gastric epithelial cell kinetics in Helicobacter pylori-associated intestinal metaplasia: implication for gastric carcinogenesis. Int J Cancer. 2000;85:192–200.
Shiotani A, Iishi H, Ishiguro S, Tatsuta M, Nakae Y, Merchant JL. Epithelial cell turnover in relation to ongoing damage of the gastric mucosa in patients with early gastric cancer: increase of cell proliferation and paramalignant lesions. J Gastroenterol. 2005;40:337–44.
Iijima K, Koike T, Sekine H, Abe Y, Asanuma K, Ara N, et al. Sustained epithelial proliferation in a functionally irreversible fundic mucosa after Helicobacter pylori eradication. J Gastroenterol. 2009;44:47–55.
Ito M, Tanaka S, Takata S, Oka S, Imagawa S, Ueda H, et al. Morphological changes in human gastric tumours after eradication therapy of Helicobacter pylori in a short-term follow-up. Aliment Pharmacol Ther. 2005;21:559–66.
Kato M, Asaka M, Ono S, Nakagawa M, Nakagawa S, Shimizu Y, et al. Eradication of Helicobacter pylori for primary gastric cancer and secondary gastric cancer after endoscopic mucosal resection. J Gastroenterol. 2007;42(Suppl 17):16–20.
Acknowledgments
This work was supported in part by the Japan Society for the Promotion of Science KAKENHI Grant Number 24591025.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kobayashi, M., Hashimoto, S., Nishikura, K. et al. Magnifying narrow-band imaging of surface maturation in early differentiated-type gastric cancers after Helicobacter pylori eradication. J Gastroenterol 48, 1332–1342 (2013). https://doi.org/10.1007/s00535-013-0764-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-013-0764-7