Summary
Background
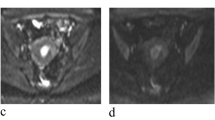
This study aimed to investigate the efficacy of quantitative apparent diffusion coefficient (ADC) measurement in the differential diagnosis of the uterine endometrial cavity tumors (UECT).
Methods
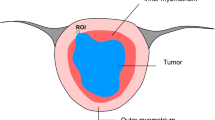
This study included consecutive 36 female patients with UECT. The groups were formed as follows: group 1, patients with endometrial carcinoma; group 2, patients with endometrial polyp; group 3, patients with submucosal leiomyoma; and additionally group A, patients with malignant lesion; group B patients with benign lesion. Tumoral and outer myometrial ADC values were measured and the tumor–myometrium ADC ratios were calculated in all patients. Kolmogorov–Smirnov tests were used to test the normality of the data distributions. The data were not normally distributed, and therefore, nonparametric tests were performed. The cut-off values, sensitivity, and specificity were determined by the receiver operating characteristic analysis.
Results
The mean ADC value and ratio of group 1 were lower than that of group 2 and 3, and the mean ADC value and ratio of group 3 were lower than that of group 2. The sensitivity and specificity for group 1, 2, and 3 were as follows: 90, 100, and 66.7 %; and 81.8, 88, and 58.8 %, respectively, in terms of the ADC values; 90, 100 and 67 %; and 77.3, 96, and 64.7 % in terms of the ADC ratios. Statistically significant differences were demonstrated between group A and B in terms of mean tumoral ADC values and ratios. Sensitivity and specificity were found to be 90 and 81.8 %, respectively, in terms of the ADC values. The sensitivity and specificity were found to be 90 and 77.3 %, respectively, in terms of the ADC ratios.
Conclusions
ADC values and ratios represent a promising parameter in the determination of the tumoral lesions in patients with UECT.







Similar content being viewed by others
References
Takeuchi M, Matsuzaki K, Uehara H, Yoshida S, Nishitani H, Shimazu H. Pathologies of the uterine endometrial cavity: usual and unusual manifestations and pitfalls on magnetic resonance. Eur Radiol. 2005;15(11):2244–55.
Aubé C, Racineux PX, Lebigot J, et al. [Diagnosis and quantification of hepatic fibrosis with diffusion weighted MR imaging: preliminary results]. J Radiol. 2004;85(3):301–6.
Fujii S, Matsusue E, Kigawa J, et al. Diagnostic accuracy of the apparent diffusion coefficient in differentiating benign from malignant uterine endometrial cavity lesions: initial results. Eur Radiol. 2008;18(2):384–9.
Tong DC, Albers GW. Diffusion and perfusion magnetic resonance imaging for the evaluation of acute stroke: potential use in guiding thrombolytic therapy. Curr Opin Neurol. 2000;13(1):45–50.
Guo Y, Cai YQ, Cai ZL, et al. Differentiation of clinically benign and malignant breast lesions using diffusion-weighted imaging. J Magn Reson Imaging. 2002;16(2):172–8.
Woodhams R, Matsunaga K, Kan S, et al. ADC mapping of benign and malignant breast tumors. Magn Reson Med Sci. 2005;4(1):35–42.
Rubesova E, Grell AS, De Maertelaer V, Metens T, Chao SL, Lemort M. Quantitative diffusion imaging in breast cancer: a clinical prospective study. J Magn Reson Imaging. 2006;24(2):319–24.
Patel J, Sigmund EE, Rusinke H, Oei M, Babbs JS, Taouli B. Diagnosis of cirrhosis with intravoxel incoherent motion diffusion MRI and dynamic contrast-enhanced MRI alone and in combination: preliminary experience. J Magn Reson Imaging. 2010;31(3):589–600.
Sener RN. Diffusion MRI: apparent diffusion coefficient (ADC) values in the normal brain, and a classification of brain disorders based on ADC values. Comput Med Imaging Graph. 2001;25(4):299–326.
Mukherji SK, Chenevert TL, Castillo M. Diffusion-weighted magnetic resonance imaging. J Neuroophthalmol. 2002;22(2):118–22.
Sugahara T, Korogi Y, Kochi M, et al. Usefulness of diffusion-weighted MRI with echo-planar technique in the evaluation of cellularity in gliomas. J Magn Reson Imaging. 1999;9(1):53–60.
Gauvain KM, McKinstry RC, Mukherjee P, et al. Evaluating pediatric brain tumor cellularity with diffusion-tensor imaging. AJR Am J Roentgenol. 2001;177(2):449–54.
Guo AC, Cummings TJ, Dash RC, Provenzale JM. Lymphomas and high-grade astrocytomas: comparison of water diffusibility and histologic characteristics. Radiology. 2002;224(1):177–83.
Rumboldt Z, Camacho DL, Lake D, Welsh CT, Castillo M. Apparent diffusion coefficients for differentiation of cerebellar tumors in children. AJNR Am J Neuroradiol. 2006;27(6):1362–9.
Hayashida Y, Hirai T, Morishita S, et al. Diffusion-weighted imaging of metastatic brain tumors: comparison with histologic type and tumor cellularity. AJNR Am J Neuroradiol. 2006;27(7):1419–25.
Silverberg SG, Kurman RJ. Tumors of the uterine corpus and gestational disease. In: Rosai J, Aovin trophoblastic L, editors. Atlas of tumor pathology. Vol. 3. Washington DC: Armed Forces Institute of Pathology; 2002. pp. 113–51.
Shimada K, Ohashi I, Kasahara I, et al. Differentiation between completely hyalinized uterine leiomyomas and ordinary leiomyomas: three-phase dynamicmagnetic resonance imaging (MRI) vs. diffusion-weighted MRI with very small b-factors. J Magn Reson Imaging. 2004;20(1):97–104.
Liapi E, Kamel IR, Bluemke DA, Jacobs MA, Kim HS. Assessment of response of uterine fibroids and myometrium to embolization using diffusion-weighted echoplanar MR imaging. J Comput Assist Tomogr. 2005;29(1):83–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karakas, O., Karakas, E., Dogan, F. et al. Diffusion-weighted MRI in the differential diagnosis of uterine endometrial cavity tumors. Wien Klin Wochenschr 127, 266–273 (2015). https://doi.org/10.1007/s00508-015-0709-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-015-0709-7