Abstract
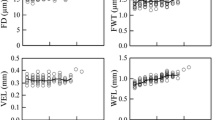
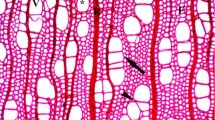
Changes in age of the hydraulic architecture of Ulmus minor and U. minor × U. pumila juvenile wood were studied and related to tolerance to Dutch elm disease (DED). The xylem vessel dimensions and the conductivity to air of 2- to 7-year-old branches were analyzed and quantified. No obvious differences in vessel length distribution and conductivity were found to explain differences in DED tolerance among the U. minor clones, or, at the taxon level, the higher DED tolerance of U. minor × U. pumila. Among the U. minor clones, the more susceptible one had wider vessels and a higher maximum vessel diameter than the more tolerant clone. Relations between vessel lengths, vessel diameters and branch sizes were highly significant, and varied between taxa. The diameter and length of vessels increased with age, and average values stabilized 1–2 years earlier for U. minor than for U. minor × pumila. Mean maximum vessel length was significantly higher in U. minor and increased more with age and maximum vessel diameter than in U. minor × pumila. With each 0.2 m increase in height up the stem, conductivities for U. minor and U. minor × U. pumila decreased by 59 and 50 %, respectively, probably due to shortening of the vessels. The implications of xylem structure for the means of pathogen movement and resistance to DED are discussed.




Similar content being viewed by others
References
Ahmed SA, Chun SK (2011) Permeability of Tectona grandis L. as affected by wood structure. Wood Sci Technol 45:487–500
Banfield WM (1941) Distribution by the sap stream of spores of three fungi that induce vascular wilt diseases of elm. J Agric Res 62:637–681
Boege K, Dirzo R, Siemens D, Brown P (2007) Ontogenetic switches from plant resistance to tolerance: minimizing costs with age? Ecol Lett 10:177–187
Chatelet DS, Wistrom CM, Purcell AH, Rost TL, Matthews MA (2011) Xylem structure of four grape varieties and 12 alternative hosts to the xylem-limited bacterium Xylella fastidious. Ann Bot 108:73–85
Cohen S, Bennink J, Tyree M (2003) Air method measurements of apple vessel length distributions with improved apparatus and theory. J Exp Bot 389:1889–1897
Elgersma DM (1970) Length and diameter of xylem vessels as a factors in resistance of elms to Ceratocystis ulmi. Neth J Plant Pathol 76:179–182
Ellmore GS, Ewers FW (1985) Hydraulic conductivity in trunk xylem of elm, Ulmus americana. IAWA Bull 4:303–307
Et-Touil A, Rioux D, Mathieu FM, Bernier L (2005) External symptoms and histopathological changes following inoculation of elms putatively resistant to Dutch elm disease with genetically close strains of Ophiostoma. Can J Bot 83:656–667
Ewers FW, Fisher JB (1989) Techniques for measuring vessel lengths and diameters in stems of woody plants. Am J Bot 76:645–656
Ewers FW, Fisher JB, Chiu ST (1990) A survey of vessel dimensions in stems of tropical lianas and other growth forms. Oecologia 84:544–552
Inch SA, Ploetz RC (2012) Impact of laurel wilt, caused by Raffaelea lauricola, on xylem function in avocado, Persea americana. For Path 42:239–245
Kitin P, Funada R, Sano Y, Beeckman H, Ohtani J (1999) Variations in the lengths of fusiform cambial cells and vessel elements in Kalopanax pictus. Ann Bot 84:621–632
Landwehr VR, Phillipsen WJ, Accerno ME, Hatch R (1981) Attraction of the native elm bark beetle to American elm after the pruning of branches. J Econ Entomol 74:577–580
Lens F, Sperry JS, Christman MA, Choat B, Rabaey D, Jansen S (2011) Testing hypotheses that link wood anatomy to cavitation resistance and hydraulic conductivity in the genus Acer. New Phytol 190:709–723
Li X, Wu HX, Southerton SG (2011) Transcriptome profiling of wood maturation in Pinus radiata identifies differentially expressed genes with implications in juvenile and mature wood variation. Gene 487:62–71
Loepfe L, Martinez-Vilalta J, Piñol J, Mencuccini M (2007) The relevance of xylem network structure for plant hydraulic efficiency and safety. J Theor Biol 247:788–803
MacHardy WE, Beckman CH (1973) Water relations in American elm infected with Ceratocystis ulmi. Phytopathology 63:98–103
Martín JA, Solla A, Woodward S, Gil L (2007) Detection of differential changes in lignin composition of elm xylem tissues inoculated with Ophiostoma novo-ulmi using Fourier transform-infrared spectroscopy. For Path 37:187–191
Martín JA, Solla A, Coimbra MA, Gil L (2008) Metabolic fingerprinting allows discrimination between Ulmus pumila and U. minor, and between U. minor clones of different susceptibility to Dutch elm disease. For Path 38:244–256
Martín JA, Solla A, Esteban LG, de Palacios P, Gil L (2009) Bordered pit and ray morphology involvement in elm resistance to Ophiostoma novo-ulmi. Can J For Res 39:420–429
Martín JA, Solla A, Gil L, García-Vallejo MC (2010) Phenological and histochemical changes of Ulmus minor due to root absorption of phenol: implications for resistance to DED. Environ Exp Bot 69:175–182
Martín JA, Solla A, García-Vallejo MC, Gil L (2012) Chemical changes in Ulmus minor xylem tissue after salicylic acid or carvacrol treatments are associated with enhanced resistance to Ophiostoma novo-ulmi. Phytochemistry 83:104–109
McNabb HS Jr, Heybroek HM, Mcdonald WL (1970) Anatomical factors in resistance to Dutch elm disease. Neth J Plant Pathol 76:196–204
Melching JB, Sinclair WA (1975) Hydraulic conductivity of stem internodes relates to resistance of American elms to Ceratocystis ulmi. Phytopathology 65:643–645
Miller HJ, Elgersma DM (1976) The growth of aggressive and non-aggressive strains of Ophiostoma ulmi in susceptible and resistant elms, a scanning electron microscopical study. Neth J Plant Pathol 82:51–65
Newbanks D, Bosh A, Zimmerman MH (1983) Evidence for xylem dysfunction by embolization in Dutch elm disease. Phytopathology 73:1060–1063
Pomerleau R, Mehran AR (1966) Distribution of spores of Ceratocystis ulmi labelled with phosphorus-32 in green shoots and leaves of Ulmus americana. Nat Can 93:577–582
Shigo AL, Tippet JT (1981) Compartmentalization of American elm tissues infected by Ceratocystis ulmi. Plant Dis 65:715–718
Sinclair WA, Zahand JP, Melching JB (1975) Anatomical markers for resistance of Ulmus americana to Ceratocystis ulmi. Phytopathology 65:349–352
Smalley EB, Guries RP (2000) Asian elms: sources of disease and insect resistance. In: Dunn CP (ed) The elms: breeding, conservation and disease management. Kluwer Academic Publishers, Boston, pp 215–230
Solla A, Gil L (2002a) Influence of water stress on Dutch elm disease symptoms in Ulmus minor Miller. Can J Bot 80:810–817
Solla A, Gil L (2002b) Xylem vessel diameter as a factor in resistance of Ulmus minor to Ophiostoma novo-ulmi. For Pathol 32:123–134
Solla A, Bohnens J, Collin E, Diamandis S, Franke A, Gil L, Burón M, Santini A, Mittempergher L, Pinon J, Broek AV (2005a) Screening European elms for resistance to Ophiostoma novo-ulmi. For Sci 51:134–141
Solla A, Martín JA, Corral P, Gil L (2005b) Seasonal changes in wood formation of Ulmus pumila and U. minor and its relation with Dutch elm disease. New Phytol 166:1025–1034
Solla A, Martín JA, Ouellette G, Gil L (2005c) Influence of plant age on symptom development in Ulmus minor following inoculation by Ophiostoma novo-ulmi. Plant Dis 89:1035–1040
Solla A, Dacasa MC, Nasmith C, Hubbes M, Gil L (2008) Analysis of Spanish populations of Ophiostoma ulmi and O. novo-ulmi using phenotypic characteristics and RAPD markers. Plant Pathol 57:33–44
Sperry JS, Hacke UG, Pittermann J (2006) Size and function in conifer tracheids and angiosperm vessels. Am J Bot 93:1490–1500
Sutherland ML, Pearson S, Brasier CM (1997) The influence of temperature and light on defoliation levels of elm by Dutch elm disease. Phytopathology 87:576–581
Tainter FH (2001) Anatomy and physiology of vascular tissues of woody plants. In: Ash CL (ed) Shade tree wilt diseases. APS Press, Minnesota, pp 1–14
Thorne ET, Young BM, Young GM, Stevenson JF, Labavitch JM, Matthews MA, Rost TL (2006) The structure of xylem vessels in grapevine (Vitaceae) and a possible passive mechanism for the systemic spread of bacterial disease. Am J Bot 93:497–504
Tyree MT (1993) Theory of vessel-length determination: the problem of nonrandom vessel distribution. Can J Bot 71:297–302
Tyree MT, Zimmermann MH (2002) Xylem structure and the ascent of sap. Springer, Verlag 285 p
Van Alfen N, Turner N (1975) Influence of a Ceratocystis ulmi toxin on water relations of elm (Ulmus americana). Plant Physiol 55:312–316
Ye ZH (2002) Vascular tissue differentiation and pattern formation in plants. Annu Rev Plant Biol 53:183–202
Zimmermann MH, Jeje A (1981) Vessel-length distribution in stems of some American woody plants. Can J Bot 59:1882–1892
Acknowledgments
The authors wish to thank Margarita Burón and David López for their technical support, and to Jane McGrath for the language editing. This study was conducted within the framework of an agreement between DGMN and PF (Ministerio de Agricultura, Alimentación y Medio Ambiente) and ETSI Montes (Universidad Politécnica de Madrid).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by W. Osswald.
Rights and permissions
About this article
Cite this article
Martín, J.A., Solla, A., Ruiz-Villar, M. et al. Vessel length and conductivity of Ulmus branches: ontogenetic changes and relation to resistance to Dutch elm disease. Trees 27, 1239–1248 (2013). https://doi.org/10.1007/s00468-013-0872-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-013-0872-2