Abstract
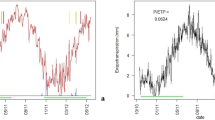
Respiration rates are reported to increase exponentially with temperature. Respiration rates of woody tissues are commonly measured as CO2 efflux rates (\(F_{{\rm CO}_{\rm 2}}\)) from that tissue. However, this paper describes clear variations in stem \(F_{{\rm CO}_{\rm 2}}\) that were not related to temperature for the case of a young beech (Fagus sylvatica L.) and oak (Quercus robur L.) tree during the dormant season. The CO2 concentration ([CO2]) in the xylem of the beech tree showed similar temperature-independent variations. The trees were grown in a growth chamber in which radiation patterns and temperature were kept constant. \(F_{{\rm CO}_{\rm 2}}\) was measured with an IRGA connected to cuvettes surrounding a stem segment. Xylem [CO2] was measured in situ using a CO2 microelectrode. Depressions in \(F_{{\rm CO}_{\rm 2}}\) and [CO2] occurred during the light period, despite equal temperatures in the light and dark period. Explanations found in literature for discrepancies in the exponential relationship between temperature and \(F_{{\rm CO}_{\rm 2}}\) are the influence of (1) sap flow or (2) decreased cell water content. However, (1) the variations were observed in the dormant season, when no sap flow was observed yet, and (2) reduced cell water content was not likely to be apparent as differences in stem transpiration rates between the dark and light period were not significant. Hence, previously formulated theories failed to explain our results. This work therefore provides a new ground for discussion on other possible causes of daytime depressions in \(F_{{\rm CO}_{\rm 2}}\). One might be the refixation of respired CO2 by corticular photosynthesis in the stem parts adjacent to the stem segment enclosed by the cuvette.





Similar content being viewed by others
References
Amthor JS (1984) The role of maintenance respiration in plant growth. Plant Cell Environ 7:561–569
Armstrong W, Armstrong J (2005) Stem photosynthesis not pressurized ventilation is responsible for light-enhanced oxygen supply to submerged roots of alder (Alnus glutinosa). Ann Bot 96:591–612
Bosc A, De Grandcourt A, Loustau D (2003) Variability of stem and branch maintenance respiration in a Pinus pinaster tree. Tree Physiol 23:227–236
Cernusak LA, Marshall JD (2000) Photosynthetic refixation in branches of Western White Pine. Funct Ecol 14:300–311
Ceschia E, Damesin C, Lebaube S, Pontailler JY, Dufrêne E (2002) Spatial and seasonal variations in stem respiration of beech trees (Fagus sylvatica). Ann For Sci 59:801–812
Coe JM, McLaughlin SB (1980) Winter season corticular photosynthesis in Cornus florida, Acer rubrum, Quercus alba, and Liriodendron tulipifera. For Sci 26:561–566
Damesin C (2003) Respiration and photosynthesis characteristics of current-year stems of Fagus sylvatica: from the seasonal pattern to an annual balance. New Phytol 158:465–475
Damesin C, Ceschia E, Le Goff N, Ottorini JM, Dufrêne E (2002) Stem and branch respiration of beech: from tree measurements to estimations at the stand level. New Phytol 153:159–172
Edwards NT, Hanson PJ (1996) Stem respiration in a closed-canopy upland oak forest. Tree Physiol 16:433–439
Edwards NT, McLaughlin SB (1978) Temperature-independent diel variations of respiration rates in Quercus alba and Liriodendron tulipifera. Oikos 31:200–206
Edwards NT, Shugart HH Jr, McLaughlin SB, Harris WF, Reichle DE (1981) Carbon metabolism in terrestrial ecosystems. In: Reichle DE (ed) Dynamic properties of forest ecosystems. Cambridge University Press, Cambridge, pp 499–536
Edwards NT, Wullschleger SD (2000) Carbon dioxide efflux rates from stems of mature Quercus prinus L. and Acer rubrum L. trees do not appear to be affected by sapflow rates. J Sust For 10:125–131
Eklund L (1990) Endogenous levels of oxygen, carbon dioxide and ethylene in stems of Norway spruce trees during one growing season. Trees 4:150–154
Eklund L (1993) Seasonal variations of O2, CO2, and ethylene in oak and maple stems. Can J For Res 23:2608–2610
Eklund L, Lavigne MB (1995) Restricted lateral gas movement in Pinus strobus branches. Trees 10:83–85
Foote K, Schaedle M (1976) Physiological characteristics of photosynthesis and respiration in stems of Populus tremuloides Michx. Plant Physiol 58:91–94
Gansert D (2004) A new type of cuvette for the measurement of daily variation of CO2 efflux from stems and branches in controlled temperature conditions. Trees 18:221–229
Hari P, Nygren P, Korpilathi E (1991) Internal circulation of carbon within a tree. Can J For Res 21:514–515
Janssens IA, Lankreijer H, Matteucci G, Kowalski AS, Buchmann N, Epron D, Pilegaard K, Kutsch W, Longdoz B, Grunwald T, Montagnani L, Dore S, Rebmann C, Moors EJ, Grelle A, Rannik U, Morgenstern K, Oltchev S, Clement R, Gudmundsson J, Minerbi S, Berbigier P, Ibrom A, Moncrieff J, Aubinet M, Bernhofer C, Jensen NO, Vesala T, Granier A, Schulze ED, Lindroth A, Dolman AJ, Jarvis PG, Ceulemans R, Valentini, R (2001) Productivity overshadows temperature in determining soil and ecosystem respiration across European forests. Global Change Biol 7:269–278
Jones HG (1992) Plants and microclimate: a quantitative approach to plant physiology. Cambridge University Press, Cambridge
Kaipiainen LK, Sofronova GI, Hari P, Yalynskaya EE (1998) The role of xylem in CO2 exchange in Pinus sylvestris woody stems. Russ J Plant Physiol 45:500–505
Kakubari, Y (1988) Diurnal and seasonal fluctuations in the bark respiration of standing Fagus sylvatica trees at Solling, West Germany. J Jpn For Soc 70:64–70
Larcher W (1995) Physiological plant ecology. Springer, New York
Lavigne MB (1987) Differences in stem respiration responses to temperature between balsam fir trees in thinned and unthinned stands. Tree Physiol 3:225–233
Lavigne MB, Franklin SE, Hunt ER (1996) Estimating stem maintenance respiration rates of dissimilar balsam fir stands. Tree Physiol 16:687–695
Lavigne MB, Ryan MG (1997) Growth and maintenance respiration rates of aspen, black spruce and jack pine stems at northern and southern BOREAS sites. Tree Physiol 17:543–551
Levy PE, Jarvis PG (1998) Stem CO2-fluxes in two Sahelian shrub species (Guiera senegalensis and Combretum micranthum). Funct Ecol 12:107–116
Levy PE, Meir P, Allen SJ, Jarvis PG (1999) The effect of aqueous transport of CO2 in xylem sap on gas exchange in woody plants. Tree Physiol 19:53–58
Little CHA (1975) Inhibition of cambial activity in Abies balsamea by internal water stress—role of abscisic acid. Can J Bot 53:3041–3050
Lockhart JA (1965) An analysis of irreversible plant cell elongation. J Theor Biol 8:264–275
Maier CA, Zarnoch SJ, Dougherty PM (1998) Effects of temperature and tissue nitrogen on dormant season stem and branch maintenance respiration in a young loblolly pine (Pinus taeda) plantation. Tree Physiol 18:11–20
Martin TA, Teskey RO, Dougherty PM (1994) Movement of respiratory CO2 in stems of loblolly pine (Pinus taeda L.) seedlings. Tree Physiol 14:481–495
McCree KJ (1970) An equation for the rate of respiration of white clover plants grown under controlled conditions. In: Setlik I (ed) Prediction and measurement of photosynthetic productivity. PUDOC, Wageningen, pp 221–229
McGuire MA, Teskey RO (2002) Microelectrode technique for in situ measurement of carbon dioxide concentrations in xylem sap of trees. Tree Physiol 22:807–811
McGuire MA, Teskey RO (2004) Estimating stem respiration in trees by a mass balance approach that accounts for internal and external fluxes of CO2. Tree Physiol 24:571–578
Negisi K (1972) Diurnal fluctuation of CO2 release from the bark of a standing Magnolia obovota tree. J Jpn For Soc 54:257–263
Negisi K (1978) Daytime depression in bark respiration and radial shrinkage in stem of a standing young Pinus densiflora tree. J Jpn For Soc 60:380–382
Pfanz H, Aschan G, Langenfeld-Heyser R, Wittmann C, Loose M (2002) Ecology and ecophysiology of tree stems: corticular and wood photosynthesis. Naturwissenschaften 89:147–162
Pilarski J (1994) Diffusion of carbon-dioxide through the cork and stomata in lilac. Acta Physiol Plant 16:137–140
Ryan MG (1990) Growth and maintenance respiration in stems of Pinus contorta and Picea engelmannii. Can J For Res 20:48–57
Ryan MG (1991) Effects of climate change on plant respiration. Ecol Appl 1:157–167
Ryan MG, Gower ST, Hubbard RM, Waring RH, Gholz HL, Cropper WP, Running, SW (1995) Woody tissue maintenance respiration of 4 conifers in contrasting climates. Oecologia 101:133–140
Schönherr J, Ziegler H (1980) Water permeability of Betula periderm. Planta 147:345–354
Sprugel DG (1990) Components of woody-tissue respiration in young Abies amabilis trees. Trees 4:88–98
Sprugel DG, Benecke U (1991) Measuring woody-tissue respiration and photosynthesis. In: Lassoie JP, Hinckley TM (eds) Techniques and approaches in forest tree ecophysiology. CRC Press, Florida, pp 329–355
Steinberg S, van Bavel CHM, McFarland MJ (1989) A gauge to measure mass flow rate of sap in stems and trunks of woody plants. J Am Soc Hortic Sci 114:466–472
Stockfors J (2000) Temperature variations and distribution of living cells within tree stems: implications for stem respiration and scaling up. Tree Physiol 20:1057–1062
Stockfors J, Linder S (1998) Effect of nitrogen on the seasonal course of growth and maintenance respiration in stems of Norway spruce trees. Tree Physiol 18:155–166
Teskey RO, McGuire MA (2002) Carbon dioxide transport in xylem causes errors in estimation of rates of respiration in stems and branches of trees. Plant Cell Environ 25:1571–1577
Teskey RO, McGuire MA (2005) CO2 transported in xylem sap affects CO2 efflux from Liquidambar styraciflua and Platanus occidentalis stems, and contributes to observed wound respiration phenomena. Trees 19:357–362
Thornley JHM, Johnson IR (1990) Plant and crop modelling. Clarendon Press, Oxford
van Bavel MG, van Bavel CHM (1990) Dynagage installation and operation manual. Dynamax Inc, Texas
Wang WJ, Yang FJ, Zu YG, Wang HM, Takagi K, Sasa K, Koike T (2003) Stem respiration of a larch (Larix gmelini) plantation in Northeast China. Acta Bot Sin 45:1387–1397
Wittmann C, Aschan G, Pfanz H (2001) Leaf and twig photosynthesis of young beech (Fagus sylvatica) and aspen (Populus tremula) trees grown under different light regime. Basic Appl Ecol 2:145–154
Acknowledgements
The authors wish to thank the Special Research Fund (B.O.F.) of Ghent University for the Ph.D. funding granted to the first author. We are also indebted to Mary Anne McGuire for her helpful answers to our questions and Philip Deman for his accurate and enthusiastic technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Pfanz
Rights and permissions
About this article
Cite this article
Saveyn, A., Steppe, K. & Lemeur, R. Report on non-temperature related variations in CO2 efflux rates from young tree stems in the dormant season. Trees 22, 165–174 (2008). https://doi.org/10.1007/s00468-006-0099-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-006-0099-6