Abstract
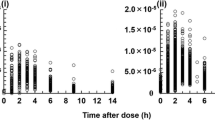
In adult renal transplant recipients the Neoral area under the curve (AUC) displays less inter- and intra- individual variability than Sandimmune, and those renal transplant recipients with reduced intra-individual variability of the AUC have a lower risk for chronic rejection. As variability of Neoral pharmacokinetic (Pk) parameters has not been investigated in pediatric renal transplant recipients, we retrospectively analyzed 453 Pk profiles in 14 pediatric patients who were switched from Sandimmune to Neoral and compared the inter- and intra-individual variability of the Pk profiles on both formulations. After the switch, we observed less inter- and intra-individual variability of AUC, the 2-h concentration, and the oral clearance. As clearance with both formulations is supposedly equal, the significantly lower intra-individual variability of oral clearance is most likely an effect of less variable absorption. While the lower inter-individual variability of the Pk parameters suggests increased success in keeping cyclosporine concentrations on target, the lower intra-individual variability leads to the hypothesis that with Neoral, a lower incidence of chronic rejection might be achieved.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 8 February 2000 / Revised: 17 May 2000 / Accepted: 22 May 2000
Rights and permissions
About this article
Cite this article
Portman, R., Meier-Kriesche, H., Swinford, R. et al. Reduced variability of Neoral pharmacokinetic studies in pediatric renal transplantation. Pediatr Nephrol 15, 2–6 (2000). https://doi.org/10.1007/s004670000435
Issue Date:
DOI: https://doi.org/10.1007/s004670000435