Abstract
Background
The impact of disorders of fluid balance, including the pathologic state of fluid overload in sick children has become increasingly apparent. With this understanding, there has been a shift from application of absolute thresholds of fluid accumulation to an appreciation of the intricacies of fluid balance, including the impact of timing, trajectory, and disease pathophysiology.
Methods
The 26th Acute Disease Quality Initiative was the first to be exclusively dedicated to pediatric and neonatal acute kidney injury (pADQI). As part of the consensus panel, a multidisciplinary working group dedicated to fluid balance, fluid accumulation, and fluid overload was created. Through a search, review, and appraisal of the literature, summative consensus statements, along with identification of knowledge gaps and recommendations for clinical practice and research were developed.
Conclusions
The 26th pADQI conference proposed harmonized terminology for fluid balance and for describing a pathologic state of fluid overload for clinical practice and research. Recommendations include that the terms daily fluid balance, cumulative fluid balance, and percent cumulative fluid balance be utilized to describe the fluid status of sick children. The term fluid overload is to be preserved for describing a pathologic state of positive fluid balance associated with adverse events. Several recommendations for research were proposed including focused validation of the definition of fluid balance, fluid overload, and proposed methodologic approaches and endpoints for clinical trials.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, the deleterious impact of fluid overload in critically ill patients across the age spectrum has become clear [1,2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20]. Evidence first described in children has led the field in highlighting the adverse outcomes associated with excessive fluid accumulation in sick children. The association between greater positive fluid balance at the time of initiation of continuous kidney replacement therapy (CKRT) and increased mortality was first described in 2001 [8]. This association has been demonstrated repeatedly across a spectrum of neonatal and pediatric populations, including those who are critically ill, receiving mechanical ventilation, on extracorporeal life support, and undergoing congenital heart surgery. Thus, the importance of documenting and monitoring fluid balance and for complications attributed to fluid accumulation is vital to avoiding adverse events and improving outcomes in sick children.
Careful examination of the concept of “fluid overload” in children reveals several gaps in our understanding of the impact of fluid accumulation. In early studies, the term fluid overload was used to simply describe positive fluid accumulation in children at the time of CKRT initiation [7,8,9, 12, 17, 20, 21]. In these studies, fluid accumulation likely represented a pathologic state was equated with fluid overload, and children by extension were perceived to benefit from intervention with CKRT. Since those initial studies, the terminology in the literature describing fluid accumulation has largely used the term fluid overload to equate with a state of positive fluid balance. However, use of the term fluid overload in this way introduces bias by the inherent assumption that all fluid accumulation is detrimental, and by extension fluid removal or “negative fluid overload” is good. Furthermore, the methodology utilized to calculate fluid balance in these studies varied [8, 12,13,14, 17, 20]. To advance our understanding and generate new knowledge, a standardization of terminology and methods for reporting fluid balance is necessary.
Fluid accumulation represents an attractive target for intervention in sick children. Several strategies to prevent or mitigate a state of fluid overload have been proposed [22]. Prior to widespread implementation of such strategies, however, it is vital to appraise the source and strength of existing evidence to support the association between positive fluid balance and outcomes in sick children. The current literature has significant limitations, including being small, single center, and observational (often retrospective) in design, along with a lack of standardized reporting on measures of fluid balance (definition, timing, epidemiology) across studies [23,24,25]. Rigorous observational studies and randomized trials evaluating prospective interventions and strategies to manage fluid balance and accumulation in sick children are needed.
Acknowledging the importance of fluid accumulation and disorders of fluid balance, the meeting chairs of the first ADQI dedicated to neonates and pediatric patients (the 26th ADQI) convened a diverse expert multidisciplinary working group dedicated to fluid balance, fluid accumulation, and fluid overload. We herein summarize the consensus to describe in detail: definitions and epidemiology of disorders of fluid balance, targets for intervention, and endpoints for clinical trials.
Methods
The methodology utilized for ADQI meetings has been developed iteratively over the last two decades [26]. The aim of ADQI meetings is to provide expert-based statements, supported by evidence where applicable, and interpretation of current knowledge for use in clinical care and research endeavors. In addition, ADQI aims to identify evidence and knowledge-to-care gaps to establish future research priorities. The 26th ADQI consensus meeting included physicians and scientists (adult and pediatric nephrology and critical care, pediatric cardiology), nurse practitioners, nurse educators, clinical pharmacists, critical care dieticians, health services researchers, and patient and family advocates for a 3-day meeting held in Napa Valley, CA on November 11–14, 2021.
The preparation began over a year prior to the in-person meeting with a detailed literature review (2001–2021), topic elicitation, question development, and proposed statements. The workgroup identified several themes around “fluid balance and fluid overload” to generate concrete questions for the in-person meeting which were iteratively refined though consensus during sequential plenary sessions.
Question 1: What defines fluid balance in sick children?
Statement: Fluid balance is the difference between total input and output that can be expressed as “daily” and “cumulative” over a defined duration of time (Table 1)
Many terms have been used interchangeably to describe the impact of fluid on outcomes in sick children, including but not limited to “fluid balance,” “fluid accumulation,” and “fluid overload.” Identifying consensus terminology is critical to advance our understanding of the impact of fluid management on outcomes across populations. Such definitions will guide the development of clinical practice guidelines, epidemiological surveillance, clinical trials, and ultimately improved care for sick children and neonates.
The concept of fluid balance is at the heart of describing fluid accumulation in any population. Fluid balance is a measure of the intake and output for a given duration. This is classically expressed over a period of time as daily fluid balance (24-h) or cumulative fluid balance (since admission, previous number of days, etc.). The cumulative fluid balance is often expressed as a percent corrected for a pre-specified anchor weight. We propose utilizing the terms daily fluid balance, cumulative fluid balance, and percent cumulative fluid balance to describe the fluid status objectively in sick children for the purpose of clinical care and research (Table 1).
The two methodologies commonly utilized in the literature to describe fluid balance include cumulative fluid input and output and weight-based methodology (Table 1). The “cumulative fluid input and output methodology” has been adopted since the aforementioned 2001 study that first described the impact of “fluid overload” on outcomes in critically ill children initiating CKRT [8, 11, 15, 20, 27, 30]. This is the most frequently used methodology of quantifying fluid accumulation in the literature.
With this methodology, practitioners must be able to accurately measure all inputs (intravenous fluids, blood products, enteral feeds, intravenous flushes, etc.) and all outputs (urine, drains, dressings, stool, etc.) over a given time period. In the absence of a Foley catheter, urine output is often calculated utilizing change in diaper weight or measured voids which introduces potential limitations. Inaccuracies or missing input/output measurements are carried forward in all subsequent fluid balance calculations, propagating any potential inaccuracy. Enteral and intravenous fluid (IVF) contributions to input are generally treated as equivalent. Furthermore, this methodology does not account for insensible losses or potential insensible gains (humidified ventilatory circuits).
The second methodology to describe fluid balance utilizes a weight-based approach [4, 14, 16,17,18] as a change in patient weight from an anchor weight. The ability to reliably measure weight in a consistent and safe matter is crucial for application of this approach, which frequently requires set protocols to account for potential barriers such as mechanical ventilation, extracorporeal membrane oxygenation (ECMO), and use of bed weights. In this context, a reliable weight does not necessarily reflect the patient’s “true weight,” but instead a weight that is measured using a consistent method from day to day. This approach removes the inherent inaccuracies of accounting for daily input and output and should theoretically capture insensible and other losses. This would also enable compensation for missed or inaccurate daily measures of fluid input and output in the calculation of cumulative fluid balance.
For either method, a weight is needed in the denominator to understand the percent change in fluid balance by serving as a baseline for the equation or the anchor weight. Anchor weights used to describe percent change in fluid balance in the literature include intensive care unit (ICU) admission weight, hospital admission weight, estimated dry weight, pre-operative weight, and birthweight in neonates [4, 7,8,9, 12, 14, 17, 28, 31]. The ICU admission weight is the weight most commonly utilized as the anchor weight for fluid balance calculations in the literature. This practice likely reflects the fact that the ICU admission weight often is the first recorded weight available to clinicians. Less commonly reported in the literature is the use of hospital admission weight, which may reflect the weight in the emergency department, ICU admission weight, or ward admission weight. This may risk introducing bias due to the lack of standardization of weighing practices across different areas. The admission weight represents patients in a variety of fluid balance states ranging from hypo- to hypervolemic. Finally, some reports have used the physician estimate of “dry weight” as the baseline for weight correction. However, clinicians do not reliably predict dry weight, recognize fluid overload clinically, and the dry weight implies a healthy state with muscle mass that may significantly change over the course of a critical illness [31]. Therefore, no clear gold standard exists against which to systematically compare different approaches, highlighting a knowledge gap requiring targeted study.
A vital point for defining fluid balance is the concept of euvolemia, commonly referred to clinically as an estimated healthy dry weight. In considering fluid balance, we must acknowledge some of the inherent assumptions commonly utilized in the literature and their weaknesses. In particular, most fluid balance calculations assume even fluid balance at ICU admission with the assumption that the anchor weight represents a euvolemic state. This assumption is not supported by evidence and is subject to many potential inaccuracies (over/inadequate fluid resuscitation prior to ICU admission, fluid management on cardiopulmonary bypass, intravenous fluids administered on the general wards, etc.).
In describing the terminology and methodology, the practicality and impact of these definitions in low- and middle-income countries (LMICs) necessitate a separate discussion. In evaluating the potential resources necessary to accurately monitor fluid balance, the “cumulative fluid input and output methodology” may be more resource intensive as it requires a detailed 24-h monitoring. The weight-based methodology has the advantage of being less resource intensive. In LMIC settings, it remains important to use a standardized method to define anchor weight. Weighing neonates and small children is a well-established method in neonatal and pediatric intensive care units (PICU) in LMICs. However, weighing older children and adolescents receiving mechanical ventilation is a challenge as ICU beds capable of weighing patients are less commonly available.
Critically ill neonates warrant separate discussion, as measuring fluid balance and fluid accumulation can be particularly challenging in this population. Special considerations for neonates include increased insensible losses, insensible losses that change with gestational age, and the expected physiologic diuresis over the first 1–2 postnatal weeks. The ascertainment of fluid balance in neonates is further complicated by the fact that sick neonates rarely have Foley catheters in place. Weight-based methods have been clearly shown to be a superior measure of fluid balance in neonates [14, 18, 32,33,34]. For the neonatal population, the most common anchor weight is the birthweight in the first two postnatal weeks [32]. After this period, there has been little research describing the accurate calculation of an anchor weight.
As the importance of fluid balance has become increasingly clear, an important step is to incorporate fluid balance as a “vital sign” in sick children. As with any other vital sign, fluid balance can be incorporated into clinical rounds and monitoring on a daily basis in sick children. Furthermore, there are high-risk populations where fluid balance may need to be evaluated on a more frequent basis (e.g., CKRT, ECMO). The availability of such data serves as a quality indicator, and quality improvement methodology can be utilized to optimize this [35, 36].
Areas for research
-
Identify the optimal anchor weight needed to calculate fluid balance measurements to be used through a patient’s clinical course (e.g., premorbid weight, dry weight, admission weight, PICU admission weight).
-
Systematically study, develop, implement, and evaluate protocols for daily weights in sick children and in different resourced settings (e.g., LMICs).
-
Evaluate the optimal method and process to adjust anchor weight for patients hospitalized for extended periods of time.
-
Evaluate the optimal method and process to adjust anchor weight for neonates outside of the first 2 postnatal weeks.
Question 2: What defines fluid overload in sick children?
Statement: Fluid overload denotes a pathologic state of positive fluid balance associated with a clinically observable event(s), which may vary by age, case-mix, acuity, and phase of illness. No specific threshold of positive fluid balance alone can define fluid overload across all sick children
In order to advance the field, the term percent cumulative fluid balance should be utilized to describe the cumulative positive fluid balance over a given time period. The term fluid overload should be defined as the degree of positive fluid balance that is associated with adverse patient-centered events such as increased length of mechanical ventilation, length of stay, and/or increased mortality. The threshold and timing of fluid balance that define the pathologic state of fluid overload differ across populations, and disease processes are variable (Table 2 and Supplemental Table S1) [2, 3, 8, 10, 11, 14,15,16, 18, 20, 23, 27,28,29, 37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89]. Observational studies have identified important characteristics that impact the thresholds associated with fluid overload, including age, underlying disease process, illness acuity, temporal profile in fluid balance, and available resources. In considering the utilization of thresholds to report fluid balance relative to reporting fluid balance as a continuous variable, one must understand the potential pitfalls to this practice. The utilization of thresholds is subject to potential bias (e.g., influence of extreme values), and this must be appreciated.
With the paradigm shift on how to ideally define fluid balance, there must be an associated shift in how we describe its epidemiology. In defining fluid overload as positive fluid balance associated with adverse clinical events, it is implicit that not all states of positive fluid balance are in fact deleterious. Furthermore, the full spectrum of fluid balance should be considered, including negative fluid balance. It is reasonable to think that negative fluid balance is not always beneficial for the patient, and that in certain disease states, may become detrimental to the patient. This paradigm and its associations with various disease states and clinical factors are illustrated in Fig. 1.
The Spectrum of Fluid Balance [90]. There is a spectrum of fluid balance for the sick child that is a U-shaped curve. (A) This spectrum can swing between positive fluid balance (+FB) and negative fluid balance (-FB) with varying levels of clinical impact depending on the severity of the abnormal fluid balance. (B) Depending on the case-mix and/or resources available, the U-shaped curve may have more or less tolerance for a positive or negative fluid balance. These depict examples of different scenarios where a patient population may have greater or lesser tolerance of a +FB or -FB scenario. (C) For any given sick child, there are several factors (i.e., host factors, interventions, adverse outcomes) that may push them towards a greater +FB or -FB or pull them back towards a state of neutral fluid balance at the center. These may vary over the course of the hospitalization and can be constantly changing
A major gap and limitation in the epidemiology of fluid balance is understanding the full continuum of fluid balance, particularly iatrogenic and clinically important negative fluid balance or clinical dehydration. The incidence of positive fluid balance, including temporal profile and magnitude, varies by center, definition, reported thresholds, and predetermined thresholds set as “clinically meaningful fluid overload” (Supplemental Table S1). The standardization of definitions and reporting will improve the understanding of the full spectrum of fluid balance and allow us to better define and describe fluid overload. The literature to date shows wide ranges in the incidence of fluid overload among critically ill children depending on the threshold used: 17–70% for > 5%, 4.5–56% for > 10%, 9–33% for > 20% (Supplemental Table S1).
In recent years, pediatric fluid research shifted from identifying absolute thresholds of fluid overload to understanding the impact of timing and trajectory of fluid accumulation on outcomes. In a single center, retrospective analysis of post-cardiac surgery patients, the median peak positive fluid balance was 4.5% and most commonly occurred on postoperative day 2 [91]. Among a cohort of mechanically ventilated children across five PICUs, the mean daily cumulative positive fluid balance was positive 20–35 ml/kg on hospital days 1–3 [92]. An evaluation of over 1000 critically ill children in Canada found that the positive fluid balance increased with each subsequent ICU day from 1.6% on hospital day 1 to 16.4% on hospital day 10 [2]. This data taken together emphasizes the importance of considering timing, magnitude, and trajectory in evaluating the impact of fluid balance.
A significant amount of work has gone into understanding the case mix and factors associated with derangements in fluid balance and the development of fluid overload (Supplemental Table S1). In post-cardiac surgery populations, young age, severity of AKI, longer cardiopulmonary bypass time, and surgical complexity were associated with derangements in fluid balance [11, 13, 37, 93]. In general, PICU populations, younger age, AKI, inotrope use, ventilatory support, shock diagnosis, and admission to a medical/surgical unit vs. a cardiac unit were associated with higher fluid balance categories [2, 3, 27, 94, 95]. Given the associations between positive fluid balance and adverse outcomes, including mortality, further investigation for modifiable factors as targets to enrich clinical trials and be actionable for intervention are needed.
Another important subset of patients with little data is in LMICs, like South Africa, that has reported lower proportions of critically ill children with fluid overload (3% with > 10%). The same cohort also had similar median peak positive fluid balance of 3.5% for their study period as compared to high-income settings [96]. Further work is needed to better understand whether different thresholds are associated with adverse events attributed to a positive fluid balance in often less resources settings encountered in LMICs.
Triggers and interventions
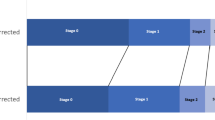
Two types of interventions can be implemented to modify fluid accumulation: limiting fluid administration and facilitating active fluid removal through pharmacologic (i.e., diuretics) and/or mechanical (i.e., kidney replacement therapy (KRT)) approaches. In sick children, administered fluids have varying degrees of necessity and include those that are life sustaining (i.e., resuscitation, blood products, medication, nutrition) and those that may be restricted (i.e., intravenous fluids, carriers for medications). A large proportion of fluid administered to critically ill children comes in forms that may be restricted, particularly maintenance fluids [97, 98]. We suggest clinicians and researchers study fluid balance in an overlapping but sequential manner: limitation, targeted diuretic therapy, and then consideration for the use of mechanical fluid removal (i.e., KRT). Figure 2 demonstrates a theoretical framework of the potential overlapping interventions based on the degree of fluid accumulation and its clinical impact.
Overlapping Interventions to Manage Fluid Balance. Interventions to manage fluid balance are overlapping based on the degree of fluid accumulation and clinical impact of the fluid. These interventions include limitation of continuous fluids, targeted diuretic therapy, and then consideration for the use of kidney support therapy. Green triangle represents limitations to continuous fluids and optimizing the osmotic gradient. Red triangle represents the need for renal replacement therapy. Purple oval outlines the role of diuretics while patients during both limitation of fluids and need for renal replacement therapy
Another important clinical consideration is to begin to consider preventative measures or fluid stewardship programs aimed at mitigating fluid accumulation in high risk populations prior to the development of overt fluid overload [3, 97, 99]. This could be done by identifying patients at high risk for developing fluid overload and/or kidney dysfunction (i.e., predictive enrichment and clinical risk prediction) that would further magnify the accumulation of IVF and by taking a targeted approach to more aggressively limit a positive fluid balance. This may include the deployment of several tools (e.g., renal angina index, novel biomarkers, furosemide stress test, fluid overload, and kidney injury score (FOKIS)) in high-risk populations [99,100,101,102,103]. These tools can be utilized to identify high-risk patients who would most benefit from a precision approach to fluid management with the targeted use of diuretics and kidney support therapy. A clinical trial currently underway that is utilizing this approach is the “Trial in AKI using NGAL and Fluid Overload to optimize CRRT Use” (TAKING FOCUS 2). This trial is evaluating the impact of sequentially mobilizing urinary biomarkers and the furosemide stress test to identify patients at risk for the development of fluid overload and severe AKI [104, 105].
Areas for future research
-
Further understanding of the incidence of both positive and negative fluid balance in all sick children (acute, critically ill, neonates, post-cardiac surgery, oncologic patients, etc.).
-
Further delineate the thresholds of positive fluid balance whereby the development of fluid overload is most likely to occur across all sick children.
-
Understand the impact of timing and trajectory of positive fluid balance on outcomes.
-
Identify the risk factors and potential modifiable factors for positive fluid balance and fluid overload in all sick children.
-
Further study and understanding of the interplay and impact of intravascular volume status and total body volume. Further work is needed to define and standardize the terminology including but not limited to hypervolemia, euvolemia, and hypovolemia relative to total body volume and intravascular volume.
-
Develop objective and reproducible measures of edema.
-
Identify the potential associations of negative fluid balance and clinical dehydration on clinical outcomes (such as venous thromboses).
-
Identify the incidence, risk factors, and associated outcomes of negative and positive fluid balance in children in LMICs. This is urgently needed to better understand and put into clinical context the findings of the FEAST trial.
-
Identify thresholds for intervention in various case mixes of sick children.
-
Evaluate the effectiveness of different interventions on the degree of positive fluid balance and the impact on fluid overload and associated outcomes.
-
Evaluate the effectiveness of a preventative strategy or fluid stewardship interventions. (i.e., reduce fluid administration, improve cumulative fluid balance, improve physiological outcomes, and improve patient reported outcomes).
Question 3: What are the challenges to translating observational data to clinical management of fluid balance?
Statement: The literature describing the association of fluid balance on outcomes is based on observational studies. Evidence from observational data alone cannot establish a causal relationship between fluid balance and outcomes
In order to move the field forward and improve outcomes related to fluid balance, a better understanding and establishment of the causal link between fluid balance and outcomes are needed. The lack of randomized trials in hospitalized children and complexity of evaluating fluid balance in critically ill patients make it challenging to causally link fluid balance to outcomes. In order to have the highest likelihood of success, these trials would benefit from utilizing many of the previously described tools to identify the highest risk patient populations that would potentially benefit. We provide further suggestions on optimizing study design and outcomes to inform the development of trials focused on the impact of fluid balance on outcomes.
Study designs
Table 3 outlines some of the ongoing and seminal studies that have been done to date in adults and children evaluating the impact of fluid balance on clinical outcomes. To date, most data surrounding the impact of fluid balance on outcomes are derived from observational studies. Observational data are important tools that allow us to study new concepts and exposures that are difficult to randomize. The confounders that have an association with both the exposure (i.e., fluid balance) and outcomes must be realized and controlled for. A useful tool for researchers is a directed acyclic graph (DAG). A DAG is a diagrammatic tool to allow researchers to visualize confounders, mediators, and intermediaries on pathways, as well as other relevant exposures. A DAG developed from observational studies describing the interactions of fluid overload and morbidity and mortality is presented in Fig. 3. A detailed understanding of the complex relationship between patient characteristics, fluid balance, and outcomes from observational studies will serve to inform clinical trial design.
Directed Acyclic Graph of Fluid Overload and Mortality [90]. The directed acyclic graph (DAG) above is an example of the utilization of this tool to describe the complex relationship between fluid overload and mortality in sick children. Different DAGs would be necessary to evaluate positive or negative fluid balance or other fluid states and different outcomes. The variables depicted in this figure are likely not exhaustive and additional nuanced variables could be considered (e.g., genetics). The figure is meant to be as comprehensive as possible with current knowledge-to-date to demonstrate the complexity of the relationship and the importance that all factors are considered to minimize bias when trying to use observational studies to evaluate the fluid overload-mortality relationship and its potential for a causal pathway. Arrows depict a known associated risk relationship (whether that is a positive or negative relationship) E=the primary exposure of interest (fluid overload in this example) O=the primary outcome of interest (mortality in this example) White circles = represent variables that are confounders in the primary exposure-outcome relationship and would be the required minimal adjustment variables to account for in order to get a fully adjusted analysis of the primary exposure-outcome relationship (i.e., the minimal adjustment set) Pink circles = represent additional variables that are also confounders but not necessary for a minimal adjustment set evaluating the primary exposure-outcome relationship Orange circle = ancestor to primary exposure of interest
In designing future observational studies, we suggest researchers to incorporate novel approaches to analysis to strengthen these studies. The first is to utilize appropriate sample sizes and to switch from logistic regression modelling (i.e., odds ratios) to linear or log-linear modelling (i.e., risk differences and risk ratios) when feasible. This analytic strategy will improve our understanding to look at risk ratios, risk differences, or attributable risks to determine the degree that a particular risk may be contributing to outcomes of interest.
Traditional randomized controlled trials, or explanatory trials, have been considered the gold standard to delineate causality in between an intervention and outcome. Randomized controlled trials can be challenging to design and implement, particularly for relationships as complex as outcomes associated with fluid balance. In recent years, pragmatic trials have been increasingly utilized to study approaches to management fluid, with fluid balance representing a key process and outcome measure. Pragmatic trials are characterized by simple eligibility, broad inclusion, simple data collection and use of core outcome sets, easily applied intervention, and group randomization (e.g., ward level, unit level, hospital level) [113]. Each of these characteristics aims to optimize the ease of the trial implementation, to evaluate interventions in “real world” scenarios, and to enable ease of translation of findings into practice. Several pragmatic and novel clinical trial design approaches have recently been applied (i.e., stepped-wedge implementation; cluster cross-over, quasi-experimental) in a range of clinical contexts that may be well-suited to evaluation of complex interventions for fluid management in sick children. Adaptive trial designs represent another important tool that may be utilized to study fluid balance in sick children. Simply put, such studies allow for modifications of study design that may be implemented at predefined interim analysis [114].
Given the complexity of fluid balance in sick children and our limited understanding of whether or not a direct causal pathway exists with adverse outcomes (i.e., mortality), we suggest innovative trial design be incorporated into future studies. A good example is the FLUID trial which proposes a hospital-wide (adults and children) cluster-randomized crossover trial to evaluate outcomes between 0.9% NaCl and Ringer’s lactate fluids (https://clinicaltrials.gov/ct2/show/NCT02721485, NCT02721485) [115].
Outcomes
The association of fluid overload and short-term outcomes during the initial hospitalization has been the most frequently reported study design, with mortality being the most common outcome. While mortality remains important, it is relatively uncommon, necessitating the need for alternative outcomes (Table 4). These outcomes include circumstances where fluid accumulation has led to an escalation in care due to the development of deteriorating clinical status or worsening organ function, including increased receipt of respiratory support, increased oxygenation index, increased ionotropic support, worsened cardiac dysfunction, subsequent or worsening AKI, receipt of KRT, or receipt of extracorporeal life support (ECLS). By extension, there are studies that report on outcomes related to the duration of such interventions (e.g., mechanical ventilation, ECLS, KRT) or hospitalization (ICU or total hospital length of stay) based on degrees of positive fluid balance. Additionally, ICU mortality and in-hospital mortality are commonly reported as outcomes of interest in observational studies. To date, there are few data describing the cost attributable to abnormalities in fluid balance in children.
Recent work suggests that AKI and disorders of fluid accumulation may impact the long-term outcomes associated with other organs and multisystem disorders. Recent work by the Life After Pediatric Sepsis Evaluation (LAPSE) investigators has evaluated the association of severe AKI with the development of new comorbidities in children following critical care admission for sepsis. In this multicenter study, children with severe AKI had an increased odds of death or new substantive functional morbidity (adjusted odds ratio, 2.78; 95% CI, 1.63–4.81; p < 0.001) [122]. More recently, the functional outcomes of children treated with CKRT were assessed by the Functional Status Scale (FSS) in a single-center retrospective study of 45 children. In this study, 31 (69%) had worse FSS score at PICU discharge, and 51% of the children had new morbidity (defined by the FSS score). Furthermore, on adjusted analysis, the degree of fluid overload at CKRT initiation predicted a worse FSS score in this population [118]. In critically ill adults, the degree of positive fluid balance at ICU discharge predicted impaired mobility and discharge to another healthcare facility [123]. To date, there has not been a systematic evaluation of the impact of fluid balance on long-term outcomes in sick children. Future studies should include a multidisciplinary evaluation of outcomes including respiratory, cardiovascular, neurodevelopmental, health-related quality of life, and functional outcomes.
In recent years, the Major Adverse Kidney Events (MAKE) have been proposed to be included as a composite outcome to be utilized and reported in all effectiveness clinical trials in AKI. This composite outcome includes death, new kidney support therapy, or persistent serum creatinine greater than two times baseline. This outcome can be assessed at 30, 60, and 90 days. An important area for future research is to understand the applicability of MAKE outcomes to children and to revise it as necessary to fit pediatric and neonatal outcomes.
Areas for future research
-
Pragmatic randomized trials such as crossovers, cluster RCTs, stepped-wedge, or other quasi-experimental designs are likely needed to further understand the complex relationship of fluid balance and mortality, and whether or not a causal link exists.
-
Capitalize on existing networks such as NINJA, collaborative pediatric critical care research network to conduct multicenter randomized trials to answer the missing epidemiological questions surrounding fluid balance and mortality and other clinical outcomes.
-
Effects on outcomes of “isolated positive fluid balance” in patients with “normal” kidney function.
-
Prospective association of fluid overload and organ function (causally or temporally associated).
-
Verification if fluid overload is “continuously” associated with outcomes (i.e., FO 10% leads to an ICU stay of 7 days; 15%, 8 days; 20%, 10 days) or if there is a “dichotomic” threshold (i.e., FO above/below the clinically relevant threshold has significant outcomes implications).
-
Improved understanding of the etiology of fluid balance and the development of the state of fluid overload. This includes contributions from severity of illness, disease pathophysiology, timing, impaired fluid clearance (e.g., AKI), inadequate fluid stewardship, endothelial dysfunction, inflammation, and capillary leak.
Conclusion
The understanding of how fluid balance impacts the clinical care and outcomes of sick children is evolving. This includes a recognition that observational data alone does not determine causality. Fluid balance is an objective measure that ideally will be considered daily in patient care. Fluid overload is a pathologic state associated with positive fluid balance and clinically observable events. Armed with these important distinctions, further research will be able to better discern the relationship between fluid balance and fluid overload as well as help evaluate future clinical practices.
References
Abulebda K, Cvijanovich NZ, Thomas NJ, Allen GL, Anas N, Bigham MT, Hall M, Freishtat RJ, Sen A, Meyer K, Checchia PA, Shanley TP, Nowak J, Quasney M, Weiss SL, Chopra A, Banschbach S, Beckman E, Lindsell CJ, Wong HR (2014) Post-ICU admission fluid balance and pediatric septic shock outcomes: a risk-stratified analysis. Crit Care Med 42:397–403
Alobaidi R, Basu RK, DeCaen A, Joffe AR, Lequier L, Pannu N, Bagshaw SM (2020) Fluid accumulation in critically ill children. Crit Care Med 48:1034–1041
Arikan AA, Zappitelli M, Goldstein SL, Naipaul A, Jefferson LS, Loftis LL (2012) Fluid overload is associated with impaired oxygenation and morbidity in critically ill children. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 13:253–258
Askenazi DJ, Koralkar R, Hundley HE, Montesanti A, Patil N, Ambalavanan N (2013) Fluid overload and mortality are associated with acute kidney injury in sick near-term/term neonate. Pediatr Nephrol 28:661–666
Bhaskar P, Dhar AV, Thompson M, Quigley R, Modem V (2015) Early fluid accumulation in children with shock and ICU mortality: a matched case-control study. Intensive Care Med 41:1445–1453
Flori HR, Church G, Liu KD, Gildengorin G, Matthay MA (2011) Positive fluid balance is associated with higher mortality and prolonged mechanical ventilation in pediatric patients with acute lung injury. Crit Care Res Pract 2011:854142
Foland JA, Fortenberry JD, Warshaw BL, Pettignano R, Merritt RK, Heard ML, Rogers K, Reid C, Tanner AJ, Easley KA (2004) Fluid overload before continuous hemofiltration and survival in critically ill children: a retrospective analysis. Crit Care Med 32:1771–1776
Goldstein SL, Currier H, Graf C, Cosio CC, Brewer ED, Sachdeva R (2001) Outcome in children receiving continuous venovenous hemofiltration. Pediatrics 107:1309–1312
Goldstein SL, Somers MJ, Baum MA, Symons JM, Brophy PD, Blowey D, Bunchman TE, Baker C, Mottes T, McAfee N, Barnett J, Morrison G, Rogers K, Fortenberry JD (2005) Pediatric patients with multi-organ dysfunction syndrome receiving continuous renal replacement therapy. Kidney Int 67:653–658
Gorga SM, Sahay RD, Askenazi DJ, Bridges BC, Cooper DS, Paden ML, Zappitelli M, Gist KM, Gien J, Basu RK, Jetton JG, Murphy HJ, King E, Fleming GM, Selewski DT (2020) Fluid overload and fluid removal in pediatric patients on extracorporeal membrane oxygenation requiring continuous renal replacement therapy: a multicenter retrospective cohort study. Pediatr Nephrol 35:871–882
Hassinger AB, Wald EL, Goodman DM (2014) Early postoperative fluid overload precedes acute kidney injury and is associated with higher morbidity in pediatric cardiac surgery patients. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 15:131–138
Hayes LW, Oster RA, Tofil NM, Tolwani AJ (2009) Outcomes of critically ill children requiring continuous renal replacement therapy. J Crit Care 24:394–400
Hazle MA, Gajarski RJ, Yu S, Donohue J, Blatt NB (2013) Fluid overload in infants following congenital heart surgery. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 14:44–49
Selewski DT, Akcan-Arikan A, Bonachea EM, Gist KM, Goldstein SL, Hanna M, Joseph C, Mahan JD, Nada A, Nathan AT, Reidy K, Staples A, Wintermark P, Boohaker LJ, Griffin R, Askenazi DJ, Guillet R, Neonatal Kidney C (2019) The impact of fluid balance on outcomes in critically ill nearterm/term neonates: a report from the AWAKEN study group. Pediatr Res 85:79–85
Selewski DT, Askenazi DJ, Bridges BC, Cooper DS, Fleming GM, Paden ML, Verway M, Sahay R, King E, Zappitelli M (2017) The impact of fluid overload on outcomes in children treated with extracorporeal membrane oxygenation: a multicenter retrospective cohort study. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 18:1126–1135
Selewski DT, Cornell TT, Blatt NB, Han YY, Mottes T, Kommareddi M, Gaies MG, Annich GM, Kershaw DB, Shanley TP, Heung M (2012) Fluid overload and fluid removal in pediatric patients on extracorporeal membrane oxygenation requiring continuous renal replacement therapy. Crit Care Med 40:2694–2699
Selewski DT, Cornell TT, Lombel RM, Blatt NB, Han YY, Mottes T, Kommareddi M, Kershaw DB, Shanley TP, Heung M (2011) Weight-based determination of fluid overload status and mortality in pediatric intensive care unit patients requiring continuous renal replacement therapy. Intensive Care Med 37:1166–1173
Selewski DT, Gist KM, Nathan AT, Goldstein SL, Boohaker LJ, Akcan-Arikan A, Bonachea EM, Hanna M, Joseph C, Mahan JD, Mammen C, Nada A, Reidy K, Staples A, Wintermark P, Griffin R, Askenazi DJ, Guillet R, Neonatal Kidney C (2020) The impact of fluid balance on outcomes in premature neonates: a report from the AWAKEN study group. Pediatr Res 87:550–557
Sinitsky L, Walls D, Nadel S, Inwald DP (2015) Fluid overload at 48 hours is associated with respiratory morbidity but not mortality in a general PICU: retrospective cohort study. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 16:205–209
Sutherland SM, Zappitelli M, Alexander SR, Chua AN, Brophy PD, Bunchman TE, Hackbarth R, Somers MJ, Baum M, Symons JM, Flores FX, Benfield M, Askenazi D, Chand D, Fortenberry JD, Mahan JD, McBryde K, Blowey D, Goldstein SL (2010) Fluid overload and mortality in children receiving continuous renal replacement therapy: the prospective pediatric continuous renal replacement therapy registry. American journal of kidney diseases : the official journal of the National Kidney Foundation 55:316–325
Lane PH, Mauer SM, Blazar BR, Ramsay NK, Kashtan CE (1994) Outcome of dialysis for acute renal failure in pediatric bone marrow transplant patients. Bone Marrow Transplant 13:613–617
Selewski DT, Goldstein SL (2018) The role of fluid overload in the prediction of outcome in acute kidney injury. Pediatr Nephrol 33:13–24
Alobaidi R, Morgan C, Basu RK, Stenson E, Featherstone R, Majumdar SR, Bagshaw SM (2018) Association between fluid balance and outcomes in critically ill children: a systematic review and meta-analysis. JAMA Pediatr 172:257–268
Bellos I, Iliopoulos DC, Perrea DN (2020) Association of postoperative fluid overload with adverse outcomes after congenital heart surgery: a systematic review and dose-response metaanalysis. Pediatr Nephrol 35:1109–1119
Weaver LJ, Travers CP, Ambalavanan N, Askenazi D (2023) Neonatal fluid overload-ignorance is no longer bliss. Pediatr Nephrol 38:47–60
Ronco C, Kellum JA, Mehta R (2001) Acute dialysis quality initiative (ADQI). Nephrology, dialysis, transplantation : official publication of the European Dialysis and Transplant Association - European Renal Association 16:1555–1558
Li Y, Wang J, Bai Z, Chen J, Wang X, Pan J, Li X, Feng X (2016) Early fluid overload is associated with acute kidney injury and PICU mortality in critically ill children. Eur J Pediatr 175:39–48
Neumayr TM, Alten JA, Bailly DK, Bhat PN, Brandewie KL, Diddle JW, Ghbeis M, Krawczeski CD, Mah KE, Raymond TT, Reichle G, Zang H, Selewski DT, Investigators N (2023) Assessment of fluid balance after neonatal cardiac surgery: a description of intake/output vs. weight-based methods. Pediatr Nephrol 38:1355–1364
Schmidt B, Roberts RS, Fanaroff A, Davis P, Kirpalani HM, Nwaesei C, Vincer M, Investigators T (2006) Indomethacin prophylaxis, patent ductus arteriosus, and the risk of bronchopulmonary dysplasia: further analyses from the trial of indomethacin prophylaxis in Preterms (TIPP). J Pediatr 148:730–734
Lima L, Menon S, Goldstein SL, Basu RK (2021) Timing of fluid overload and association with patient outcome. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 22:114–124
Lombel RM, Kommareddi M, Mottes T, Selewski DT, Han YY, Gipson DS, Collins KL, Heung M (2012) Implications of different fluid overload definitions in pediatric stem cell transplant patients requiring continuous renal replacement therapy. Intensive Care Med 38:663–669
Starr MC, Charlton JR, Guillet R, Reidy K, Tipple TE, Jetton JG, Kent AL, Abitbol CL, Ambalavanan N, Mhanna MJ, Askenazi DJ, Selewski DT, Harer MW, Neonatal Kidney Collaborative B (2021) Advances in Neonatal Acute Kidney injury. Pediatrics 148
van Asperen Y, Brand PL, Bekhof J (2012) Reliability of the fluid balance in neonates. Acta Paediatr 101:479–483
Bontant T, Matrot B, Abdoul H, Aizenfisz S, Naudin J, Jones P, Dauger S (2015) Assessing fluid balance in critically ill pediatric patients. Eur J Pediatr 174:133–137
Harer MW, Selewski DT, Kashani K, Basu RK, Gist KM, Jetton JG, Sutherland SM, Zappitelli M, Goldstein SL, Mottes TA, Askenazi DJ (2021) Improving the quality of neonatal acute kidney injury care: neonatal-specific response to the 22nd Acute disease quality initiative (ADQI) conference. J Perinatol 41:185–195
Selewski DT, Askenazi DJ, Kashani K, Basu RK, Gist KM, Harer MW, Jetton JG, Sutherland SM, Zappitelli M, Ronco C, Goldstein SL, Mottes TA (2021) Quality improvement goals for pediatric acute kidney injury: pediatric applications of the 22nd Acute disease quality initiative (ADQI) conference. Pediatr Nephrol 36:733–746
Lex DJ, Toth R, Czobor NR, Alexander SI, Breuer T, Sapi E, Szatmari A, Szekely E, Gal J, Szekely A (2016) Fluid overload is associated with higher mortality and morbidity in Pediatric patients undergoing cardiac surgery. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 17:307–314
Bailly DK, Alten JA, Gist KM, Mah KE, Kwiatkowski DM, Valentine KM, Diddle JW, Tadphale S, Clarke S, Selewski DT, Banerjee M, Reichle G, Lin P, Gaies M, Blinder JJ, Investigators N (2022) Fluid accumulation after Neonatal congenital cardiac operation: clinical implications and outcomes. Ann Thorac Surg
Rusmawatiningtyas D, Rahmawati A, Makrufardi F, Mardhiah N, Murni IK, Uiterwaal C, Savitri AI, Kumara IF, Nurnaningsih (2021) Factors associated with mortality of pediatric sepsis patients at the pediatric intensive care unit in a low-resource setting. BMC Pediatr 21:471
Marquez-Gonzalez H, Casanova-Bracamontes L, Munoz-Ramirez CM, Peregrino-Bejarano L, Bolanos-Tellez B, Yanez-Gutierrez L (2019) Relation between fluid overload and mortality in children with septic shock. Arch Argent Pediatr 117:105–113
Maitland K, Kiguli S, Opoka RO, Engoru C, Olupot-Olupot P, Akech SO, Nyeko R, Mtove G, Reyburn H, Lang T, Brent B, Evans JA, Tibenderana JK, Crawley J, Russell EC, Levin M, Babiker AG, Gibb DM, Group FT (2011) Mortality after fluid bolus in African children with severe infection. N Engl J Med 364:2483–2495.
Gillespie RS, Seidel K, Symons JM (2004) Effect of fluid overload and dose of replacement fluid on survival in hemofiltration. Pediatr Nephrol 19:1394–1399
Michael M, Kuehnle I, Goldstein SL (2004) Fluid overload and acute renal failure in pediatric stem cell transplant patients. Pediatr Nephrol 19:91–95
Flores FX, Brophy PD, Symons JM, Fortenberry JD, Chua AN, Alexander SR, Mahan JD, Bunchman TE, Blowey D, Somers MJ, Baum M, Hackbarth R, Chand D, McBryde K, Benfield M, Goldstein SL (2008) Continuous renal replacement therapy (CRRT) after stem cell transplantation. A report from the prospective pediatric CRRT Registry Group Pediatric nephrology 23:625–630
Baird JS, Wald EL (2010) Long-duration (> 4 weeks) continuous renal replacement therapy in critical illness. Int J Artif Organs 33:716–720
Elbahlawan L, West NK, Avent Y, Cheng C, Liu W, Barfield RC, Jones DP, Rajasekaran S, Morrison RR (2010) Impact of continuous renal replacement therapy on oxygenation in children with acute lung injury after allogeneic hematopoietic stem cell transplantation. Pediatr Blood Cancer 55:540–545
Askenazi DJ, Goldstein SL, Koralkar R, Fortenberry J, Baum M, Hackbarth R, Blowey D, Bunchman TE, Brophy PD, Symons J, Chua A, Flores F, Somers MJ (2013) Continuous renal replacement therapy for children </=10 kg: a report from the prospective pediatric continuous renal replacement therapy registry. J Pediatr 162:587–592 e583
Boschee ED, Cave DA, Garros D, Lequier L, Granoski DA, Guerra GG, Ryerson LM (2014) Indications and outcomes in children receiving renal replacement therapy in pediatric intensive care. J Crit Care 29:37–42
Jhang WK, Kim YA, Ha EJ, Lee YJ, Lee JH, Park YS, Park SJ (2014) Extrarenal sequential organ failure assessment score as an outcome predictor of critically ill children on continuous renal replacement therapy. Pediatr Nephrol 29:1089–1095
Modem V, Thompson M, Gollhofer D, Dhar AV, Quigley R (2014) Timing of continuous renal replacement therapy and mortality in critically ill children*. Crit Care Med 42:943–953
de Galasso L, Emma F, Picca S, Di Nardo M, Rossetti E, Guzzo I (2016) Continuous renal replacement therapy in children: fluid overload does not always predict mortality. Pediatr Nephrol 31:651–659
Lee ST, Cho H (2016) Fluid overload and outcomes in neonates receiving continuous renal replacement therapy. Pediatr Nephrol 31:2145–2152
Choi SJ, Ha EJ, Jhang WK, Park SJ (2017) Factors associated with mortality in continuous renal replacement therapy for Pediatric patients with Acute Kidney injury. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 18:e56–e61
Kaempfen S, Dutta-Kukreja P, Mok Q (2017) Continuous Venovenous hemofiltration in children less than or equal to 10 kg: a single-center experience. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 18:e70–e76
Barhight MF, Lusk J, Brinton J, Stidham T, Soranno DE, Faubel S, Goebel J, Mourani PM, Gist KM (2018) Hyperchloremia is independently associated with mortality in critically ill children who ultimately require continuous renal replacement therapy. Pediatr Nephrol 33:1079–1085
Miklaszewska M, Korohoda P, Zachwieja K, Sobczak A, Kobylarz K, Stefanidis CJ, Gozdzik J, Drozdz D (2019) Factors affecting mortality in children requiring continuous renal replacement therapy in pediatric intensive care unit. Adv Clin Exp Med 28:615–623
Cortina G, McRae R, Hoq M, Donath S, Chiletti R, Arvandi M, Gothe RM, Joannidis M, Butt W (2019) Mortality of critically ill children requiring continuous renal replacement therapy:effect of fluid overload, underlying disease, and timing of initiation. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 20:314–322
Chen Z, Wang H, Wu Z, Jin M, Chen Y, Li J, Wei Q, Tao S, Zeng Q (2021) Continuous renal-replacement therapy in critically ill children: practice changes and association with outcome. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies
Askenazi D, Patil NR, Ambalavanan N, Balena-Borneman J, Lozano DJ, Ramani M, Collins M, Griffin RL (2015) Acute kidney injury is associated with bronchopulmonary dysplasia/mortality in premature infants. Pediatr Nephrol 30:1511–1518
Matsushita FY, Krebs VLJ, Ferraro AA, de Carvalho WB (2020) Early fluid overload is associated with mortality and prolonged mechanical ventilation in extremely low birth weight infants. Eur J Pediatr 179:1665–1671
Rallis D, Balomenou F, Drougia A, Benekos T, Vlahos A, Tzoufi M (2021) Giapros V. Association of fluid overload with patent ductus arteriosus during the first postnatal day, Minerva Pediatr (Torino)
Sasser WC, Dabal RJ, Askenazi DJ, Borasino S, Moellinger AB, Kirklin JK, Alten JA (2014) Prophylactic peritoneal dialysis following cardiopulmonary bypass in children is associated with decreased inflammation and improved clinical outcomes. Congenit Heart Dis 9:106–115
Sampaio TZ, O'Hearn K, Reddy D, Menon K (2015) The influence of fluid overload on the length of mechanical ventilation in Pediatric congenital heart surgery. Pediatr Cardiol 36:1692–1699
Piggott KD, Soni M, Decampli WM, Ramirez JA, Holbein D, Fakioglu H, Blanco CJ, Pourmoghadam KK (2015) Acute Kidney injury and fluid overload in neonates following surgery for congenital heart disease. World J Pediatr Congenit Heart Surg 6:401–406
Park SK, Hur M, Kim E, Kim WH, Park JB, Kim Y, Yang JH, Jun TG, Kim CS (2016) Risk factors for Acute Kidney injury after congenital cardiac surgery in infants and children: a retrospective observational study. PLoS One 11:e0166328
Wilder NS, Yu S, Donohue JE, Goldberg CS, Blatt NB (2016) Fluid overload is associated with late poor outcomes in neonates following cardiac surgery. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 17:420–427
Kwiatkowski DM, Goldstein SL, Cooper DS, Nelson DP, Morales DL, Krawczeski CD (2017) Peritoneal Dialysis vs furosemide for prevention of fluid overload in infants after cardiac surgery: a randomized clinical trial. JAMA Pediatr 171:357–364
Delpachitra MR, Namachivayam SP, Millar J, Delzoppo C, Butt WW (2017) A case-control analysis of postoperative fluid balance and mortality after Pediatric cardiac surgery. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 18:614–622
SooHoo MM, Patel SS, Jaggers J, Faubel S, Gist KM (2018) Acute Kidney injury defined by fluid corrected creatinine in neonates after the Norwood procedure. World J Pediatr Congenit Heart Surg 9:513–521
Wang J, Wang C, Wang Y, Gao Y, Tian Y, Wang S, Li J, Yang L, Peng YG, Yan F (2020) Fluid overload in special Pediatric cohorts with anomalous origin of the left coronary artery from the pulmonary artery following surgical repair. J Cardiothorac Vasc Anesth 34:1565–1572
Gist KM, Henry BM, Borasino S, Rahman A, Webb T, Hock KM, Kim JS, Smood B, Mosher Z, Alten JA (2021) Prophylactic peritoneal Dialysis after the arterial switch operation: a retrospective cohort study. Ann Thorac Surg 111:655–661
Anderson NM, Bond GY, Joffe AR, MacDonald C, Robertson C, Urschel S, Morgan CJ, Western Canadian Complex Pediatric Therapies Program Follow-up G (2021) Post-operative fluid overload as a predictor of hospital and long-term outcomes in a pediatric heart transplant population. Pediatr Transplant 25:e13897
Zanaboni D, Min J, Seshadri R, Gaynor JW, Dreher M, Blinder JJ (2021) Higher total ultrafiltration volume during cardiopulmonary bypass-assisted infant cardiac surgery is associated with acute kidney injury and fluid overload. Pediatric nephrology.
Diaz F, Benfield M, Brown L, Hayes L (2017) Fluid overload and outcomes in critically ill children: a single center prospective cohort study. J Crit Care 39:209–213
Ingelse SA, Wiegers HM, Calis JC, van Woensel JB, Bem RA (2017) Early fluid overload prolongs mechanical ventilation in children with viral-lower respiratory tract disease. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 18:e106–e111
Samaddar S, Sankar J, Kabra SK, Lodha R (2018) Association of Fluid Overload with mortality in critically-ill mechanically ventilated children. Indian Pediatr 55:957–961
Sethi SK, Raghunathan V, Shah S, Dhaliwal M, Jha P, Kumar M, Paluri S, Bansal S, Mhanna MJ, Raina R (2018) Fluid overload and renal angina index at admission are associated with worse outcomes in critically ill children. Front Pediatr 6:118
Muttath A, Annayappa Venkatesh L, Jose J, Vasudevan A, Ghosh S (2019) Adverse outcomes due to aggressive fluid resuscitation in children: a prospective observational study. J Pediatr Intensive Care 8:64–70
Vaewpanich J, Akcan-Arikan A, Coss-Bu JA, Kennedy CE, Starke JR, Thammasitboon S (2019) Fluid overload and Kidney injury score as a predictor for ventilator-associated events. Front Pediatr 7:204
Barhight MF, Brinton JT, Soranno DE, Faubel S, Mourani PM, Gist KM (2020) Effects of hyperchloremia on renal recovery in critically ill children with acute kidney injury. Pediatr Nephrol
Barhight MF, Brinton J, Stidham T, Soranno DE, Faubel S, Griffin BR, Goebel J, Mourani PM, Gist KM (2018) Increase in chloride from baseline is independently associated with mortality in critically ill children. Intensive Care Med 44:2183–2191
Black CG, Thomas NJ, Yehya N (2021) Timing and clinical significance of fluid overload in Pediatric Acute respiratory distress syndrome. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 22:795–805
Rameshkumar R, Chidambaram M, Bhanudeep S, Krishnamurthy K, Sheriff A, Selvan T, Mahadevan S (2021) Prospective cohort study on cumulative fluid balance and outcome in critically ill children using a restrictive fluid protocol. Indian J Pediatr
Armenda S, Rusmawatiningtyas D, Makrufardi F, Arguni E (2021) Factors associated with clinical outcomes of pediatric dengue shock syndrome admitted to pediatric intensive care unit: a retrospective cohort study. Ann Med Surg (Lond) 66:102472
Hoover NG, Heard M, Reid C, Wagoner S, Rogers K, Foland J, Paden ML, Fortenberry JD (2008) Enhanced fluid management with continuous venovenous hemofiltration in pediatric respiratory failure patients receiving extracorporeal membrane oxygenation support. Intensive Care Med 34:2241–2247
Blijdorp K, Cransberg K, Wildschut ED, Gischler SJ, Jan Houmes R, Wolff ED, Tibboel D (2009) Haemofiltration in newborns treated with extracorporeal membrane oxygenation: a case comparison study. Crit Care 13:R48
Murphy HJ, Cahill JB, Twombley KE, Annibale DJ, Kiger JR (2018) Implementing a practice change: early initiation of continuous renal replacement therapy during neonatal extracorporeal life support standardizes care and improves short-term outcomes. J Artif Organs 21:76–85
Murphy HJ, Cahill JB, Twombley KE, Kiger JR (2018) Early continuous renal replacement therapy improves nutrition delivery in neonates during extracorporeal life support. J Ren Nutr 28:64–70
Mallory PP, Selewski DT, Askenazi DJ, Cooper DS, Fleming GM, Paden ML, Murphy L, Sahay R, King E, Zappitelli M, Bridges BC (2020) Acute Kidney injury, fluid overload, and outcomes in children supported with extracorporeal membrane oxygenation for a respiratory indication. ASAIO J 66:319–326
Goldstein SL, Akcan-Arikan A, Alobaidi R, Askenazi DJ, Bagshaw SM, Barhight M, Barreto E, Bayrakci B, Bignall ONR, Bjornstad E, Brophy PD, Chanchlani R, Charlton JR, Conroy AL, Deep A, Devarajan P, Dolan K, Fuhrman DY, Gist KM et al (2022) Consensus-based recommendations on priority activities to address Acute Kidney injury in children: a modified Delphi consensus statement. JAMA Netw Open 5:e2229442
Seguin J, Albright B, Vertullo L, Lai P, Dancea A, Bernier PL, Tchervenkov CI, Calaritis C, Drullinsky D, Gottesman R, Zappitelli M (2014) Extent, risk factors, and outcome of fluid overload after pediatric heart surgery*. Crit Care Med 42:2591–2599
Valentine SL, Sapru A, Higgerson RA, Spinella PC, Flori HR, Graham DA, Brett M, Convery M, Christie LM, Karamessinis L, Randolph AG, Pediatric Acute Lung I, Sepsis Investigator's N, Acute Respiratory Distress Syndrome Clinical Research N (2012) Fluid balance in critically ill children with acute lung injury. Crit Care Med 40:2883–2889
Mah KE, Hao S, Sutherland SM, Kwiatkowski DM, Axelrod DM, Almond CS, Krawczeski CD, Shin AY (2018) Fluid overload independent of acute kidney injury predicts poor outcomes in neonates following congenital heart surgery. Pediatr Nephrol 33:511–520
Chen J, Li X, Bai Z, Fang F, Hua J, Li Y, Pan J, Wang J, Feng X, Li Y (2016) Association of Fluid Accumulation with clinical outcomes in critically ill children with severe Sepsis. PLoS One 11:e0160093
Gist KM, Selewski DT, Brinton J, Menon S, Goldstein SL, Basu RK (2020) Assessment of the independent and synergistic effects of fluid overload and Acute Kidney injury on outcomes of critically ill children. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 21:170–177
Ketharanathan N, McCulloch M, Wilson C, Rossouw B, Salie S, Ahrens J, Morrow BM, Argent AC (2014) Fluid overload in a south African pediatric intensive care unit. J Trop Pediatr 60:428–433
Al-Lawati ZH, Sur M, Kennedy CE, Akcan Arikan A (2020) Profile of fluid exposure and recognition of fluid overload in critically ill children. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 21:760–766
Barhight MF, Nelson D, Chong G, Basu RK, Sanchez-Pinto LN (2022) Non-resuscitation fluid in excess of hydration requirements is associated with higher mortality in critically ill children. Pediatr Res 91:235–240
Akcan-Arikan A, Gebhard DJ, Arnold MA, Loftis LL, Kennedy CE (2017) Fluid overload and Kidney injury score: a multidimensional real-time assessment of renal disease burden in the critically ill patient. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 18:524–530
Basu RK, Chawla LS, Wheeler DS, Goldstein SL (2012) Renal angina: an emerging paradigm to identify children at risk for acute kidney injury. Pediatr Nephrol 27:1067–1078
Basu RK, Wang Y, Wong HR, Chawla LS, Wheeler DS, Goldstein SL (2014) Incorporation of biomarkers with the renal angina index for prediction of severe AKI in critically ill children. Clin J Am Soc Nephro 9:654–662
Basu RK, Zappitelli M, Brunner L, Wang Y, Wong HR, Chawla LS, Wheeler DS, Goldstein SL (2014) Derivation and validation of the renal angina index to improve the prediction of acute kidney injury in critically ill children. Kidney Int 85:659–667
Chawla LS, Davison DL, Brasha-Mitchell E, Koyner JL, Arthur JM, Shaw AD, Tumlin JA, Trevino SA, Kimmel PL, Seneff MG (2013) Development and standardization of a furosemide stress test to predict the severity of acute kidney injury. Crit Care 17:R207
Roy JP, Krallman KA, Basu RK, Chima RS, Fei L, Wilder S, Schmerge A, Gerhardt B, Fox K, Kirby C, Goldstein SL (2020) Early sequential risk stratification assessment to optimize fluid dosing, CRRT initiation and discontinuation in critically ill children with Acute Kidney injury: taking focus 2 process article. J Clin Trials 10
Goldstein SL, Krallman KA, Kirby C, Roy JP, Collins M, Fox K, Schmerge A, Wilder S, Gerhardt B, Chima R, Basu RK, Chawla L, Fei L (2022) Integration of the renal angina index and urine neutrophil gelatinase-associated Lipocalin improves severe Acute Kidney injury prediction in critically ill children and young adults. Kidney Int Rep 7:1842–1849
Ricci Z, Haiberger R, Pezzella C, Garisto C, Favia I, Cogo P (2015) Furosemide versus ethacrynic acid in pediatric patients undergoing cardiac surgery: a randomized controlled trial. Crit Care 19:2
Ingelse SA, Geukers VG, Dijsselhof ME, Lemson J, Bem RA, van Woensel JB (2019) Less is more?-a feasibility study of fluid strategy in critically ill children with Acute respiratory tract infection. Front Pediatr 7:496
Hjortrup PB, Haase N, Bundgaard H, Thomsen SL, Winding R, Pettila V, Aaen A, Lodahl D, Berthelsen RE, Christensen H, Madsen MB, Winkel P, Wetterslev J, Perner A, Group CT, Scandinavian Critical Care Trials G (2016) Restricting volumes of resuscitation fluid in adults with septic shock after initial management: the CLASSIC randomised, parallel-group, multicentre feasibility trial. Intensive Care Med 42:1695–1705
Vaara ST, Ostermann M, Bitker L, Schneider A, Poli E, Hoste E, Fierens J, Joannidis M, Zarbock A, van Haren F, Prowle J, Selander T, Backlund M, Pettila V, Bellomo R, team R-As (2021) Restrictive fluid management versus usual care in acute kidney injury (REVERSE-AKI): a pilot randomized controlled feasibility trial. Intensive Care Med 47:665–673
Myles PS, Bellomo R, Corcoran T, Forbes A, Peyton P, Story D, Christophi C, Leslie K, McGuinness S, Parke R, Serpell J, MTV C, Painter T, McCluskey S, Minto G, Wallace S, Australian, New Zealand College of Anaesthetists Clinical Trials N, the A, New Zealand Intensive Care Society Clinical Trials G (2018) Restrictive versus Liberal fluid therapy for major abdominal surgery. N Engl J Med 378:2263–2274
National Heart L, Blood Institute Acute Respiratory Distress Syndrome Clinical Trials N, Wiedemann HP, Wheeler AP, Bernard GR, Thompson BT, Hayden D, deBoisblanc B, Connors AF Jr, Hite RD, Harabin AL (2006) Comparison of two fluid-management strategies in acute lung injury. N Engl J Med 354:2564–2575
Parker MJ, Thabane L, Fox-Robichaud A, Liaw P, Choong K, Canadian Critical Care Trials G, the Canadian Critical Care Translational Biology G (2016) A trial to determine whether septic shock reversal is quicker in pediatric patients randomized to an early goal-directed fluid-sparing strategy versus usual care (SQUEEZE): study protocol for a pilot randomized controlled trial. Trials 17:556
Legrand M, Bagshaw SM, Koyner JL, Schulman IH, Mathis MR, Bernholz J, Coca S, Gallagher M, Gaudry S, Liu KD, Mehta RL, Pirracchio R, Ryan A, Steubl D, Stockbridge N, Erlandsson F, Turan A, Wilson FP, Zarbock A et al (2022) Optimizing the design and analysis of future AKI trials. J Am Soc Nephrol 33:1459–1470
Gilholm P, Ergetu E, Gelbart B, Raman S, Festa M, Schlapbach LJ, Long D, Gibbons KS, Australian, New Zealand Intensive Care Society Paediatric Study G (2023) Adaptive clinical trials in Pediatric critical care: a systematic review. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies.
McIntyre L, Taljaard M, McArdle T, Fox-Robichaud A, English SW, Martin C, Marshall J, Menon K, Muscedere J, Cook DJ, Weijer C, Saginur R, Maybee A, Iyengar A, Forster A, Graham ID, Hawken S, McCartney C, Seely AJ et al (2018) FLUID trial: a protocol for a hospital-wide open-label cluster crossover pragmatic comparative effectiveness randomised pilot trial. BMJ Open 8:e022780
Killien EY, Loftis LL, Clark JD, Muszynski JA, Rissmiller BJ, Singleton MN, White BR, Zimmerman JJ, Maddux AB, Pinto NP, Fink EL, Watson RS, Smith M, Ringwood M, Graham RJ, Post P, Injury PCIotPAL, Sepsis I, the Eunice Kennedy Shriver National Institute of Child H, Human Development Collaborative Pediatric Critical Care Research N (2021) Health-related quality of life outcome measures for children surviving critical care: a scoping review. Qual Life Res 30:3383–3394
Killien EY, Rivara FP, Dervan LA, Smith MB, Watson RS (2022) Components of health-related quality of life most affected following pediatric critical illness. Crit Care Med 50:e20–e30
Smith M, Bell C, Vega MW, Tufan Pekkucuksen N, Loftis L, McPherson M, Graf J, Akcan Arikan A (2022) Patient-centered outcomes in pediatric continuous kidney replacement therapy: new morbidity and worsened functional status in survivors. Pediatr Nephrol 37:189–197
Pollack MM, Holubkov R, Funai T, Clark A, Moler F, Shanley T, Meert K, Newth CJ, Carcillo J, Berger JT, Doctor A, Berg RA, Dalton H, Wessel DL, Harrison RE, Dean JM, Jenkins TL (2014) Relationship between the functional status scale and the pediatric overall performance category and pediatric cerebral performance category scales. JAMA Pediatr 168:671–676
Pollack MM, Holubkov R, Glass P, Dean JM, Meert KL, Zimmerman J, Anand KJ, Carcillo J, Newth CJ, Harrison R, Willson DF, Nicholson C, Eunice Kennedy Shriver National Institute of Child H, Human Development Collaborative Pediatric Critical Care Research N (2009) Functional status scale: new pediatric outcome measure. Pediatrics 124:e18–e28
Heneghan JA, Sobotka SA, Hallman M, Pinto N, Killien EY, Palumbo K, Murphy Salem S, Mann K, Smith B, Steuart R, Akande M, Graham RJ (2021) Outcome measures following critical illness in children with disabilities: a scoping review. Front Pediatr 9:689485
Starr MC, Banks R, Reeder RW, Fitzgerald JC, Pollack MM, Meert KL, PS MQ, Mourani PM, Chima RS, Sorenson S, Varni JW, Hingorani S, Zimmerman JJ, Life After Pediatric Sepsis Evaluation I (2020) Severe Acute Kidney injury is associated with increased risk of death and new morbidity after Pediatric septic shock. Pediatric critical care medicine : a journal of the Society of Critical Care Medicine and the World Federation of Pediatric Intensive and Critical Care Societies 21:e686–e695
Mitchell KH, Carlbom D, Caldwell E, Leary PJ, Himmelfarb J, Hough CL (2015) Volume overload: prevalence, risk factors, and functional outcome in survivors of septic shock. Ann Am Thorac Soc 12:1837–1844
Acknowledgements
The ADQI 26 workgroup:
The following individuals contributed to the formulation and content of this work in accordance with their participation in the 26th Acute Disease Quality Initiative (ADQI XXVI) and should be citable as collaborators on Pubmed
Rashid Alobaidi1, David J. Askenazi2, Erin Barreto3, Benan Bayrakci4, O.N. Ray Bignall II5, Patrick Brophy6, Jennifer Charlton7, Rahul Chanchlani8, Andrea L. Conroy9, Akash Deep10, Prasad Devarajan11, Kristin Dolan12, Dana Fuhrman13, Katja M. Gist11, Stephen M. Gorga14, Jason H. Greenberg15, Denise Hasson11, Emma Heydari1, Arpana Iyengar16, Jennifer Jetton17, Catherine Krawczeski5, Leslie Meigs18, Shina Menon19, Catherine Morgan1, Jolyn Morgan11, Theresa Mottes20, Tara Neumayr21, Danielle Soranno9, Natalja Stanski11, Michelle Starr9, Scott M. Sutherland22, Jordan Symons19, Molly Vega23, Michael Zappitelli24, Claudio Ronco25, Ravindra L. Mehta26, John Kellum27, Marlies Ostermann28
1Alberta Health Sciences University, Edmonton, Alberta, Canada
2University of Alabama at Birmingham, Children’s Hospital of Alabama, Birmingham, AL, USA
3Hacettepe University, Ankara, Turkey
4Mayo Clinic, Rochester, MN, USA
5Nationwide Children’s Hospital, Ohio State University, Columbus, OH, USA
6Golisano Children’s Hospital, Rochester University Medical Center, Rochester, NY, USA
7University of Virginia, Charlottesville, VA, USA
8McMaster University, McMaster Children’s Hospital, Hamilton, ON, Canada
9Riley Children’s Hospital, Indiana University School of Medicine, Indianapolis, IN, USA
10King’s College London, King’s College Hospital NHS Foundation Trust, London, UK
11Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA
12Mercy Children’s Hospital, University of Missouri-Kansas City School of Medicine, Kansas City, MO, USA
13Children’s Hospital of Pittsburgh, University of Pittsburgh Medical Center, Pittsburgh, PA, USA
14C.S. Mott Children’s Hospital, University of Michigan, Ann Arbor, MI, USA
15Yale University Medical Center, New Haven, CT, USA
16St. John’s National Academy of Health Sciences, Bangalore, India
17Stead Family Children’s Hospital, University of Iowa, Iowa City, IA, USA
18University of St. Thomas, Houston, TX USA
19Seattle Children’s Hospital, University of Washington, Seattle, WA, USA
20Ann & Robert H. Lurie Children’s Hospital, Chicago, IL, USA
21Washington University School of Medicine, St. Louis, MO, USA
22Lucille Packard Children’s Hospital, Stanford University, Palo Alto, CA, USA
23Texas Children’s Hospital, Baylor College of Medicine, Houston, TX, USA
24Hospital for Sick Children, Toronto, Ontario, Canada
25San Bartolo Hospital, Universiti di Padova, Vicenza, Italy
26University of California San Diego School of Medicine, La Jolla, CA, USA
27University of Pittsburgh Medical Center, Pittsburgh, PA, USA
28Guy’s and St. Thomas’ NHS Foundation Trust, London, UK
Funding
Open access funding provided by Università degli Studi di Firenze within the CRUI-CARE Agreement. The in-person meeting was supported by educational grants from AM-Pharma, Baxter, bioMerieux, BioPorto Diagnostics, Cincinnati Children’s Hospital, ExThera Medical, Leadiant, Lowell Therapeutics, MediBeacon Inc., Medtronic, Nuwellis Inc., Potrero, RES Seminars, SeaStar Medical, and Stavro Medical. Role of the funder/sponsor: the funders had no involvement in the selection of the panelists, design or conduct of the study; collection, management, analysis, or interpretation of the data; preparation, review, and approval of the manuscript; and decision to submit the manuscript for publication. Representatives from the funders were present at the face-to-face meeting but did not participate in the panel discussions, voting, or final decisions.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
Dr. Goldstein reported receiving personal fees from BioPorto Diagnostics, grant support and personal fees from Medtronic, Nuwellis Inc., Leadiant, personal fees from MediBeacon Inc., stock options and personal fees from Baxter Healthcare, grant support and personal fees from SeaStar Medical, and grants from ExThera Medical outside the submitted work. Dr. Akcan-Arikan reported receiving financial support for research from BioPorto research funds paid to her institution outside the submitted work. Dr. Askenazi reported receiving personal fees from Baxter, Nuwellis Inc., Medtronic, SeaStar, and BioPorto, and financial support for research from Zorro-Flow outside the submitted work; in addition, Dr. Askenazi reported having a patent for Zorro-Flow, an external urine collection device pending, and a patent for continuous renal replacement therapy (CRRT) advancements pending. Dr. Bagshaw reported receiving personal fees from Baxter and BioPorto during the conduct of the study. Dr. Barreto reported receiving financial support for research from FAST Biomedical Consultant and Wolters-Kluwer Consultant outside the submitted work. Dr. Brophy reported receiving personal fees from UpToDate, during the conduct of the study, and American Board of Medical Specialities finance board. Dr. Charlton reported receiving personal fees from Medtronics Carpediem Clinical Events Committee outside the submitted work. Dr. Devarajan reported receiving grants from National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases during the conduct of the study; holding a patent as co-inventor on patents licensed to Abbott Diagnostics and BioPorto Inc., on use of neutrophil gelatinase-associated lipocalin as a biomarker of kidney injury. Dr. Gist reported receiving financial support for research from Medtronic Speaker Honorarium and financial support for research from BioPorto Diagnostics consulting fees outside the submitted work. Dr. Menon reported receiving personal fees from Nuwellis Inc. outside the submitted work. Dr. J. Morgan reported receiving personal fees from Medtronic Consultant outside the submitted work. Dr. Mottes reported receiving financial support for research from Medtronic outside the submitted work. Dr. Stanski reported receiving grants from National Center for Advancing Translational Sciences of the National Institutes of Health Institutional CT2 grant, 2UL1TR001425-05A1 and travel reimbursement from pADQI Travel funds to travel to consensus meeting (flight and hotel) during the conduct of the study; in addition, Dr. Stanski reported holding a patent for PERSEVERE-II AKI Prediction Model pending. Dr. Zappitelli reported receiving financial support for research from BioPorto Inc. adjudicator/consultant for a study on neutrophil gelatinase-associated lipocalin and financial support for research from Baxter Inc. Honorarium for a national talk on CRRT (not viewed by company ahead of time) outside the submitted work. Dr. Kellum reported receiving personal fees from Dialco; being an employee of Spectral Medical and its wholly owned subsidiary Dialco outside the submitted work; and paid consultant for Astute Medical. Dr. Ostermann reported receiving grants from Fresenius Research funding, grants from Baxter Research funding, and grants from bioMerieux Research funding during the conduct of the study. Dr. Basu reported receiving personal fees from BioPorto Diagnostics outside of the submitted work and personal fees from bioMerieux outside of the submitted work. During the conduct of the study, in addition, Dr. Basu reported having a patent for Renal Angina Index pending outside of the submitted work, and a patent for olfactomedin-4 pending outside of the submitted work. No other disclosures were reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
All authors are members of the Pediatric ADQI Collaborative
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Selewski, D.T., Barhight, M.F., Bjornstad, E.C. et al. Fluid assessment, fluid balance, and fluid overload in sick children: a report from the Pediatric Acute Disease Quality Initiative (ADQI) conference. Pediatr Nephrol 39, 955–979 (2024). https://doi.org/10.1007/s00467-023-06156-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-023-06156-w