Abstract
Background
The aim of this study was to analyze the long-term efficacy and safety of angiotensin-converting enzyme inhibitor (ACEi) and ACEi + angiotensin receptor blocker (ARB) treatments in a cohort of children with Alport syndrome (AS).
Methods
This was a respective review of 79 Chinese children with AS who received ACEi alone or combined ACEi + ARB therapy.
Results
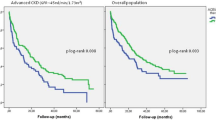
The mean age of the pediatric patients with AS at onset of treatment was 8.6 ± 4.1 (range 1.5–16.3) years. The mean duration of follow-up was 2.5 ± 1.8 (range 0.5–7.8) years. For analysis, we separated the children into three groups according to proteinuria level before treatment, namely, <25, 25–50, and ≥50 mg/kg/day, respectively; after 1 year of treatment the proteinuria had decreased from 11.0 to 9.7 mg/kg/day, from 34.6 to 15.2 mg/kg/day, and from 73.0 to 50.0 mg/kg/day in each group, respectively. Proteinuria decreased significantly during the first 2 years of treatment and was stable from the third to fifth years of treatment. There was no statistically significant difference in the antiproteinuric effect of the ACEi and ACEi + ARB treatments in patients with severe or less severe mutations after 1 year of therapy. Five children stopped the ACEi + ARB treatment due to a decline in creatinine clearance.
Conclusion
Our findings demonstrate that early and long-term ACEi and ARB treatments in children with AS is efficient and well tolerated. The antiproteinuric effect of ACEi and ARB is of equal value in children with severe and less severe mutations in the COL4An gene.

Similar content being viewed by others
References
Barker DF, Hostikka SL, Zhou J, Chow LT, Oliphant AR, Gerken SC, Gregory MC, Skolnick MH, Atkin CL, Tryggvason K (1990) Identification of mutations in the COL4A5 collagen gene in Alport syndrome. Science 248:1224–1227
Nagel M, Nagorka S, Gross O (2005) Novel COL4A5, COL4A4, and COL4A3 mutations in Alport syndrome. Hum Mutat 26:60
Mochizuki T, Lemmink HH, Mariyama M, Antignac C, Gubler MC, Pirson Y, Verellen-Dumoulin C, Chan B, Schröder CH, Smeets HJ (1994) Identification of mutations in the alpha 3(IV) and alpha 4(IV) collagen genes in autosomal recessive Alport syndrome. Nat Genet 8:77–81
Jais JP, Knebelmann B, Giatras I, De Marchi M, Rizzoni G, Renieri A, Weber M, Gross O, Netzer KO, Flinter F, Pirson Y, Verellen C, Wieslander J, Persson U, Tryggvason K, Martin P, Hertz JM, Schröder C, Sanak M, Krejcova S, Carvalho MF, Saus J, Antignac C, Smeets H, Gubler MC (2000) X-linked Alport syndrome: natural history in 195 families and genotype- phenotype correlations in males. J Am Soc Nephrol 11:649–657
Wang F, Zhao D, Ding J, Zhang H, Zhang Y, Yu L, Xiao H, Yao Y, Zhong X, Wang S (2012) Skin biopsy is a practical approach for the clinical diagnosis and molecular genetic analysis of x-linked Alport’s syndrome. J Mol Diagn 14:586–593
Zhang Y, Wang F, Ding J, Zhang H, Zhao D, Yu L, Xiao H, Yao Y, Zhong X, Wang S (2012) Genotype-phenotype correlations in 17 Chinese patients with autosomal recessive Alport syndrome. Am J Med Genet A 158A:2188–2193
Kashtan CE, Ding J, Gregory M, Gross O, Heidet L, Knebelmann B, Rheault M, Licht C, Alport Syndrome Research Collaborative (2013) Clinical practice recommendations for the treatment of Alport syndrome: a statement of the Alport Syndrome Research Collaborative. Pediatr Nephrol 28:5–11
Gross O, Licht C, Anders HJ, Hoppe B, Beck B, Tönshoff B, Höcker B, Wygoda S, Ehrich JH, Pape L, Konrad M, Rascher W, Dötsch J, Müller-Wiefel DE, Hoyer P, Study Group Members of the Gesellschaft für Pädiatrische Nephrologie, Knebelmann B, Pirson Y, Grunfeld JP, Niaudet P, Cochat P, Heidet L, Lebbah S, Torra R, Friede T, Lange K, Müller GA, Weber M (2012) Early angiotensin-converting enzyme inhibition in Alport syndrome delays renal failure and improves life expectancy. Kidney Int 81:494–501
Webb NJ, Shahinfar S, Wells TG, Massaad R, Gleim GW, McCrary Sisk C, Lam C (2013) Losartan and enalapril are comparable in reducing proteinuria in children with Alport syndrome. Pediatr Nephrol 28:737–743
Li JG, Ding J, Wang F, Zhang HW (2009) Drugs controlling proteinuria of patients with Alport syndrome. World J Pediatr 5:308–311
Proesmans W, Knockaert H, Trouet D (2000) Enalapril in paediatric patients with Alport syndrome: 2 years’ experience. Eur J Pediatr 159:430–433
Webb NJ, Lam C, Shahinfar S, Strehlau J, Wells TG, Gleim GW, Le Bailly De Tilleghem C (2011) Efficacy and safety of losartan in children with Alport syndrome—results from a subgroup analysis of a prospective, randomized, placebo- or amlodipine-controlled trial. Nephrol Dial Transplant 26:2521–2526
Adler L, Mathew R, Futterweit S, Frank R, Gauthier BG, Kashtan CE, Trachtman H (2002) Angiotensin converting enzyme inhibitor therapy in children with Alport syndrome: effect on urinary albumin, TGF-beta, and nitrite excretion. BMC Nephrol 3:2
Proesmans W, Van Dyck M (2004) Enalapril in children with Alport syndrome. Pediatr Nephrol 19:271–275
Conflict of interest
The authors declared that they have no conflict of interest.
Ethical disclosure
This study was approved by the Ethical Committee of Peking University First Hospital and parental/patient consent was not required due to the retrospective nature of the study.
Financial statement
This study was supported by grants from the National Nature Science Foundation (30801252 81070545, and 81400685), the Beijing Nature Science Foundation (7102148), and the National “Twelfth Five-Year” Science and Technology Support Project (2012BAI03B02).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, Y., Wang, F., Ding, J. et al. Long-term treatment by ACE inhibitors and angiotensin receptor blockers in children with Alport syndrome. Pediatr Nephrol 31, 67–72 (2016). https://doi.org/10.1007/s00467-015-3184-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-015-3184-5