Abstract
Background
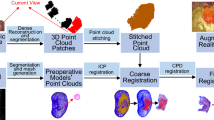
Augmented reality (AR) is the fusion of computer-generated and real-time images. AR can be used in surgery as a navigation tool, by creating a patient-specific virtual model through 3D software manipulation of DICOM imaging (e.g., CT scan). The virtual model can be superimposed to real-time images enabling transparency visualization of internal anatomy and accurate localization of tumors. However, the 3D model is rigid and does not take into account inner structures’ deformations. We present a concept of automated AR registration, while the organs undergo deformation during surgical manipulation, based on finite element modeling (FEM) coupled with optical imaging of fluorescent surface fiducials.
Methods
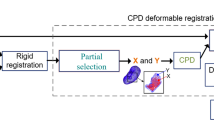
Two 10 × 1 mm wires (pseudo-tumors) and six 10 × 0.9 mm fluorescent fiducials were placed in ex vivo porcine kidneys (n = 10). Biomechanical FEM-based models were generated from CT scan. Kidneys were deformed and the shape changes were identified by tracking the fiducials, using a near-infrared optical system. The changes were registered automatically with the virtual model, which was deformed accordingly. Accuracy of prediction of pseudo-tumors’ location was evaluated with a CT scan in the deformed status (ground truth). In vivo: fluorescent fiducials were inserted under ultrasound guidance in the kidney of one pig, followed by a CT scan. The FEM-based virtual model was superimposed on laparoscopic images by automatic registration of the fiducials.
Results
Biomechanical models were successfully generated and accurately superimposed on optical images. The mean measured distance between the estimated tumor by biomechanical propagation and the scanned tumor (ground truth) was 0.84 ± 0.42 mm. All fiducials were successfully placed in in vivo kidney and well visualized in near-infrared mode enabling accurate automatic registration of the virtual model on the laparoscopic images.
Conclusions
Our preliminary experiments showed the potential of a biomechanical model with fluorescent fiducials to propagate the deformation of solid organs’ surface to their inner structures including tumors with good accuracy and automatized robust tracking.






Similar content being viewed by others
References
Kim SP, Thompson RH (2013) Kidney function after partial nephrectomy: current thinking. Curr Opin Urol 23:105–111
Tang YH, Wen TF, Chen X (2013) Anatomic versus non-anatomic liver resection for hepatocellular carcinoma: a systematic review. Hepato-gastroenterology 60:2019–2025
Hou W, Yan W, Ji Z (2015) Anatomic features involved in technical complexity of partial nephrectomy. Urology 85:1–7
Santambrogio R, Opocher E, Ceretti AP, Barabino M, Costa M, Leone S, Montorsi M (2007) Impact of intraoperative ultrasonography in laparoscopic liver surgery. Surg Endosc 21:181–188
Marescaux J, Diana M (2015) Next step in minimally invasive surgery: hybrid image-guided surgery. J Pediatr Surg 50:30–36
D’Agostino J, Diana M, Vix M, Soler L, Marescaux J (2012) Three-dimensional virtual neck exploration before parathyroidectomy. N Engl J Med 367:1072–1073
Marescaux J, Diana M (2015) Inventing the future of surgery. World J Surg 39:615–622
Marzano E, Piardi T, Soler L, Diana M, Mutter D, Marescaux J, Pessaux P (2013) Augmented reality-guided artery-first pancreatico-duodenectomy. J Gastrointest Surg 17:1980–1983
Pessaux P, Diana M, Soler L, Piardi T, Mutter D, Marescaux J (2014) Robotic duodenopancreatectomy assisted with augmented reality and real-time fluorescence guidance. Surg Endosc 28:2493–2498
Pessaux P, Diana M, Soler L, Piardi T, Mutter D, Marescaux J (2015) Towards cybernetic surgery: robotic and augmented reality-assisted liver segmentectomy. Langenbecks Arch Surg 400:381–385
Haouchine N, Cotin S, Peterlik I, Dequidt J, Lopez MS, Kerrien E, Berger MO (2015) Impact of soft tissue heterogeneity on augmented reality for liver surgery. IEEE Trans Vis Comput Graph 21:584–597
Collins BT, Erickson K, Reichner CA, Collins SP, Gagnon GJ, Dieterich S, McRae DA, Zhang Y, Yousefi S, Levy E, Chang T, Jamis-Dow C, Banovac F, Anderson ED (2007) Radical stereotactic radiosurgery with real-time tumor motion tracking in the treatment of small peripheral lung tumors. Radiat Oncol 2:39
Dieterich S, Gibbs IC (2011) The CyberKnife in clinical use: current roles, future expectations. Front Radiat Ther Oncol 43:181–194
Chavalitdhamrong D, DiMaio CJ, Siersema PD, Wagh MS (2015) Technical advances in endoscopic ultrasound-guided fiducial placement for the treatment of pancreatic cancer. Endosc Int Open 3:E373–E377
Kong SH, Noh YW, Suh YS, Park HS, Lee HJ, Kang KW, Kim HC, Lim YT, Yang HK (2015) Evaluation of the novel near-infrared fluorescence tracers pullulan polymer nanogel and indocyanine green/gamma-glutamic acid complex for sentinel lymph node navigation surgery in large animal models. Gastric Cancer 18:55–64
Crum WR, Camara O, Hill DL (2006) Generalized overlap measures for evaluation and validation in medical image analysis. IEEE Trans Med Imaging 25:1451–1461
Faure F, Duriez C, Delingette H, Allard J, Gilles B, Marchesseau S, Talbot H, Courtecuisse H, Bousquet G, Peterlik I (2012) Sofa: a multi-model framework for interactive physical simulation. Soft tissue biomechanical modeling for computer assisted surgery. Springer, New York, pp 283–321
Yamada H, Evans FG (1970) Strength of biological materials. Williams & Wilkins, Philadelphia
Marescaux J, Diana M, Soler L (2013) Augmented reality and minimally invasive surgery. J Gastroenterol Hepatol Res 2:555–560
Pilet J, Lepetit V, Fua P (2008) Fast non-rigid surface detection, registration and realistic augmentation. Int J Comput Vis 76:109–122
Gay-Bellile V, Bartoli A, Sayd P (2010) Direct estimation of nonrigid registrations with image-based self-occlusion reasoning. IEEE Trans Pattern Anal 32:87–104
Haouchine N, Dequidt J, Berger MO, Cotin S (2015) Monocular 3D reconstruction and augmentation of elastic surfaces with self-occlusion handling. IEEE Trans Vis Comput Graph 21:1363–1376
Marescaux J, Rubino F, Arenas M, Mutter D, Soler L (2004) Augmented-reality-assisted laparoscopic adrenalectomy. JAMA J Am Med Assoc 292:2214–2215
Teber D, Guven S, Simpfendorfer T, Baumhauer M, Guven EO, Yencilek F, Gozen AS, Rassweiler J (2009) Augmented reality: a new tool to improve surgical accuracy during laparoscopic partial nephrectomy? Preliminary in vitro and in vivo results. Eur Urol 56:332–338
Chen Y, Li H, Wu D, Bi K, Liu C (2014) Surgical planning and manual image fusion based on 3D model facilitate laparoscopic partial nephrectomy for intrarenal tumors. World J Urol 32:1493–1499
Wang D, Zhang B, Yuan X, Zhang X, Liu C (2015) Preoperative planning and real-time assisted navigation by three-dimensional individual digital model in partial nephrectomy with three-dimensional laparoscopic system. Int J Comput Assist Radiol Surg 10:1461–1468
Marvik R, Lango T, Tangen GA, Andersen JO, Kaspersen JH, Ystgaard B, Sjolie E, Fougner R, Fjosne HE, Nagelhus Hernes TA (2004) Laparoscopic navigation pointer for three-dimensional image-guided surgery. Surg Endosc 18:1242–1248
Ieiri S, Uemura M, Konishi K, Souzaki R, Nagao Y, Tsutsumi N, Akahoshi T, Ohuchida K, Ohdaira T, Tomikawa M, Tanoue K, Hashizume M, Taguchi T (2012) Augmented reality navigation system for laparoscopic splenectomy in children based on preoperative CT image using optical tracking device. Pediatr Surg Int 28:341–346
Nam WH, Kang DG, Lee D, Lee JY, Ra JB (2012) Automatic registration between 3D intra-operative ultrasound and pre-operative CT images of the liver based on robust edge matching. Phys Med Biol 57:69–91
Shekhar R, Dandekar O, Bhat V, Philip M, Lei P, Godinez C, Sutton E, George I, Kavic S, Mezrich R, Park A (2010) Live augmented reality: a new visualization method for laparoscopic surgery using continuous volumetric computed tomography. Surg Endosc 24:1976–1985
Noh YW, Kong SH, Choi DY, Park HS, Yang HK, Lee HJ, Kim HC, Kang KW, Sung MH, Lim YT (2012) Near-infrared emitting polymer nanogels for efficient sentinel lymph node mapping. ACS Nano 6:7820–7831
Sutherland SE, Resnick MI, Maclennan GT, Goldman HB (2002) Does the size of the surgical margin in partial nephrectomy for renal cell cancer really matter? J Urol 167:61–64
Kim SP, Thompson RH, Boorjian SA, Weight CJ, Han LC, Murad MH, Shippee ND, Erwin PJ, Costello BA, Chow GK, Leibovich BC (2012) Comparative effectiveness for survival and renal function of partial and radical nephrectomy for localized renal tumors: a systematic review and meta-analysis. J Urol 188:51–57
Sun M, Trinh QD, Bianchi M, Hansen J, Hanna N, Abdollah F, Shariat SF, Briganti A, Montorsi F, Perrotte P, Karakiewicz PI (2012) A non-cancer-related survival benefit is associated with partial nephrectomy. Eur Urol 61:725–731
Lam JS, Bergman J, Breda A, Schulam PG (2008) Importance of surgical margins in the management of renal cell carcinoma. Nat Clin Pract Urol 5:308–317
Marszalek M, Carini M, Chlosta P, Jeschke K, Kirkali Z, Knuchel R, Madersbacher S, Patard JJ, Van Poppel H (2012) Positive surgical margins after nephron-sparing surgery. Eur Urol 61:757–763
Di Pierro GB, Tartaglia N, Aresu L, Polara A, Cielo A, Cristini C, Grande P, Gentile V, Grosso G (2014) Laparoscopic partial nephrectomy for endophytic hilar tumors: feasibility and outcomes. Eur J Surg Oncol 40:769–774
Autorino R, Khalifeh A, Laydner H, Samarasekera D, Rizkala E, Eyraud R, Stein RJ, Haber GP, Kaouk JH (2014) Robot-assisted partial nephrectomy (RAPN) for completely endophytic renal masses: a single institution experience. BJU Int 113:762–768
Moreno-Noguer F, Lepetit V, Fua P (2007) Accurate non-iterative o (n) solution to the pnp problem. In: IEEE 11th international conference on computer vision. IEEE, pp 1–8
Kim JH, Hong SS, Kim JH, Park HJ, Chang YW, Chang AR, Kwon SB (2012) Safety and efficacy of ultrasound-guided fiducial marker implantation for CyberKnife radiation therapy. Korean J Radiol 13:307–313
Kothary N, Heit JJ, Louie JD, Kuo WT, Loo BW Jr, Koong A, Chang DT, Hovsepian D, Sze DY, Hofmann LV (2009) Safety and efficacy of percutaneous fiducial marker implantation for image-guided radiation therapy. J Vasc Interv Radiol JVIR 20:235–239
Paulus CJ, Haouchine N, Cazier D, Cotin S (2015) Surgical augmented reality with topological changes. International conference on medical image computing and computer-assisted intervention. Springer, New York, pp 413–420
Acknowledgments
Authors are grateful to Christopher Burel, professional in medical English proofreading, for their valuable help in revising the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
SH Kong, N Haouchine, R Soares, A Klymchenko, B Andreiuk, B Marques, G Shabat, T Piechaud, M Diana, S Cotin, and J Marescaux have no conflicts of interest or financial ties to disclose.
Rights and permissions
About this article
Cite this article
Kong, SH., Haouchine, N., Soares, R. et al. Robust augmented reality registration method for localization of solid organs’ tumors using CT-derived virtual biomechanical model and fluorescent fiducials. Surg Endosc 31, 2863–2871 (2017). https://doi.org/10.1007/s00464-016-5297-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-016-5297-8