Abstract
Background
Venous thromboembolic events (VTE) are a morbidity and mortality concern for patients undergoing laparoscopic bariatric surgery. Although VTE prophylaxis is recommended in bariatric surgery, data with regard to monitoring and appropriate dosing of low-molecular-weight heparin are limited. Enoxaparin prophylactic doses ranging from 30 to 60 mg every 12 h have been used for this population. The authors hypothesized that higher prophylactic enoxaparin doses (60 mg) would yield more appropriate heparin antifactor Xa (anti-Xa) concentrations than the 40-mg dosage for bariatric surgery patients.
Methods
Patients undergoing laparoscopic bariatric surgery by two surgeons during a 5-month period at one institution received enoxaprin 40 or 60 mg every 12 h. Anti-Xa levels were obtained 4 h after the first and third doses. Therapeutic levels were defined as 0.18 to 0.44 U/ml. Paired and unpaired t-tests and chi-square tests were used for statistical analysis as appropriate.
Results
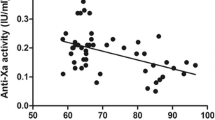
The first-dose mean anti-Xa concentration was 0.173 U/ml in the 40-mg group and 0.261 U/ml in the 60-mg group (p < 0.005), compared with the third-dose mean anti-Xa levels of 0.21 and 0.43 U/ml, respectively (p < 0.001). After the third dose of enoxaparin, the percentage of patients with anti-Xa concentrations who remained subtherapeutic showed a statistically significant difference: 44% in the 40-mg group versus 0% in the 60-mg group (p = 0.02). However, no supratherapeutic anti-Xa concentrations were observed in the 40-mg group, whereas 57% of the third-dose levels in the 60-mg group were supratheraputic. The highest anti-Xa level was 0.54 U/ml, but none of the patients with this level experienced bleeding events.
Conclusions
Enoxaparin 60-mg every 12 h was superior to a dosage of 40 mg every 12 h in achieving therapeutic anti-Xa concentrations and avoiding subtherapeutic anti-Xa levels. However, the 60-mg group had a number of supratherapeutic levels. Future studies evaluating the relationship of anti-Xa concentrations and outcomes with larger numbers of morbidly obese patients are needed.


Similar content being viewed by others
References
Scholten DJ, Hoedema RM, Scholten SW (2002) A comparison of two different prophylactic dose regimens of low-molecular-weight enoxaparin in bariatric surgery. Obes Surg 12:19–24
Hamad GG, Choban P (2005) Enoxaparin for thromboprophylaxis in morbidly obese patients undergoing bariatric surgery: findings of the prophylaxis against VTE outcomes in bariatric surgery patients receiving enoxaparin (PROBE) study. Obes Surg 15:1368–1374
Sapala JA, Wood MH, Schuhknecht MP, Sapala MA (2003) Fatal pulmonary embolism after bariatric operations for morbid obesity: a 24-year retrospective analysis. Obes Surg 13:819–825
Lovenox package insert
Hirsh J, Guyatt G, Albers GW, Schunemann HJ (2004) The seventh ACCP conference on antithrombotic and thrombolytic therapy: evidence-based guidelines. Chest 126:172S–173S
Lexi-Comp Inc, (Lexi-Complete). Lexi-Comp, Inc., 15 March 2007
Enoxaparin. DRUGDEX System. Greenwood Village, CO: Tomson Micromedex. Retrieved 15 March 2007 at http://www.thomsonhc.com
Borkgren-Okonek MJ, Hart RW, Pantano JA, Rantis PC Jr, Guske PJ, Kane JM Jr, Verseman SR, Wallace JW, Sambol N, Vu T (2005) Stratified enoxaparin dosing achieves prophylactic anti-factor Xa concentration in gastric bypass surgery patients. Surg Obes Relat Dis 1:226
Rowan BO, Kuhl DA, Lee MD, Tichansky DS, Madan AK (2008) Anti-Xa levels in bariatric surgery patients receiving prophylactic enoxaparin. Obes Surg 18:162–166
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented at the Society of American Gastrointestinal and Endoscopic Surgeons (SAGES) 2008 annual meeting at Philadelphia, PA.
Rights and permissions
About this article
Cite this article
Simone, E.P., Madan, A.K., Tichansky, D.S. et al. Comparison of two low-molecular-weight heparin dosing regimens for patients undergoing laparoscopic bariatric surgery. Surg Endosc 22, 2392–2395 (2008). https://doi.org/10.1007/s00464-008-9997-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-008-9997-6