Abstract
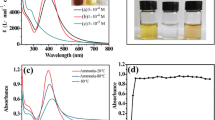
A facile method for the aqueous phase synthesis of cysteine-functionalized silver nanoparticles by potato extract has been reported in the present work. These functionalized nanoparticles were then used for the covalent immobilization of a biomolecule, alkaline phosphatase, on its surface through carbodiimide coupling. Different reaction parameters such as cysteine concentration, reducing agent concentration, temperature, pH and reaction time were varied during the nanoparticles’ formation, and their effects on plasmon resonance were studied using Ultraviolet–visible spectroscopy. Fourier transform infrared spectroscopy was used to confirm the surface modification of silver nanoparticles by cysteine and the particle size analysis was done using particle size analyzer, which showed the average nanoparticles’ size of 61 nm for bare silver nanoparticles and 201 nm for the enzyme-immobilized nanoparticles. The synthesized nanoparticles were found to be highly efficient for the covalent immobilization of alkaline phosphatase on its surface and retained 67 % of its initial enzyme activity (9.44 U/mg), with 75 % binding efficiency. The shelf life of the enzyme-nanoparticle bioconjugates was found to be 60 days, with a 12 % loss in the initial enzyme activity. With a simple synthesis strategy, high immobilization efficiency and enhanced stability, these enzyme-coated nanoparticles have the potential for further integration into the biosensor technology.








Similar content being viewed by others
References
Nair LS, Laurencin CT (2007) Silver nanoparticles: synthesis and therapeutic applications. J Biomed Nanotechnol 3:301–316
Zhang W, Qiao X, Chen J (2007) Synthesis of silver nanoparticles—effects of concerned parameters in water/oil microemulsion. Mater Sci Eng B 142:1–15
Shi W, Ma Z (2010) Amperometric glucose biosensor based on a triangular silver nanoprisms/chitosan composite film as immobilization matrix. Biosens Bioelectron 26:1098–1103
Christensen L, Vivekanandhan S, Misra M, Mohanty AK (2011) Biosynthesis of silver nanoparticles using murraya koenigii (curry leaf): an investigation on the effect of broth concentration in reduction mechanism and particle size. Adv Mater Lett 2:429–434
Chen M, Feng YG, Wang X, Li TC, Zhang JY, Qian DJ (2007) Silver nanoparticles capped by oleylamine: formation, growth, and self-organization. Langmuir 23:5296–5304
Filippo E, Serra A, Manno D (2009) Poly (vinyl alcohol) capped silver nanoparticles as localized surface plasmon resonance-based hydrogen peroxide sensor. Sens Actuators B Chem 138:625–630
Henglein A, Giersig M (1999) Formation of colloidal silver nanoparticles: capping action of citrate. J Phys Chem B 103:9533–9539
Li X, Zhang J, Xu W, Jia H, Wang X, Yang B, Zhao B, Li B, Ozaki Y (2003) Mercaptoacetic acid-capped silver nanoparticles colloid: formation, morphology, and SERS activity. Langmuir 19:4285–4290
Tong MC, Chen W, Sun J, Ghosh D, Chen S (2006) Dithiocarbamate-capped silver nanoparticles. J Phys Chem B 110:19238–19242
Wang W, Chen X, Efrima S (1999) Silver nanoparticles capped by long-chain unsaturated carboxylates. J Phys Chem B 103:7238–7246
Wei G, Wang L, Zhou H, Liu Z, Song Y, Li Z (2005) Electrostatic assembly of CTAB-capped silver nanoparticles along predefined λ-DNA template. Appl Surf Sci 252:1189–1196
Badea M, Curulli A, Palleschi G (2003) Oxidase enzyme immobilisation through electropolymerised films to assemble biosensors for batch and flow injection analysis. Biosens Bioelectron 18:689–698
Hamilton TA, Górnicki SZ, Sussman HH (1979) Alkaline phosphates from human milk. Comparison with isoenzymes from placenta and liver. Biochem J 177:197
Mandal S, Gole A, Lala N, Gonnade R, Ganvir V, Sastry M (2001) Studies on the reversible aggregation of cysteine-capped colloidal silver particles interconnected via hydrogen bonds. Langmuir 17:6262–6268
Song JY, Kim BS (2009) Rapid biological synthesis of silver nanoparticles using plant leaf extracts. Bioprocess Biosyst Eng 32:79–84
Brause R, Moeltgen H, Kleinermanns K (2002) Characterization of laser-ablated and chemically reduced silver colloids in aqueous solution by UV/VIS spectroscopy and STM/SEM microscopy. Appl Phys B 75:711–716
Mie G (1908) Contributions to the optics of turbid media, especially colloidal metal solutions. An Phy 25:377–445
Aryal S, BKC R, Dharmaraj N, Bhattarai N, Kim CH, Kim HY (2006) Spectroscopic identification of SAu interaction in cysteine capped gold nanoparticles. Spectrochim Acta A 63:160–163
Khan MM, Kalathil S, Lee J, Cho MH (2012) Synthesis of cysteine capped silver nanoparticles by electrochemically active biofilm and their antibacterial activities. Bull Korean Chem Soc 33:2592–2596
Nafady A, Afridi H, Sara S, Shah A, Niaz A (2011) Direct synthesis and stabilization of Bi-sized cysteine-derived gold nanoparticles: reduction catalyst for methylene blue. J Iran Chem Soc 8:S34–S43
He S, Yao J, Jiang P, Shi D, Zhang H, Xie S, Pang S, Gao H (2001) Formation of silver nanoparticles and self-assembled two-dimensional ordered superlattice. Langmuir 17:1571–1575
Amin M, Anwar F, Janjua MRSA, Iqbal MA, Rashid U (2012) Green synthesis of silver nanoparticles through reduction with Solanum xanthocarpum L. berry extract: characterization, antimicrobial and urease inhibitory activities against Helicobacter pylori. Int J Mol Sci 13:9923–9941
Ravindran A, Dhas SP, Chandrasekaran N, Mukherjee A (2013) Differential interaction of silver nanoparticles with cysteine. J Exp Nanosci 8:589–595
Petean I, Tomoaia G, Horovitz O, Mocanu A, Tomoaia-Cotisel M (2008) Cysteine mediated assembly of gold nanoparticles. J Optoelectron Adv M 10:2289–2292
Singh M, Singh A, Mandal R, Sinha I (2011) Synthesis of anisotropic silver nanostructures in presence of polyvinyl pyrrolidone (PVP): LSPR and SAXS analysis. Colloids Surf Physicochem Eng Aspects 390:167–172
Morales-Sánchez E, Guajardo-Pacheco J, Noriega-Treviño M, Quintero-González C, Compeán-Jasso M, López-Salinas F, González-Hernández J, Ruiz F (2011) Synthesis of silver nanoparticles using albumin as a reducing agent. Mater Sci Appl 2:578–581
Si S, Mandal TK (2007) Tryptophan-based peptides to synthesize gold and silver nanoparticles: a mechanistic and kinetic study. Chem Eur J 13:3160–3168
Amaladhas TP, Sivagami S, Devi TA, Ananthi N, Velammal SP (2012) Biogenic synthesis of silver nanoparticles by leaf extract of Cassia angustifolia. Adv Nat Sci: Nanosci Nanotechnol 3:045006
Jiang X, Chen W, Chen C, Xiong S, Yu A (2010) Role of temperature in the growth of silver nanoparticles through a synergetic reduction approach. Nanoscale Res Lett 6:32
Frederix F, Bonroy K, Reekmans G, Laureyn W, Campitelli A, Abramov MA, Dehaen W, Maes G (2004) Reduced nonspecific adsorption on covalently immobilized protein surfaces using poly (ethylene oxide) containing blocking agents. J Biochem Biophys Methods 58:67–74
Hsu FY, Yu DS, Chang JC, Chuang CL (2011) Silver nanoparticles as a glucose oxidase immobilization matrix for amperometric glucose biosensor construction. J Chin Chem Soc 58:756–760
Tan S-Z, Hu Y-J, Gong F-C, Cao Z, Xia J-Y, Zhang L (2009) A novel fluorescence sensor based on covalent immobilization of 3-amino-9-ethylcarbazole by using silver nanoparticles as bridges and carriers. Anal Chim Acta 636:205–209
Ren Y, Rivera J, He L, Kulkarni H, Lee D-K, Messersmith P (2011) Facile, high efficiency immobilization of lipase enzyme on magnetic iron oxide nanoparticles via a biomimetic coating. BMC Biotechnol 11:63
Khoshnevisan K, Bordbar A-K, Zare D, Davoodi D, Noruzi M, Barkhi M, Tabatabaei M (2011) Immobilization of cellulase enzyme on superparamagnetic nanoparticles and determination of its activity and stability. Chem Eng J 171:669–673
Kouassi GK, Irudayaraj J, McCarty G (2005) Activity of glucose oxidase functionalized onto magnetic nanoparticles. Biomagn Res Technol 3:1
Li GY, Huang KL, Jiang YR, Yang DL, Ding P (2008) Preparation and characterization of Saccharomyces cerevisiae alcohol dehydrogenase immobilized on magnetic nanoparticles. Int J Biol Macromol 42:405–412
Shaw S-Y, Chen Y-J, Ou J–J, Ho L (2006) Preparation and characterization of Pseudomonas putida esterase immobilized on magnetic nanoparticles. Enzyme Microb Technol 39:1089–1095
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Upadhyay, L.S.B., Verma, N. Synthesis and characterization of cysteine functionalized silver nanoparticles for biomolecule immobilization. Bioprocess Biosyst Eng 37, 2139–2148 (2014). https://doi.org/10.1007/s00449-014-1191-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-014-1191-8