Abstract
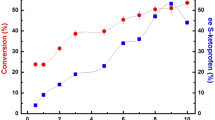
Lipase-catalyzed remote resolution of the tertiary alcohol, citalopram intermediate (diol acetate), has been achieved. The chiral discrimination was obtained by the Novozym435-catalyzed alcoholysis of the primary hydroxyl ester which was four bonds away from the center. The influence of acyl acceptor structure and the organic solvents on the reaction rate and enantioselectivity were investigated. Based on the thermodynamic analysis, the difference of activation free energy between the two enantiomers which dominated the enantioselectivity was significantly affected by the organic solvents, while the acyl acceptor showed less effect. In addition, the enantiomer discrimination was driven by both the difference of activation enthalpy and activation entropy. The thermodynamic analysis provides further insights into the prediction and optimization of enantioselectivity and reaction rate in remote resolution.




Similar content being viewed by others
References
Kourist R, de Maria PD, Bornscheuer UT (2008) Enzymatic synthesis of optically active tertiary alcohols: expanding the biocatalysis toolbox. ChemBioChem 9:491–498
Kourist R, Bornscheuer UT (2011) Biocatalytic synthesis of optically active tertiary alcohols. Appl Microbiol Biotechnol 91:505–517
Bassegoda A, Nguyen GS, Schmidt M, Kourist R, Diaz P, Bornscheuer UT (2010) Rational protein design of Paenibacillus barcinonensis esterase EstA for kinetic resolution of tertiary alcohols. ChemCatChem 2:962–967
Henke E, Bornscheuer UT, Schmid RD, Pleiss J (2003) A molecular mechanism of enantiorecognition of tertiary alcohols by carboxylesterases. ChemBioChem 4:85–493
Effenberger F, Graef BW, Osswald S (1997) Preparation of (S)-naproxen by enantioselective hydrolysis of racemic naproxen amide with resting cells of Rhodococcus erythropolis MP50 in organic solvents. Tetrahedron Asymmetry 8:749–2755
Wiggers M, Holt J, Kourist R, Bartsch S, Arends IWCE, Minnaard AJ, Bornscheuer UT, Hanefeld U (2009) Probing the enantioselectivity of Bacillus subtilis esterase BS2 for tert. alcohols. J Mol Catal B Enzym 60:82–86
Joubert AF, Sanchez C, Larsen F (2000) Citalopram. Hum Psychopharmacol Clin Exp 15:439–451
Moen AR, Ruud K, Anthonsen T (2008) Combination of stereospecific dihydroxylation and enzyme catalyzed enantioselective resolution for synthesis of enantiopure vicinal diols. J Mol Cat B Enzym 50:74–79
Heinze B, Kourist R, Fransson L, Hult K, Bornscheuer UT (2007) Highly enantioselective kinetic resolution of two tertiary alcohols using mutants of an esterase from Bacillus subtilis. Protein Eng Des Sel 20:125–131
Holt J, Arends IWCE, Minnaard AJ, Hanefeld U (2007) Hydrolase-catalysed preparation of chiral alpha, alpha-disubstituted cyanohydrin acetates. Adv Synth Catal 349:1341–1344
Bogeso KP (1987) New phenyl-fluoro-phenyl di-methylamino-propyl-carbinol is new and is an intermediate in preparation of antidepressant di-hydro-iso-benzofuran. US 4650884
Bech SM, Nielsen O, Petersen H, Ahmadian H, Pedersen H, Brosen P (2003) Preparation of escitalopram useful as an antidepressant drug involves liquid chromatographic separation of enantiomers of chiral citalopram with a chiral stationary phase. WO03/006449
Elati CR, Kolla N, Vankawala PJ, Gangula S, Chalamala S, Sundaram V, Bhattacharya A, Vurimidi H, Mathad VT (2007) Substrate modification approach to achieve efficient resolution: didesmethylcitalopram: a key intermediate for escitalopram. Org Process Res Dev 11:289–292
Petersen H, Boegesoe KP, Sommer MB (2001) Method for the preparation of citalopram. US6291689
Solares LF, Brieva R, Quiros M, Llorente I, Bayod M, Gotor V (2004) Enzymatic resolution of a quaternary stereogenic centre as the key step in the synthesis of (S)-(+)-citalopram. Tetrahedron Asymmetry 15:341–345
Taoka N, Kato T, YamamotoS, Yoshida T (2004) Preparation of S- or R-diol compounds and/or opposite enantiomer of acylated diol compounds useful in escitalopram preparation, by selective enzymatic acylation or deacylation of racemic compound. WO014821
Wang SZ, Wu JP, Xu G, Yang LR (2009) Kinetic modelling of lipase-catalyzed remote resolution of citalopram intermediate in solvent-free system. Biochem Eng J 45:113–119
Angoli M, Barilli A, Lesma G, Passarella D, Riva S, Silvani A, Danieli B (2003) Remote stereocenter discrimination in the enzymatic resolution of piperidine-2-ethanol. Short enantioselective synthesis of sedamine and allosedamine. J Org Chem 68:9525–9527
Santaniello E, Casati S, Ciuffreda P (2006) Lipase-catalyzed deacylation by alcoholysis: a selective, useful transesterification reaction. Curr Org Chem 10:1095–1123
Micaelo NM, Soares CM (2007) Modeling hydration mechanisms of enzymes in nonpolar and polar organic solvents. FEBS J 274:2424–2436
Overbeeke PLA, Ottosson J, Hult K, Jongejan JA, Duine JA (1999) The temperature dependence of enzymatic kinetic resolutions reveals the relative contribution of enthalpy and entropy to enzymatic enantioselectivity. Biocatal Biotransform 17:61–79
Canelas AB, Ras C, ten Pierick A, van Gulik WM, Heijnen JJ (2011) An in vivo data-driven framework for classification and quantification of enzyme kinetics and determination of apparent thermodynamic data. Metab Eng 13:294–306
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Key Program) (No. 20936002), Key Project of Chinese National Programs for Fundamental Research and Development (No. 2011CB710800), and Hi-Tech Research and Development Program of China (No. 2011AA02A209).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, SZ., Wu, JP., Xu, G. et al. Lipase-catalyzed remote kinetic resolution of citalopram intermediate by asymmetric alcoholysis and thermodynamic analysis. Bioprocess Biosyst Eng 35, 1043–1048 (2012). https://doi.org/10.1007/s00449-012-0690-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-012-0690-8