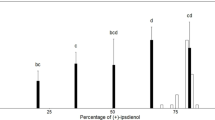
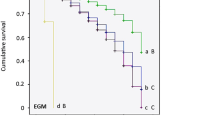
Abstract
Timing of arrival at a resource often determines an individual’s reproductive success. Tree-killing bark beetles can reproduce in healthy trees by attacking in adequate numbers to overcome host defences that could otherwise be lethal. This process is mediated by aggregation and antiaggregation pheromones. Beetles that arrive early in such a “mass attack” must contend with undiminished tree defences, and produce enough pheromones to attract more beetles, but have a head start on gallery construction and egg-laying. Beetles that arrive late may be impeded by competition and diminishing availability of phloem, but should experience fewer costs associated with pheromone production and battling tree defences. We investigated relationships between timing of arrival, body size, pheromone production and fitness in the southern pine beetle, Dendroctonus frontalis. In field experiments, we captured beetles that arrived early (pioneers) and late on slash pine trees, Pinus elliottii, and measured pheromone amounts in their hindguts. We marked gallery entrances of beetles as they landed on a tree and measured their reproductive success after the attack terminated. We found no difference in body size or pheromone amounts between early and late arrivers. Most beetles arrived at the middle of the attack sequence, and excavated longer galleries per day than early arrivers. The number of offspring produced per day by beetles that established galleries midway through mass attack was higher than those that arrived early or very late in the sequence. Our results suggest that beetles do not exhibit adaptive phenotypic plasticity in pre-landing pheromone production, depending on the extent of previous colonisation of a host. Rather, it appears that stabilising selection favours beetles that attack in the middle of the sequence, and contributes to attack synchrony. Synchronous attack on trees is essential before population booms characteristic of tree-killing bark beetles can occur in nature.



Similar content being viewed by others
References
Aukema BH, Raffa KF (2004) Gender- and sequence-dependent predation within group colonizers of defended plants: a constraint on cheating among bark beetles? Oecologia 138:253–258
Avilés L, Tufino P (1998) Colony size and individual fitness in the social spider Anelosimus eximus. Am Nat 152:403–418
Avilés L, Abbot P, Cutter AD (2002) Population ecology, nonlinear dynamics, and social evolution. I. Associations among nonrelatives. Am Nat 159:115–127
Axelrod R, Hamilton WD (1981) The evolution of cooperation. Science 211:1390–1396
Ayres BD, Ayres MP, Abrahmson MD, Teale SA (2001) Resource partitioning and overlap in three sympatric species of Ips bark beetles. Oecologia 128:443–453
Barkawi LS, Franke W, Blomquist GJ, Seybold SJ (2003) Frontalin: de novo biosynthesis of an aggregation pheromone component by Dendroctonus spp. bark beetles (Coleoptera: Scolytidae). Insect Biochem Mol Biol 33:773–788
Berryman AA, Dennis B, Raffa KF, Stenseth NS (1985) Evolution of optimal group attack with particular reference to bark beetles (Coleoptera: Scolytidae). Ecology 66:898–903
Birgersson GF, Schlyter F, Bergstrom G, Lofqvist J (1988) Individual variation in aggregation pheromone content of the bark beetle, Ips typographus. J Chem Ecol 14:1737–1761
Blums P, Nichols JD, Hines JE, Lindberg MS, Mednis A (2005) Individual quality, survival variation and patterns of phenotypic selection on body condition and timing of nesting in birds. Oecologia 143:365–376
Byers JA (2005) A cost of alarm pheromone production in cotton aphids, Aphis gossypii. Naturwissenschaften 92:69–72
Caraco T, Wolf LL (1975) Ecological determinants of group sizes of foraging lions. Am Nat 109:343–352
Clarke AE, Webb JW, Franklin RT (1979) Fecundity of the southern pine beetle in laboratory pine bolts. Ann Entomol Soc Am 72:229–231
Clarke SR, Salom SM, Billings RF, Berisford CW, Upton WW, McClellan QC, Dalusky MJ (1999) A scentsible approach to controlling southern pine beetles. J Forestry 97:26–31
Cooper ME, Stephen FM (1978) Parental adult reemergence in southern pine beetle populations. Environ Entomol 7:574–577
Coster JE (1970) Production of aggregating pheromones in re-emerged parent females of the southern pine beetle. Ann Entomol Soc Am 63:1186–1187
Dahlsten DL, Six DL, Rowney DL, Lawson AB, Erbilgin N, Raffa KF (2004) Attraction of Ips pini (Coleoptera: Scolyitdae) and its predators to natural attractants and synthetic semiochemicals in northern California: implications for population monitoring. Environ Entomol 33:1554–1561
Day RW, Quinn GP (1989) Comparisons of treatments after analysis of variance in ecology. Ecol Monogr 59:433–463
Dickerson BR, Brinck KW, Willson MF, Bentzen P, Quinn TP (2005) Relative importance of salmon body size and arrival time at breeding grounds to reproductive success. Ecology 86:347–352
Drooz AT (ed) (1985) Insects of eastern forests. USDA Forest Service Miscellaneous Publication No. 1426, p 608
Dugatkin LA (1997) The evolution of cooperation. Bioscience 47:355–362
Foltz JL, Mayyasi AM, Hain FP, Coulson RN, Martin WC (1976) Egg-gallery length relationship and within-tree analyses for the southern pine beetle, Dendroctonus frontalis (Coleoptera: Scolytidae) Zimm. Can Entomol 108:341–352
Foster KR (2004) Diminishing returns in social evolution: the not so tragic commons. J Evol Biol 17:1058–1072
Hughes PR (1973) Dendroctonus: production of pheromones and related compounds in response to host monoterpenes. Z Angew Entomol 73:294–312
Hughes PR (1975) Pheromones of Dendroctonus: origin of a-pinene oxidation products present in emergent adults. J Insect Physiol 21:687–691
Hunt DWA, Borden JH, H.D. Pierce J, Slessor KN, King GGS, Czyewska EK (1986) Sex-specific production of ipsdienol and myrcenol by Dendroctonus ponderosae (Coleoptera: Scolytidae) exposed to myrcene vapours. J Chem Ecol 12:1579–1586
Johansson BG, Jones TM, Widemo F (2005) Cost of pheromone production in a lekking Drosophila. Anim Behav 69:851–858
Lindgren BS (1983) A multiple funnel trap for scolytid bark beetles (Coleoptera). Can Entomol 115:229–302
Marzluff JM, Heinrich B (1991) Foraging by common ravens in the presence and absence of territory holders: an experimental analysis of social foraging. Anim Behav 42:755–770
McGhehey JH (1971) Female size and egg production of the mountain pine beetle Dendroctonus ponderosae Hopkins. Nor For Res Cent, Edmonton, Alberta Inf Rep NOR-X-9
Miller DR, Borden JH, Slessor KN (1989) Inter- and intrapopulation variation of the pheromone ipsdienol produced by male pine engravers, Ips pini (Say) (Coleoptera: Scolytidae). J Chem Ecol 15:233–247
Osgood EAJ, Clark EW (1963) Methods of sexing and sex ratios of the southern pine beetle, Dendroctonus frontalis Zimmermann. Can Entomol 95:1106–1109
Payne TL, Coster JE, Richerson JV, Edson LJ, Hart ER (1978) Field responses of the southern pine beetle to behavioral chemicals. Environ Entomol 7:578-582
Pitman GB, Vité JP, Kinzer GW, Fentimann AFF Jr. (1969) Specificity of population-aggregating pheromones in Dendroctonus. J Insect Physiol 15:363–366
Price TS, Doggett C, Pye JM, Smith B (1998) A history of southern pine beetle outbreaks in the southeastern United States. Georgia Forestry Commission, Macon, GA, 71 pp
Pureswaran DS (1999) Dynamics of pheromone production and communication in the mountain pine beetle, Dendroctonus ponderosae Hopkins and the pine engraver, Ips pini (Say). Biological Sciences, Simon Fraser University, Burnaby, BC, Canada
Pureswaran DS, Borden JH (2003) Is bigger better? Size and pheromone production in the mountain pine beetle, Dendroctonus ponderosae Hopkins (Coleoptera: Scolytidae). J Insect Behav 16:765–782
Raffa KF (2001) Mixed messages across multiple trophic levels: the ecology of bark beetle chemical communication systems. Chemoecology 11:49–65
Raffa KF, Berryman AA (1983) The role of host plant resistance in the colonization behavior and ecology of bark beetles (Coleoptera: Scolytidae). Ecolog Monog 53:27–49
Raffa KF, Berryman AA (1987) Interacting selective pressures in conifer-bark beetle systems: a basis for reciprocal adaptations? Am Nat 129:234–262
Raffa KF, Phillips TW, Salom SM (1993) Strategies and mechanisms of host colonization by bark beetles. In: Schowalter TD, Filip GM (eds) Beetle-pathogen interactions in conifer forests. Academic Press, New York, pp 103–128
Reeve JD (1997) Predation and bark beetle dynamics. Oecologia 112:48–54
Reeve JD, Strom BL (2004) Statistical problems encountered in trapping studies of scolytids and associated insects. J Chem Ecol 30:1575–1590
Reid RW (1962) Biology of the mountain pine beetle, Dendroctonus monticolae Hopkins, in the east Kootenay region of British Columbia. I. Life cycle, brood development and flight periods. Can Entomol 94:531–538
Reid ML, Roitberg BD (1995) Effects of body size on investment in individual broods by male pine engravers (Coleoptera: Scolytidae). Can J Zool 73:1396–1401
Renwick JAA, Hughes PR, Pitman GB, Vité JP (1976) Oxidation products of terpenes identified from Dendroctonus and Ips bark beetles. J Insect Physiol 22:725–727
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Robertson IC, Roitberg BD (1998) Duration of paternal care in pine engraver beetles: Why do larger males care less? Behav Ecol Sociobiol 43:379–386
Safranyik L (1976) Size- and sex-related emergence and survival in cold storage of mountain pine beetles. Can Entomol 108:209–212
SAS (1999–2001) SAS Institute Inc. SAS/STAT Users guide release. Cary, NC
Schlyter F, Birgersson G (1989) Individual variation in bark beetle and moth pheromones: a comparison and an evolutionary background. Holarctic Ecol 12:457–465
Sullivan BT (2005) Electrophysiological and behavioral responses of southern pine beetle, Dendroctonus frontalis Zimmermann (Coleoptera: Curculionidae), to volatiles isolated from conspecifics. J Econ Entomol 98:2067–2078
Svihra P, Paine TD, Birch MC (1980) Interspecific olfactory communications in sourthern pine beetles. Naturwissenschaften 67S
Trivers RL (1971) The evolution of reciprocal altruism. Quat Rev Biol 46:35–57
Vanderwel D, Gries G, Singh SM, Borden JH, Oehlschlager AC (1992) (E)- and (Z)-6-nonen-2-one: biosynthetic precursors of endo- and exo-brevicomin in two bark beetles (Coleoptera: Scolytidae). J Chem Ecol 18:1389–1404
Vité JP, Billings RF, Ware CW, Mori K (1985) Southern pine beetle: enhancement or inhibition of aggregation response mediated by enantiomers of endo-brevicomin. Naturwissenschaften 72:99
Wood DL (1982) The role of pheromones, kairomones and allomones in the host selection and colonization of bark beetles. Annu Rev Entomol 27:411–446
Zhang QH, Birgersson G, Schlyter F, Chen GF (2000) Pheromone components in the larch bark beetle, Ips cembrae, from China: quantitative variation among attack phases and individuals. J Chem Ecol 26:841–858
Acknowledgements
We thank P. Marino and L. Evans for assistance in the field, L. Avilés for theoretical discussions, K. Raffa and R. Karban for reviews. This project was supported by CSREES NRI 2004-35302-14820 to MPA and a Natural Sciences and Engineering Research Council of Canada Postdoctoral Fellowship to DSP.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Richard Karban
Rights and permissions
About this article
Cite this article
Pureswaran, D.S., Sullivan, B.T. & Ayres, M.P. Fitness consequences of pheromone production and host selection strategies in a tree-killing bark beetle (Coleoptera: Curculionidae: Scolytinae) . Oecologia 148, 720–728 (2006). https://doi.org/10.1007/s00442-006-0400-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-006-0400-9