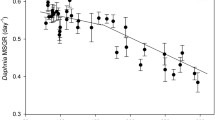
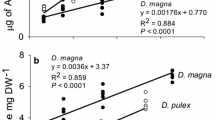
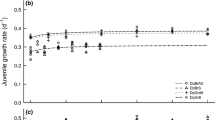
Abstract
Animals that maintain near homeostatic elemental ratios may get rid of excess ingested elements from their food in different ways. C regulation was studied in juveniles of Daphnia magna feeding on two Selenastrum capricornutum cultures contrasting in P content (400 and 80 C:P atomic ratios). Both cultures were labelled with 14C in order to measure Daphnia ingestion and assimilation rates. No significant difference in ingestion rates was observed between P-low and P-rich food, whereas the net assimilation of 14C was higher in the treatment with P-rich algae. Some Daphnia were also homogeneously labelled over 5 days on radioactive algae to estimate respiration rates and excretion rates of dissolved organic C (DOC). The respiration rate for Daphnia fed with high C:P algae (38.7% of body C day-1) was significantly higher than for those feeding on low C:P algae (25.3% of body C day-1). The DOC excretion rate was also higher when animals were fed on P-low algae (13.4% of body C day-1) than on P-rich algae (5.7% of body C day-1) . When corrected for respiratory losses, total assimilation of C did not differ significantly between treatments (around 60% of body C day-1). Judging from these experiments, D. magna can maintain its stoichiometric balance when feeding on unbalanced diets (high C:P) primarily by disposing of excess dietary C via respiration and excretion of DOC.






Similar content being viewed by others
References
Andersen T, Hessen DO (1991) Carbon, nitrogen, and phosphorus content of freshwater zooplankton. Limnol Oceanogr 36:807–814
Båmstedt U (1986) Chemical composition and energy content. In: Corner EDS, O'Hara (eds) The biological chemistry of marine copepods. Oxford University Press, Oxford, pp 1–58
Biddanda B, Ogdahl M, Cotner J (2001) Dominance of bacterial metabolism in oligotrophic relative to eutrophic waters. Limnol Oceanogr 46:730–739
Billen G (1984) Heterotrophic utilization and regeneration of nitrogen. In: Hobbie JE, Williams PJleB (eds) Heterotrophic activity in the sea. Plenum Press, New York, pp 313–355
Cimbleris ACP, Kalff J (1998) Planktonic bacterial respiration as a function of C:N:P ratios accross temperate lakes. Hydrobiologia 384:89–100
Conover RJ, Francis V (1973) The use of radioactive isotopes to measure the transfer of materials in aquatic food chains. Mar Biol 18:272–283
Copping AE, Lorenzen CJ (1980) Carbon budget of a marine phytoplankton-herbivore system with carbon-14 as a tracer. Limnol Oceanogr 25:873–882
DeMott WR, Gulati RD, Siewertsen K (1998) Effects of phosphorus-deficient diets on the carbon and phosphorus balance of Daphnia magna. Limnol Oceanogr 43:1147–1161
Elser JJ, Hassett RP (1994) A stoichiometric analysis of the zooplankton-phytoplankton interaction in marine and freshwater ecosystems. Nature 370:211–213
Elser JJ, Dobberfuhl DR, MacKay NA, Schampel JH (1996) Organism size, life history, and N:P stoichiometry: towards a unified view of cellular and ecosystem processes. BioScience 46:674–684
Elser JJ, Fagan WF, Denno RF, Dobberfuhl DR, Folarin A, Huberty A, Interlandi S, Kilham SS, McCauley E, Schulz KL, Siemann EH, Sterner RW (2000) Nutritional constraints in terrestrial and freshwater food webs. Nature 408:578–580
Gardner WS, Paffenhöfer G-A (1982) Nitrogen regeneration by the subtropical marine copepod Eucalanus pileatus. J Plankton Res 4:725–734
Goldman JC, Caron DA, Dennett MR (1987) Regulation of gross growth efficiency and ammonium regeneration in bacteria by substrate C:N ratio. Limnol Oceanogr 32:1239–1252
Goulden CE, Place AR (1990) Fatty acid synthesis and accumulation rates in daphniids. J Exp Zool 256:168–178
Groeger AW, Schram MD, Marzolf GR (1991) Influence of food quality on growth and reproduction in Daphnia. Freshwater Biol 26:11–19
Harder W, Dijkhuizen L (1983) Physiological responses to nutrient limitation. Annu Rev Microbiol 37:1–23
Hessen DO (1992) Nutrient element limitation of zooplankton production. Am Nat 140:799–814
Hessen DO (1997) Stoichiometry in food webs—Lotka revisited. Oikos 79:195–200
Hessen DO, Faafeng BA (2000) Elemental ratios in freshwater seston; implications for community structure and energy transfer in food webs. Arch Hydrobiol Adv Limnol 55:349–363
Hessen DO, Faerøvig PJ, Andersen T (2002) Light, nutrients, and P:C ratios in algae: grazer performance related to food quality and food quantity. Ecology 83:1886–1898
Hopkinson CS, Sherr B, Wiebe WJ (1989) Size-fractionated metabolism of coastal microbial plankton. Mar Ecol Prog Ser 51:155–166
Hygum BH, Petersen JW, Søndergaard M (1997) Dissolved organic carbon released by zooplankton grazing activity—a high-quality substrate pool for bacteria. J Plankton Res 19:97–111
Jumars PA, Penry DL, Baross JA, Perry MJ, Frost BW (1989) Closing the microbial loop: dissolved carbon pathway to heterotrophic bacteria from incomplete ingestion, digestion and absorption in animals. Deep-Sea Res PT A 36:483–495
Kilham SS, Kreeger DA, Lynn SG, Goulden CE, Herrera L (1998) COMBO: a defined freshwater culture medium for algae and zooplankton. Hydrobiologia 377:147–159
Lampert W (1975) A tracer study on the carbon turnover of Daphnia pulex. Verh Int Verein Limnol 19:2913–2921
Lampert W (1977) Studies on the carbon balance of Daphnia pulex as related to environmental conditions. I. Methodological problems of the use of 14C for the measurement of carbon assimilation. Arch Hydrobiol [Suppl] 48:287-309
Lampert W (1978) Release of dissolved organic matter by grazing zooplankton. Limnol Oceanogr 23:831–834
Lampert W (1987) Feeding and nutrition in Daphnia. Mem Ist Ital Idrobiol Dott Marco de Marchi Pallanza Italy 45:143–192
Lampert W, Bohrer R (1984) Effect of food availability on the respiratory quotient of Daphnia magna. Comp Biochem Phys A 78:221–223
Lampert W, Gabriel W (1984) Tracer kinetics in Daphnia: an improved two-compartment model and experimental test. Arch Hydrobiol 100:1–20
Lancelot C, Billen G (1985) Carbon-nitrogen relationships in nutrient metabolism of coastal marine ecosystems. Adv Aquat Microbiol 3:263–321
Olsen Y, Varum KM, Jensen A (1986) Some characteristics of the carbon compounds released by Daphnia. J Plankton Res 8:505–517
Park J-Ch, Aizaki M, Fukushima T, Otsuki A (1997) Production of labile and refractory dissolved organic carbon by zooplankton excretion: an experimental study using large outdoor continuous flow-through ponds. Can J Fish Aquat Sci 54:434–443
Plath K, Boersma M (2001) Mineral limitation of zooplankton: stoichiometric constraints and optimal foraging. Ecology 82:1260–1269
Reynolds CS (1997) Vegetation processes in the pelagic: a model for ecosystem theory. In: Kinne O (ed) Excellence in ecology, vol 9. Ecology Institute, Oldendorf/Luhe, Germany
Richardot M, Debroas D, Thouvenot A, Sargos D, Dévaux J (2001) Influence of cladoceran grazing activity on dissolved organic matter, enzymatic hydrolysis and bacterial growth. J Plankton Res 23:1249–1261
Rosenstock B, Simon M (2001) Sources and sinks of dissolved free amino acids and protein in a large and deep mesotrophic lake. Limnol Oceanogr 46:644–654
Rothhaupt KO (1995) Algal nutrient limitation affects rotifer growth rate but not ingestion rate. Limnol Oceanogr 40:1201–1208
Salisbury FB, Ross CW (1992) Plant physiology, 4th edn. Wadsmorth Belmont, Calif.
Sterner RW (1993) Daphnia growth on varying quality of Scenedesmus: mineral limitation of zooplankton. Ecology 74:2351–2360
Sterner RW (1997) Modelling interactions of food quality and quantity in homeostatic consumers. Freshwater Biol 38:473–481
Sterner RW (1998) Demography of a natural population of Daphnia retrocurva in a lake with low food quality. J Plankton Res 20:471–489
Sterner RW, Hessen DO (1994) Algal nutrient limitation and the nutrition of aquatic herbivores. Annu Rev Ecol Syst 25:1–29
Sterner RW, Smith RF (1993) Clearance, ingestion and release of N and P by Daphnia obtusa feeding on Scenedesmus acutus of varying quality. Bull Mar Sci 53:228–239
Sterner RW, Hagemeier DD, Smith RF, Smith WL (1992) Lipid-ovary indices in food-limited Daphnia. J Plankton Res 14:1449–1460
Sterner RW, Hagemeier DD, Smith WL, Smith RF (1993) Phytoplankton nutrient limitation and food quality for Daphnia. Limnol Oceanogr 38:857–871
Sterner RW, Clasen J, Lampert W, Weisse T (1998) Carbon:phosphorus stoichiometry and food chain production. Ecol Lett 1:146–150
Strom SL, Benner R, Ziegler S, Dagg MJ (1997) Planktonic grazers are a potentially important source of marine dissolved organic carbon. Limnol Oceanogr 42:1364–1374
Tessier AJ, Goulden CE (1982) Estimating food limitation in cladoceran populations. Limnol Oceanogr 27:707–717
Urabe J, Watanabe Y (1992) Possibility of N or P limitation for planktonic cladocerans: an experimental test. Limnol Oceanogr 37:244–251
Urabe J, Clasen J, Sterner RW (1997) Phosphorus limitation of Daphnia growth: Is it real? Limnol Oceanogr 42:1436–1443
Van Donk E, Hessen DO (1993) Grazing resistance in nutrient-stressed phytoplankton. Oecologia 93:508–511
Vrede T, Andersen T, Hessen DO (1999) Phosphorus distribution in three crustacean zooplankton species. Limnol Oceanogr 44:225–229
Acknowledgements
The authors would like to thank Prof. J. Henrard for his help in resolving the equation systems. We are grateful to S. Diehl for his helpful suggestions and improvements to an earlier draft of the manuscript. F. D. was supported by an exchange Linkecol grant provided by the European Science Foundation, which funded travel and accommodation for the duration of the experiments at the University of Oslo.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A
Demonstration of equations modelling evolution over time of total radioactivity in homogeneously labelled Daphnia feeding on unlabelled algae
The kinetics of the tracer in each pool is described by a system of two differential equations (see Table 1 for explanation of symbols):
We know that this kind of differential system has a solution of the type:
In using this type of solution in the system, we found:
In simplifying this by exp(λ j t), we found algebraic equations for a and b:
This system will give a non-trivial solution only if the next determinant
is equal to 0. This gives a quadratic equation in λ j with two solutions:
and
We then rewrite the equations in system 8 (Eq. 8) under their decomposed form:
We then rewrite the second equation in system 9 (Eq. 9) with decomposition of λ j into λ 1 and λ 2:
and
If we insert these two equations into system 12 (Eq. 12), we obtain a new system of two linear equations with two variables a 1 and a 2:
Under initial conditions, system 13 (Eq. 13) becomes:
Thus,
Appendix B
Demonstration of equations modelling evolution over time of radioactivity in unlabelled Daphnia feeding on labelled algae
The kinetics of the tracer in each pool is described by (see Table 1 for explanation of symbols):
Let us define
and
that we insert in the system of differential equations:
We can now determine α et β which nullify the non-homogeneous terms:
Both equations of system 17 (Eq. 17) become homogeneous:
As for the problem defined in Appendix A, we know that the solution of this linear differential system is a linear combination:
where, as in Appendix A, b 1 and b 2 are defined, respectively, by \( b_1 = {{{{\rho _{12} a_1 } \mathord{\left/ {\vphantom {{\rho _{12} a_1 } M}} \right. \kern-\nulldelimiterspace} M}} \over {\lambda _1 + {{\rho _{21} } \mathord{\left/ {\vphantom {{\rho _{21} } S}} \right. \kern-\nulldelimiterspace} S}}} \), and \( b_2 = {{{{\rho _{12} a_2 } \mathord{\left/ {\vphantom {{\rho _{12} a_2 } M}} \right. \kern-\nulldelimiterspace} M}} \over {\lambda _2 + {{\rho _{21} } \mathord{\left/ {\vphantom {{\rho _{21} } S}} \right. \kern-\nulldelimiterspace} S}}} \), and λ 1 and λ 2, respectively, by Eqs. 10 and 11.
Under initial conditions, m 0=0 and s 0=0.
The insertion of Eqs. 15 and 16 into system 18 (Eq. 18) gives:
Thus,
Rights and permissions
About this article
Cite this article
Darchambeau, F., Faerøvig, P.J. & Hessen, D.O. How Daphnia copes with excess carbon in its food. Oecologia 136, 336–346 (2003). https://doi.org/10.1007/s00442-003-1283-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-003-1283-7