Abstract
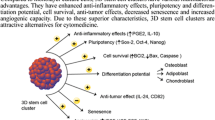
Mesenchymal stem cell (MSC) transplantation is a promising treatment of many diseases. However, conventional techniques with cells being cultured as a monolayer result in slow cell proliferation and insufficient yield to meet clinical demands. Three-dimensional (3D) culture systems are gaining attention with regard to recreating a complex microenvironment and to understanding the conditions experienced by cells. Our aim is to establish a novel 3D system for the culture of human umbilical cord MSCs (hUC-MSCs) within a real 3D microenvironment but with no digestion or passaging. Primary hUC-MSCs were isolated and grown in serum-free medium (SFM) on a suspension Rocker system. Cell characteristics including proliferation, phenotype and multipotency were recorded. The therapeutic effects of 3D-cultured hUC-MSCs on carbon tetrachloride (CCl4)-induced acute liver failure in mouse models were examined. In the 3D Rocker system, hUC-MSCs formed spheroids in SFM and maintained high viability and active proliferation. Compared with monolayer culture, the 3D-culture system yielded more hUC-MSCs cells within the same volume. The spheroids expressed higher levels of stem cell markers and displayed stronger multipotency. After transplantation into mouse, 3D hUC-MSCs significantly promoted the secretion of interferon-γ and interleukin-6 but inhibited that of tumor necrosis factor-α, thereby alleviating liver necrosis and promoting regeneration following CCl4 injury. The 3D culture of hUC-MSCs thus promotes cell yield and stemness maintenance and represents a promising strategy for hUC-MSCs expansion on an industrial scale with great potential for cell therapy and biotechnology.






Similar content being viewed by others
References
Anzalone R, Iacono ML, Corrao S, Magno F, Loria T, Cappello F, Zummo G, Farina F, La Rocca G (2010) New emerging potentials for human Wharton’s jelly mesenchymal stem cells: immunological features and hepatocyte-like differentiative capacity. Stem Cells Dev 19:423–438
Bao J, Fisher JE, Lillegard JB, Wang W, Amiot B, Yu Y, Dietz AB, Nahmias Y, Nyberg SL (2013) Serum-free medium and mesenchymal stromal cells enhance functionality and stabilize integrity of rat hepatocyte spheroids. Cell Transplant 22:299–308
Baraniak PR, McDevitt TC (2012) Scaffold-free culture of mesenchymal stem cell spheroids in suspension preserves multilineage potential. Cell Tissue Res 347:701–711
Bieback K, Kern S, Klüter H, Eichler H (2004) Critical parameters for the isolation of mesenchymal stem cells from umbilical cord blood. Stem Cells 22:625–634
Bonab MM, Alimoghaddam K, Talebian F, Ghaffari SH, Ghavamzadeh A, Nikbin B (2006) Aging of mesenchymal stem cell in vitro. BMC 7:14
Bortolomai I, Canevari S, Facetti I, De Cecco L, Castellano G, Zacchetti A, Alison MR, Miotti S (2010) Tumor initiating cells. Cell Cycle 9:1194–1206
Carpenedo RL, Sargent CY, McDevitt TC (2007) Rotary suspension culture enhances the efficiency, yield, and homogeneity of embryoid body differentiation. Stem Cells 25:2224–2234
Cheng NC, Wang S, Young TH (2012) The influence of spheroid formation of human adipose-derived stem cells on chitosan films on stemness and differentiation capabilities. Biomaterials 33:1748–1758
Deans RJ, Moseley AB (2000) Mesenchymal stem cells: biology and potential clinical uses. Exp Hematol 28:875–884
English K, Barry FP, Field-Corbett CP, Mahon BP (2007) IFN-gamma and TNF-alpha differentially regulate immunomodulation by murine mesenchymal stem cells. Immunol Lett 110:91–100
Francese R, Fiorina P (2010) Immunological and regenerative properties of cord blood stem cells. Clin Immunol 136:309–322
Friedenstein AJ, Gorskaja J, Kulagina N (1976) Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol 4:267–274
Gregory CA, Grady Gunn W, Peister A, Prockop DJ (2004) An Alizarin red-based assay of mineralization by adherent cells in culture: comparison with cetylpyridinium chloride extraction. Anal Biochem 329:77–84
Gurkan UA, El Assal R, Yildiz SE, Sung Y, Trachtenberg AJ, Kuo WP, Demirci U (2014) Engineering anisotropic biomimetic fibrocartilage microenvironment by bioprinting mesenchymal stem cells in nanoliter gel droplets. Mol Pharm 11:2151–2159
Hartmann I, Hollweck T, Haffner S, Krebs M, Meiser B, Reichart B, Eissner G (2010) Umbilical cord tissue-derived mesenchymal stem cells grow best under GMP-compliant culture conditions and maintain their phenotypic and functional properties. J Immunol Methods 363:80–89
Krontiras P, Gatenholm P, Hägg DA (2014) Adipogenic differentiation of stem cells in three‐dimensional porous bacterial nanocellulose scaffolds. J Biomed Mater Res B Appl Biomater (in press)
Kuo TK, Hung SP, Chuang CH, Chen CT, Shih YR, Fang SC, Yang VW, Lee OK (2008) Stem cell therapy for liver disease: parameters governing the success of using bone marrow mesenchymal stem cells. Gastroenterology 134:2111–2121
La Rocca G, Anzalone R, Corrao S, Magno F, Loria T, Lo Iacono ML, Di Stefano A, Giannuzzi P, Marasà L, Cappello F, Zummo G, Farina F (2009) Isolation and characterization of Oct-4+/HLA-G+ mesenchymal stem cells from human umbilical cord matrix: differentiation potential and detection of new markers. Histochem Cell Biol 131:267–282
Lam MT, Longaker MT (2012) Comparison of several attachment methods for human iPS, embryonic and adipose-derived stem cells for tissue engineering. J Tissue Eng Regen Med 6 (Suppl 3):s80–s86
Marcus-Sekura C, Richardson JC, Harston RK, Sane N, Sheets RL (2011) Evaluation of the human host range of bovine and porcine viruses that may contaminate bovine serum and porcine trypsin used in the manufacture of biological products. Biologicals 39:359–369
Nishiguchi A, Matsusaki M, Asano Y, Shimoda H, Akashi M (2014) Effects of angiogenic factors and 3D-microenvironments on vascularization within sandwich cultures. Biomaterials 35:4739–4748
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284:143–147
Rim J-S, Mynatt RL, Gawronska-Kozak B (2005) Mesenchymal stem cells from the outer ear: a novel adult stem cell model system for the study of adipogenesis. FASEB J 19:1205–1207
Romanov YA, Svintsitskaya VA, Smirnov VN (2003) Searching for alternative sources of postnatal human mesenchymal stem cells: candidate MSC-like cells from umbilical cord. Stem Cells 21:105–110
Su H, Wang L, Huang W, Qin D, Cai J, Yao X, Feng C, Li Z, Wang Y, So KF, Pan G, Wu W, Pei D (2013) Immediate expression of Cdh2 is essential for efficient neural differentiation of mouse induced pluripotent stem cells. Stem Cell Res 10:338–348
Trivedi P, Hematti P (2008) Derivation and immunological characterization of mesenchymal stromal cells from human embryonic stem cells. Exp Hematol 36:350–359
Tunma S, Inthanon K, Chaiwong C, Pumchusak J, Wongkham W, Boonyawan D (2013) Improving the attachment and proliferation of umbilical cord mesenchymal stem cells on modified polystyrene by nitrogen-containing plasma. Cytotechnology 65:119–134
Wagner W, Horn P, Castoldi M, Diehlmann A, Bork S, Saffrich R, Benes V, Blake J, Pfister S, Eckstein V, Ho AD (2008) Replicative senescence of mesenchymal stem cells: a continuous and organized process. PLoS One 3:e2213
Wang W, Itaka K, Ohba S, Nishiyama N, Chung UI, Yamasaki Y, Kataoka K (2009) 3D spheroid culture system on micropatterned substrates for improved differentiation efficiency of multipotent mesenchymal stem cells. Biomaterials 30:2705–2715
Weiss ML, Troyer DL (2006) Stem cells in the umbilical cord. Stem Cell Rev 2:155–162
Weiss ML, Anderson C, Medicetty S, Seshareddy KB, Weiss RJ, VanderWerff I, Troyer D, McIntosh KR (2008) Immune properties of human umbilical cord Wharton’s jelly-derived cells. Stem Cells 26:2865–2874
Zagoura DS, Roubelakis MG, Bitsika V, Trohatou O, Pappa KI, Kapelouzou A, Antsaklis A, Anagnou NP (2012) Therapeutic potential of a distinct population of human amniotic fluid mesenchymal stem cells and their secreted molecules in mice with acute hepatic failure. Gut 61:894–906
Zhang B, Liu R, Shi D, Liu X, Chen Y, Dou X, Zhu X, Lu C, Liang W, Liao L, Zenke M, Zhao RC (2009) Mesenchymal stem cells induce mature dendritic cells into a novel Jagged-2–dependent regulatory dendritic cell population. Blood 113:46–57
Zuk PA, Zhu M, Mizuno H, Huang J, Futrell JW, Katz AJ, Benhaim P, Lorenz HP, Hedrick MH (2001) Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng 7:211–228
Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH (2002) Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell 13:4279–4295
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by grants from the National Key Clinical Project, National Sciences and Technology Major Project of China (2012ZX10002-017) and the Sichuan Provincial Science and Technology Support Project (2012SZ0016).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Table 1
(DOCX 12 kb)
Rights and permissions
About this article
Cite this article
Li, Y., Guo, G., Li, L. et al. Three-dimensional spheroid culture of human umbilical cord mesenchymal stem cells promotes cell yield and stemness maintenance. Cell Tissue Res 360, 297–307 (2015). https://doi.org/10.1007/s00441-014-2055-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-014-2055-x