Abstract
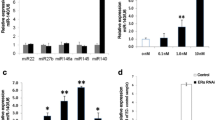
Clinical observations have suggested a relationship between osteoarthritis and a changed sex-hormone metabolism, especially in menopausal women. This study analyzes the effect of 17β-estradiol on expression of matrix metalloproteinases-1, -3, -13 (MMP-1, -3, -13) and tissue inhibitors of metalloproteinases-1, -2 (TIMP-1, -2) in articular chondrocytes. An imbalance of matrix metalloproteinases (MMPs) specialized on degradation of articular cartilage matrix over the respective inhibitors of these enzymes (TIMPs) that leads to matrix destruction was postulated in the pathogenesis of osteoarthritis. Primary human articular chondrocytes from patients of both genders were cultured in alginate beads at 5% O2 to which 10−11M–10−5M 17β-estradiol had been added and analyzed by means of immunohistochemistry, immunocytochemistry and real-time RT-PCR. Since articular chondrocytes in vivo are adapted to a low oxygen tension, culture was performed at 5% O2. Immunohistochemical staining in articular cartilage tissue from patients and immunocytochemical staining in articular chondrocytes cultured in alginate beads was positive for type II collagen, estrogen receptor α, MMP-1, and -13. It was negative for type I collagen, MMP-3, TIMP-1 and -2. Using real-time RT-PCR, it was demonstrated that physiological and supraphysiological doses of 17β-estradiol suppress mRNA levels of MMP-3 and -13 significantly in articular chondrocytes of female patients. A significant suppressing effect was also seen in MMP-1 mRNA after a high dose of 10−5M 17β-estradiol. Furthermore, high doses of this hormone led to tendentially lower TIMP-1 levels whereas the TIMP-2 mRNA level was not influenced. In male patients, only incubations with high doses (10−5M) of 17β-estradiol were followed by a tendency to suppressed MMP-1 and TIMP-1 levels while TIMP-2 mRNA level was decreased significantly. There was no effect on MMP-13 expression of cells from male patients. Taken together, application of 17β-estradiol in physiological doses will improve the imbalance between the amounts of MMPs and TIMPs in articular chondrocytes from female patients. Downregulation of TIMP-2 by 17β-estradiol in male patients would not be articular cartilage protective.






Similar content being viewed by others
References
Appleton CT, Pitelka V, Henry J, Beier F (2007) Global analyses of gene expression in early experimental osteoarthritis. Arthritis Rheum 56:1854–1868
Billinghurst FC, Dahlberg L, Ionescu M, Reiner A, Bourne R, Rorabeck C, Mitchell P, Hambor J, Diekmann O, Tschesche H, Chen J, Wart HV, Poole AR (1997) Enhanced cleavage of by collagenases in osteoarthritic articular cartilage. J Clin Invest 99:1534–1545
Chubinskaya S, Huch K, Schulze M, Otten L, Aydelotte MB, Cole AA (2001) Gene expression by human articular chondrocytes cultured in alginate beads. J Histochem Cytochem 49:1211–1220
Dean DD, Woessner JF (1984) Extracts of human articular cartilage contain an inhibitor of tissue metalloproteinases. Biochem J 218:277–280
Dean DD, Martel-Pelletier J, Pelletier JP, Howell DS, Woessner JF (1989) Evidence for metalloproteinase and metalloproteinase inhibitor imbalance in human osteoarthritic cartilage. J Clin Invest 84:678–685
Dean DD (1991) Proteinase-mediated cartilage degradation in osteoarthritis. Semin Arthritis Rheum 20:2–11
Domm C, Schünke M, Christesen K, Kurz B (2001) Redifferentiation of dediffentiated bovine articular chondrocytes in alginate culture under low oxygen tension. Osteoarthr Cartil 10:13–22
Erb A, Brenner H, Gunther KP, Sturmer T (2000) Hormone replacement therapy and patterns of osteoarthritis: baseline data from the Ulm Osteoarthritis Study. Ann Rheum Dis 59:105–109
Fernandez JC, Martel-Pelletier J, Lascau-Coman V, Moldovan F, Jovanovic D, Raynauld JP, Pelletier JP (1998) Collagenase-1 and collagenase-3 synthesis in normal and early experimental osteoarthritic canine cartilage: an immunohistochemical study. J Rheumatol 25:1585–1594
Fichter M, Körner U, Schömburg J, Jennings L, Cole AA, Mollenhauer J (2006) Collagen degradation products modulate matrix metalloproteinase expression in cultured articular chondrocytes. J Orthop Res 24:63–70
Freemont AJ, Hampson V, Tilman R, Goupille P, Taiwo Y, Hoyland JA (1997) Gene expression of matrix metalloproteinases 1, 3, and 9 by chondrocytes in osteoarthritic human knee articular cartilage is zone and grade specific. Ann Rheum Dis 56:542–549
Gokhale JA, Frenkel SR, Dicesare PE (2004) Estrogen and osteoarthritis. Am J Orthop 33:71–80
Goupille P, Jayson MIV, Valat JP, Freemont AJ (1998) Matrix metalloproteinases: The clue to intervertebral disc degeneration? Spine 23:1612–1626
Greenwald RA (1994) Treatment of destructive arthritic disorders with MMP inhibitors. Potential role of tetracyclines. Ann N Y Acad Sci 732:181–198
Ham KD, Loeser RF, Lindgren BR, Carlson CS (2002) Effects of long-term estrogen replacement therapy on osteoarthritis severity in cynomolgus monkeys. Arthritis Rheum 46:1956–1964
Hanna FS, Wluka AE, Bell RJ, Davis SR, Cicuttini FM (2004) Osteoarthritis and the postmenopausal woman: Epidemiological, magnetic resonance imaging, and radiological findings. Semin Arthritis Rheum 34:631–636
Hart DJ, Doyle DV, Spector TD (1999) Incidence and risk factors for radiographic knee osteoarthritis in middle-aged women: the Chingford Study. Arthritis Rheum 42:17–24
Hui W, Rowan AD, Cawston T (2001) Modulation of the expression of matrix metalloproteinase and tissue inhibitors of metalloproteinases by TGF-beta1 and IFG-1 in primary human articular and bovine nasal chondrocytes stimulated with TNF-alpha. Cytokine 16:31–35
Huppertz B, Kertschanska S, Demir AY, Frank HG, Kaufmann P (1998) Immunohistochemistry of matrix metalloproteinases (MMP), their substrates, and their inhibitors (TIMP) during trophoblast invasion in the human placenta. Cell Tissue Res 291:133–148
Kinney RC, Schwartz Z, Week K, Lotz MK, Boyan BD (2005) Human articular chondrocytes exhibit sexual dimorphism in their responses to 17β-estradiol. Osteoarthr Cartil 13:330–337
Lee YJ, Lee EB, Kwon YE, Lee JJ, Cho WS, Kim HA, Song YW (2003) Effect of estrogen on the expression of matrix metalloproteinase (MMP)-1, MMP-3, and MMP-13 and tissue inhibitor of metalloproteinase-1 in osteoarthritis chondrocytes. Rheumatol Int 23:282–288
Lo YYC, Conquer JA, Grinstein S, Cruz TF (1998) Interleukin-1β induction of c-fos and collagenase expression in articular chondrocytes: Involvement of reactive oxygen species. J Cell Biochem 69:19–29
Ma HL, Blanchet TJ, Peluso D, Hopkins B, Morris EA, Glasson SS (2007) Osteoarthritis severity is sex dependent in a surgical mouse model. Osteoarthr Cartil 15:695–700
Malemud CJ, Hering TM (1992) Regulation of chondrocytes in osteoarthrosis. In: Adolphe M (ed) Biological regulation of chondrocytes. CRC Press, Boca Raton, pp 295–319
Martel-Pelletier JM, McCollum R, Fujimoto N, Obata K, Cloutier JM, Pelletier JP (1994) Excess of metalloproteinases over tissue inhibitor of metalloproteinases may contribute to cartilage degradation in osteoarthritis and rheumatoid arthritis. Lab Invest 70:807–815
Mehraban F, Lark MW, Ahmed FN, Xu F, Moskowitz RW (1998) Increased secretion and activity of matrix metalloproteinase-3 in synovial tissues and chondrocytes from experimental osteoarthritis. Osteoarthr Cartil 6:286–294
Murphy CL, Sambanis A (2001) Effect of oxygen tension on chondrocytes extracellular matrix accumulation. Connect Tissue Res 42:87–96
Oestergaard S, Sondergaard BC, Hoegh-Andersen P, Henriksen K, Qvist P, Christiansen C, Tanko LB, Karsdal MA (2006) Effects of ovariectomy and estrogen therapy on degradation and structural integrity of articular cartilage in rats: implications of the time of initiation. Arthritis Rheum 54:2441–2451
Okada Y, Shinmei M, Tanaka O, Naka K, Kimura A, Nakanishi I, Bayliss MT, Iwata K, Nagase H (1992) Localization of matrix metalloproteinase-3 (stromelysin) in osteoarthritic cartilage and synovium. Lab Invest 66:680–690
Parazzini F (2003) Menopausal status, hormone replacement therapy use and risk of self-reported physician-diagnosed osteoarthritis in women attending menopause clinics in Italy. Maturitas 46:207–212
Pelletier JP, Mineau F, Faure MP, Martel-Pelletier J (1990) Imbalance between the mechanism of activation and inhibition of metalloproteinases in the early lesions of experimental osteoarthritis. Arthritis Rheum 33:1466–1476
Pufe T, Lemke A, Kurz B, Petersen W, Tillmann B, Grodzinsky AJ, Mentlein R (2004) Mechanical overload induces VEGF in cartilage discs via hypoxia-inducible factor. Am J Pathol 164:185–192
Ravn P, Warming L, Christgau S, Christiansen C (2004) The effect on cartilage of different forms of application of postmenopausal estrogen therapy: comparison of oral and transdermal therapy. Bone 35:1216–1221
Reginster JY, Kvasz A, Bruyere O, Henrotin Y (2003) Is there any rationale for prescribing hormone replacement therapy (HRT) to prevent or to treat osteoarthritis? Osteoarthr Cartil 11:87–91
Ries C, Petrides PE (1995) Cytokine regulatioon of matrix metalloproteinase activity and its regulatory dysfunction in disease. Biol Chem Hoppe-Seyler 376:345–355
Song YJ, Wu ZH, Lin SQ, Weng XS, Qiu GX (2003) The effect of estrogen and progestin on the expression of matrix metalloproteinases, tissue inhibitor of metalloproteinase and interleukin-1beta mRNA in synovia of OA rabbit model. Zhonghua Yi Xue Za Zhi 83:498–503
Spector TD, Campion GD (1989) Generalised osteoarthritis: a hormonally mediated disease. Ann Rheum Dis 48:523–527
Spector TD, Perry LA, Jubb RW (1991a) Endogenous sex steroid levels in women with generalised osteoarthritis. Clin Rheum 10:316–319
Spector TD, Hart DJ, Brown P (1991b) Frequency of osteoarthritis in hysterectomized women. J Rheumatol 18:1877–1883
Spector TD, Nandra D, Hart DJ, Doyle DV (1997) Is hormone replacement therapy protective for hand and knee osteoarthritis in women? The Chingford study. Ann Rheum Dis 56:432–434
Takahashi K, Goomer RS, Harwood F, Kubo T, Hirasawa Y, Amiel D (1999) The effects of hyaluronan on matrix metalloproteinase-3 (MMP-3), interleukin-1β (IL-1β), and tissue inhibitor of metalloproteinase-1 (TIMP-1) gene expression during the development of osteoarthritis. Osteoarthr Cartil 7:182–190
Tankó LB, Sondergaard BC, Oestergaard S, Karsdal MA, Christiansen C (2008) An update review of cellular mechanisms conferring the indirect and direct effects of estrogen on articular cartilage. Climacteric 11:4–16
Tetlow LC, Adlam DJ, Wooley DE (2001) Matrix metalloproteinase and proinflammatory cytokine production by chondrocytes of human osteoarthritic cartilage: associations with degenerative changes. Arthritis Rheum 44:585–594
Thole HH, Jakob F (1993) Characterization of five monoclonal antibodies raised against domain E of the porcine estradiol receptor. Exp Clin Endocrinol 101:112–118
von Muhlen D, Morton D, von Muhlen CA, Barrett-Connor E (2002) Postmenopausal estrogen and increased risk of clinical osteoarthritis at the hip, hand, and knee in older women. J Womens Health Gend Based Med 11:511–518
Walter H, Kawashima A, Nebelung W, Neumann W, Roessner A (1998) Immunohistochemical analysis of several proteolytic enzymes as parameters of cartilage degradation. Pathol Res Pract 194:73–81
Wluka AE, Cicuttini FM, Spector TD (2000) Menopause, oestrogens and arthritis. Maturitas 35:183–199
Wluka AE, Davis SR, Bailey M, Stuckey SL, Cicuttini FM (2001) Users of oestrogen replacement therapy have more knee cartilage than non-users. Ann Rheum Dis 60:332–336
Woessner JF, Gunja-Smith Z (1991) Role of metalloproteinases in human osteoarthritis. J Rheumatol Suppl 27:99–101
Wu JJ, Lark MW, Chun LE, Eyre DR (1991) Sites of stromelysin cleavage in collagen type II, IX, X, and XI of cartilage. J Biol Chem 266:5625–5628
Wu W, Billinghurst RC, Pidoux I, Antomiou J, Zukor D, Tanzer M, Poole AR (2002) Sites of collagenase cleavage and denaturation of in aging and osteoarthritis articular cartilage and their relationship to the distribution of matrix metalloproteinase 1 and matrix metalloproteinase 13. Arthritis Rheum 46:2087–2094
Yasuda T, Tchetina E, Ohsawa K, Roughley PJ, Wu W, Mousa A, Ionescu M, Pidoux I, Poole AR (2006) Peptides of type II collagen can induce the cleavage of type I collagen and aggrecan in articular cartilage. Matrix Biol 25:419–429
Zafarullah M, Su S, Martel-Pelletier J, DiBattista JA, Costello BG, Stetler-Stevenson WG, Pelletier JP (1996) Tissue inhibitor of metalloproteinase-2 (TIMP-2) mRNA is constitutively expressed in bovine, human normal, and osteoarthritic articular chondrocytes. J Cell Biochem 60:211–217
Acknowledgements
This study was supported by a research grant, project 989039, from the “Musculoskeletal Research Center“ of the Medical Faculty of the Christian-Albrechts-University of Kiel to Horst Claassen. We further thank the Deutsche Forschungsgemeinschaft (DFG) for support (PU 2/4/3-2; 4-2; 5-2). Especially, we would like to thank Ms. Inka Geurink, Ms. Claudia Kremling and Ms. Sabine Lorenzen (Institute of Anatomy Kiel) for their excellent assistance during the experiments. Furthermore, we wish to thank Wolfgang Graulich (Institute of Anatomy and Cell Biology Aachen) for final grafik art work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lars Ove Brandenburg and Thomas Pufe both contributed equally to the present work
Rights and permissions
About this article
Cite this article
Claassen, H., Steffen, R., Hassenpflug, J. et al. 17β-estradiol reduces expression of MMP-1, -3, and -13 in human primary articular chondrocytes from female patients cultured in a three dimensional alginate system. Cell Tissue Res 342, 283–293 (2010). https://doi.org/10.1007/s00441-010-1062-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-010-1062-9