Abstract
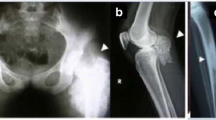
Hyperphosphatemic Familial Tumoral Calcinosis (HFTC; MIM211900) is a rare autosomal recessive disorder characterized by the progressive deposition of calcified masses in cutaneous and subcutaneous tissues, associated with elevated circulating levels of phosphate. The disease was initially found to result from mutations in GALNT3 encoding a glycosyltransferase. However, more recently, the S71G missense mutation in FGF23, encoding a potent phosphaturic protein, was identified in two families. In the present report, we describe a second mutation in FGF23 underlying a severe case displaying calcifications of cutaneous and numerous extracutaneous tissues. The mutation (M96T) was found to affect a highly conserved methionine residue at position 96 of the protein. These observations illustrate the extent of genetic and phenotypic heterogeneity in HFTC.


Similar content being viewed by others
References
ADHR consortium (2000) Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet 26:345–348
Benet-Pagès A, Orlik P, Strom TM, Lorenz-Depiereux B (2005) An FGF-23 missense mutation causes familial tumoral calcinosis with hyperphosphatemia. Hum Mol Genet 14:385–390
Frishberg Y, Topaz O, Bergman R et al (2005) Identification of a recurrent mutation in GALNT3 demonstrates that hyperostosis-hyperphosphatemia syndrome and familial tumoral calcinosis are allelic disorders. J Mol Med 83:33–38
Giard A (1898) Sur la calcification tibernale. CR Soc Biol 10:1015–1021
Ichikawa S, Lyles KW, Econs MJ (2005) A novel GALNT3 mutation in a pseudo-autosomal dominant form of tumoral calcinosis: evidence that the disorder is autosomal recessive. J Clin Endocrinol Metab 90:2420–2423
Jan de Beur SM, Levine MA (2002) Molecular pathogenesis of hypophosphatemic rickets. J Clin Endocrinol Metab 87:2467–2473
Larsson T, Yu X, Davis SI, Draman MS, Mooney SD, Cullen MJ, White KE (2005) A novel recessive mutation in Fibroblast growth factor-23 (FGF23) causes familial tumoral calcinosis. J Clin Endocrinol Metab. 90:2424–2427
McClatchie S, Bremner AD (1969) Tumoural calcinosis–an unrecognized disease. Brit Med J 1:153–155
Metzker A, Eisenstein B, Oren J, Samuel R (1988) Tumoral calcinosis revisited–common and uncommon features. Report of ten cases and review. Eur J Pediatr 147:128–132
Neu CM, Manz F, Rauch F et al (2001) Bone densities and bone size at the distal radius in healthy children and adolescents: a study using peripheral quantitative CT. Bone 28:227–232
Polykandriotis EP, Beutel FK, Horch RE, Grunert J (2004) A case of familial tumoral calcinosis in a neonate and review of the literature. Arch Orthop Trauma Surg 124:563–567
Prince MJ, Schaeffer PC, Goldsmith RS, Chausmer AB (1982) Hyperphosphatemic tumoral calcinosis: association with elevation of serum 1,25-dihydroxycholecalciferol concentrations. Ann Intern Med 96:586–591
Schiavi SC, Kumar R (2004) The phosphatonin pathway: new insights in phosphate homeostasis. Kidney Int 65:1–14
Shimada T, Kakitani M, Yamazaki Y et al (2004) Targeted ablation of FGF23 demonstrates an essential physiological role of FGF23 in phosphate and vitamin D metabolism. J Clin Invest 113:561–568
Smack D, Norton SA, Fitzpatrick JE (1996) Proposal for a pathogenesis-based classification of tumoral calcinosis. Int J Dermatol 35:265–271
Ten Hagen KG, Fritz TA, Tabak LA (2003) All in the family: the UDP-GalNAc:polypeptide N-acetylgalactosaminyltransferases. Glycobiology 13:1R–16R
Topaz O, Bergman R, Mandel U et al (2005) Absence of intraepidermal glycosyltransferase ppGalNAc-T3 expression in familial tumoral calcinosis. Am J Dermatopathol, in press
Topaz O, Shurman D, Bergman R et al (2004) Mutations in GALNT3, encoding a protein involved in O-linked glycosylation, cause familial tumoral calcinosis. Nat Genet 36:579–581
Touart DM, Sau P (1998) Cutaneous deposition diseases. Part II. J Am Acad Dermatol 39:527–544
Walton, Bijvoet (1975) Nomogram for derivation of renal threshold phosphate concentration. Lancet 2(7929):309–310
Yamashita T, Konishi M, Miyake A et al (2002) Fibroblast growth factor (FGF)-23 inhibits renal phosphate reabsorption by activation of the mitogen-activated protein kinase pathway. J Biol Chem 277:28265–28270
Ackowledgements
We acknowledge the family for having participated in the present study. We are grateful to V. Friedman for DNA sequencing services. This study was supported in part by grants provided by The Israeli Ministry of Health-Chief Scientist Office and by the General Trustee Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chefetz, I., Heller, R., Galli-Tsinopoulou, A. et al. A novel homozygous missense mutation in FGF23 causes Familial Tumoral Calcinosis associated with disseminated visceral calcification. Hum Genet 118, 261–266 (2005). https://doi.org/10.1007/s00439-005-0026-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-005-0026-8