Abstract
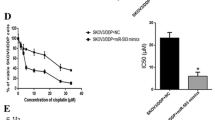
Cisplatin is one of the most popular chemotherapeutic drugs in treating ovarian cancer. Resistance to cisplatin is a common clinical challenge that needs to be solved to increase its anti-tumor effects. The relation of miR-514 expression with prognosis in ovarian cancer patients was analyzed based on GSE73584 datasets. The regulation of miR-514 on proliferation and cisplatin chemosensitivity of ovarian cells was examined by MTT assay, colony-formation assay and soft-agar colony-formation assay. Dual luciferase assay was performed to detect the direct interaction of miR-514 with its downstream targets. Immunobloting and qRT-PCR were performed for target gene expression analysis. Low expression of miR-514 was related to poor prognosis in ovarian cancer patients. MiR-514 repressed proliferation and decreased cisplatin chemosensitivity in ovarian cancer cells by targeting ATP binding cassette subfamily. MiR-514 is of clinically significance in ovarian cancer by attenuating proliferation of ovarian cancer cells and decreasing chemoresistance of cisplatin by targeting ATP binding cassette subfamily.





Similar content being viewed by others
References
Bao L, Wu J, Dodson M, Rojo de la Vega EM, Ning Y, Zhang Z, Yao M, Zhang DD, Xu C, Yi X (2017) ABCF2, an Nrf2 target gene, contributes to cisplatin resistance in ovarian cancer cells. Mol Carcinog 56:1543–1553. https://doi.org/10.1002/mc.22615
Buechler C, Eisinger K, Krautbauer S (2013) Diagnostic and prognostic potential of the macrophage specific receptor CD163 in inflammatory diseases. Inflamm Allergy Drug Targets 12:391–402
Dean M (2009) ABC transporters, drug resistance, and cancer stem cells. J Mammary Gland Biol Neoplasia 14:3–9. https://doi.org/10.1007/s10911-009-9109-9
Donninger H, Bonome T, Radonovich M, Pise-Masison CA, Brady J, Shih JH, Barrett JC, Birrer MJ (2004) Whole genome expression profiling of advance stage papillary serous ovarian cancer reveals activated pathways. Oncogene 23:8065–8077. https://doi.org/10.1038/sj.onc.1207959
Franken NA, Rodermond HM, Stap J, Haveman J, van Bree C (2006) Clonogenic assay of cells in vitro. Nat Protoc 1:2315–2319. https://doi.org/10.1038/nprot.2006.339
Friedlander ML (1998) Prognostic factors in ovarian cancer. Semin Oncol 25:305–314
Glavinas H, Krajcsi P, Cserepes J, Sarkadi B (2004) The role of ABC transporters in drug resistance, metabolism and toxicity. Curr Drug Deliv 1:27–42
Horibata S, Vo TV, Subramanian V, Thompson PR, Coonrod SA (2015) Utilization of the soft agar colony formation assay to identify inhibitors of tumorigenicity in breast cancer cells. J Vis Exp. https://doi.org/10.3791/52727
Jemal A, Murray T, Samuels A, Ghafoor A, Ward E, Thun MJ (2003) Cancer statistics, 2003. CA Cancer J Clin 53:5–26
Jung YS, Kim HJ, Seo SK, Choi YS, Nam EJ, Kim S, Kim SW, Han HD, Kim JW, Kim YT (2016) Anti-proliferative and apoptotic activities of mullerian inhibiting substance combined with calcitriol in ovarian cancer cell lines. Yonsei Med J 57:33–40. https://doi.org/10.3349/ymj.2016.57.1.33
Kanaji N, Kamitori K, Hossain A, Noguchi C, Katagi A, Kadowaki N, Tokuda M (2018) Additive antitumour effect of dallose in combination with cisplatin in non-small cell lung cancer cells. Oncol Rep. https://doi.org/10.3892/or.2018.6192
Lee S, Choi EJ, Jin C, Kim DH (2005) Activation of PI3K/Akt pathway by PTEN reduction and PIK3CA mRNA amplification contributes to cisplatin resistance in an ovarian cancer cell line. Gynecol Oncol 97:26–34. https://doi.org/10.1016/j.ygyno.2004.11.051
Lin C, Xie L, Lu Y, Hu Z, Chang J (2018) miR-133b reverses cisplatin resistance by targeting GSTP1 in cisplatin-resistant lung cancer cells. Int J Mol Med. https://doi.org/10.3892/ijmm.2018.3382
Liu J, Bi J, Li Z, Li Z, Liu X, Kong C (2018) miR214 reduces cisplatin resistance by targeting netrin1 in bladder cancer cells. Int J Mol Med 41:1765–1773. https://doi.org/10.3892/ijmm.2018.3374
Lou H, Dean M (2007) Targeted therapy for cancer stem cells: the patched pathway and ABC transporters. Oncogene 26:1357–1360. https://doi.org/10.1038/sj.onc.1210200
Proceedings of the Conference on Antilymphocyte Serum, Brook Lodge, Augusta, Michigan, May 2–3, 1969 (1970) Fed Proc 29:93–229
Rogers LC, Davis RR, Said N, Hollis T, Daniel LW (2018) Blocking LPA-dependent signaling increases ovarian cancer cell death in response to chemotherapy. Redox Biol 15:380–386. https://doi.org/10.1016/j.redox.2018.01.002
Saed GM, Fletcher NM, Diamond MP, Morris RT, Gomez-Lopez N, Memaj I (2018) Novel expression of CD11b in epithelial ovarian cancer: potential therapeutic target. Gynecol Oncol. https://doi.org/10.1016/j.ygyno.2017.12.018
Staff P (2018) Correction: risk of thromboembolism in cisplatin versus carboplatin-treated patients with lung cancer. PLoS One 13:e0191454. https://doi.org/10.1371/journal.pone.0191454
Su Z, Zhang M, Xu M, Li X, Tan J, Xu Y, Pan X, Chen N, Chen X, Zhou Q (2018) MicroRNA181c inhibits prostate cancer cell growth and invasion by targeting multiple ERK signaling pathway components. Prostate. https://doi.org/10.1002/pros.23478
Todorovic L, Stanojevic B, Mandusic V, Petrovic N, Zivaljevic V, Paunovic I, Diklic A, Saenko V, Yamashita S (2018) Expression of VHL tumor suppressor mRNA and miR-92a in papillary thyroid carcinoma and their correlation with clinical and pathological parameters. Med Oncol 35:17. https://doi.org/10.1007/s12032-017-1066-3
Wang CW, Lee BH, Tai CJ (2017a) The inhibition of cordycepin on cancer stemness in TGF-beta induced chemo-resistant ovarian cancer cell. Oncotarget 8:111912–111921. https://doi.org/10.18632/oncotarget.22951
Wang Z, Wang W, Huang K, Wang Y, Li J, Yang X (2017b) MicroRNA-34a inhibits cells proliferation and invasion by downregulating Notch1 in endometrial cancer. Oncotarget 8:111258–111270. https://doi.org/10.18632/oncotarget.22770
Wotschofsky Z, Busch J, Jung M, Kempkensteffen C, Weikert S, Schaser KD, Melcher I, Kilic E, Miller K, Kristiansen G, Erbersdobler A, Jung K (2013) Diagnostic and prognostic potential of differentially expressed miRNAs between metastatic and non-metastatic renal cell carcinoma at the time of nephrectomy. Clin Chim Acta 416:5–10. https://doi.org/10.1016/j.cca.2012.11.010
Wu D, Lu P, Mi X, Miao J (2018) Downregulation of miR-503 contributes to the development of drug resistance in ovarian cancer by targeting PI3K p85. Arch Gynecol Obstet. https://doi.org/10.1007/s00404-018-4649-0
Xu Y, Miao C, Jin C, Qiu C, Li Y, Sun X, Gao M, Lu N, Kong B (2018) SUSD2 promotes cancer metastasis and confers cisplatin resistance in high grade serous ovarian cancer. Exp Cell Res. https://doi.org/10.1016/j.yexcr.2017.12.029
Yang X, Fraser M, Moll UM, Basak A, Tsang BK (2006) Akt-mediated cisplatin resistance in ovarian cancer: modulation of p53 action on caspase-dependent mitochondrial death pathway. Cancer Res 66:3126–3136. https://doi.org/10.1158/0008-5472.CAN-05-0425
Zhang X, Shao R, Gao W, Sun G, Liu Y, Fa X (2018) Inhibition of miR-361-5p suppressed pulmonary artery smooth muscle cell survival and migration by targeting ABCA1 and inhibiting the JAK2/STAT3 pathway. Exp Cell Res. https://doi.org/10.1016/j.yexcr.2018.01.015
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
Additional information
Communicated by S. Hohmann.
Rights and permissions
About this article
Cite this article
Xiao, S., Zhang, M., Liu, C. et al. MiR-514 attenuates proliferation and increases chemoresistance by targeting ATP binding cassette subfamily in ovarian cancer. Mol Genet Genomics 293, 1159–1167 (2018). https://doi.org/10.1007/s00438-018-1447-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-018-1447-0