Abstract
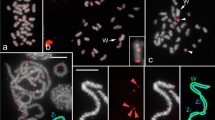
Squamate reptiles show a striking diversity in modes of sex determination, including both genetic (XY or ZW) and temperature-dependent sex determination systems. The genomes of only a handful of species have been sequenced, analyzed and assembled including the genome of Anolis carolinensis. Despite a high genome coverage, only macrochromosomes of A. carolinensis were assembled whereas the content of most microchromosomes remained unclear. Most of the Anolis species have homomorphic XY sex chromosome system. However, some species have large heteromorphic XY chromosomes (e.g., A. sagrei) and even multiple sex chromosomes systems (e.g. A. pogus), that were shown to be derived from fusions of the ancestral XY with microautosomes. We applied next generation sequencing of flow sorting-derived chromosome-specific DNA pools to characterize the content and composition of microchromosomes in A. carolinensis and A. sagrei. Comparative analysis of sequenced chromosome-specific DNA pools revealed that the A. sagrei XY sex chromosomes contain regions homologous to several microautosomes of A. carolinensis. We suggest that the sex chromosomes of A. sagrei are derived by fusions of the ancestral sex chromosome with three microautosomes and subsequent loss of some genetic content on the Y chromosome.


Similar content being viewed by others
References
Alföldi J, Di Palma F, Grabherr M et al (2011) The genome of the green anole lizard and a comparative analysis with birds and mammals. Nature 477:587–591
Bertolotto CEV, Rodrigues MT, Yonenaga-Yassuda Y (2001) Banding patterns, multiple sex chromosome system and localization of telomeric (TTAGGG)n sequences by FISH on two species of Polychrus (Squamata, Polychrotidae). Caryologia 54:217–226
Brinkrolf K, Rupp O, Laux H et al (2013) Chinese hamster genome sequenced from sorted chromosomes. Nat Biotechnol 31:694–695
Crawford PA, Dorn C, Sadovsky Y, Milbrandt J (1998) Nuclear receptor DAX-1 recruits nuclear receptor corepressor N-CoR to steroidogenic factor 1. Mol Cell Biol 18:2949–2956
De Smet W (1981) Description of the orcein stained karyotypes of 27 lizard species (Lacertilia Reptilia) belonging to the families Iguanidae, Agamidae, Chamaeleontidae and Gekkonidae (Ascalabota). Acta Zool Pathol Antwerp 76:35–72
Eggers S, Sinclair A (2012) Mammalian sex determination—insights from humans and mice. Chromosome Res 20:215–238
Ellegren H (2011) Sex-chromosome evolution: recent progress and the influence of male and female heterogamety. Nat Rev Genet 12:157–166
Etheridge R (1960) The relationships of the anoles (Reptilia: Sauria: Iguanidae): an interpretation based on skeletal morphology. PhD Dissertation, University of Michigan
Ezaz T, Graves JAM (2012) Foreword: sex and sex chromosomes—new clues from nonmodel species. Chromosome Res 20:1–5
Ezaz T, Sarre SD, O’Meally D et al (2009) Sex chromosome evolution in lizards: independent origins and rapid transitions. Cytogenet Genome Res 127:249–260
Gamble T, Geneva AJ, Glor RE, Zarkower D (2014) Anolis sex chromosomes are derived from a single ancestral pair. Evolution 68:1027–1041
Giovannotti M, Trifonov V, Paoletti A et al (2016) New insights into sex chromosome evolution in anole lizards (Reptilia, Dactyloidae). Chromosoma. doi:10.1007/s00412-016-0585-6
Gorman GC (1973) The chromosomes of the reptilia, a cytotaxonomic interpretation. Cytotaxon Vertebr Evol:349–424
Gorman G, Atkins L (1967) The zoogeography of lesser Antillean Anolis lizards—an analysis based upon chromosomes and lactic dehydrogenases. Bull Mus Comp Zool 138:53–80
Graves JAM (2006) Sex chromosome specialization and degeneration in mammals. Cell 124:901–914
Graves JAM (2013) How to evolve new vertebrate sex determining genes. Dev Dyn 242:354–359
Graves JAM, Peichel CL (2010) Are homologies in vertebrate sex determination due to shared ancestry or to limited options? Genome Biol 11:205
Gruetzner F, Ashley T, Rowell DM, Graves JAM (2005) How did the platypus get its sex chromosome chain? A comparison of meiotic multiples and sex chromosomes in plants and animals. Chromosoma 115:75–88
Hedges SB, Poling LL (1999) A molecular phylogeny of reptiles. Science 283:998–1001
Janzen FJ, Phillips PC (2006) Exploring the evolution of environmental sex determination, especially in reptiles. J Evol Biol 19:1775–1784
Kawagoshi T, Uno Y, Matsubara K et al (2009) The ZW micro-sex chromosomes of the chinese soft-shelled turtle (Pelodiscus sinensis, Trionychidae, Testudines) have the same origin as chicken chromosome 15. Cytogenet Genome Res 125:125–131
Kikuchi K, Hamaguchi S (2013) Novel sex-determining genes in fish and sex chromosome evolution. Dev Dyn 242:339–353
Kitano J, Peichel CL (2011) Turnover of sex chromosomes and speciation in fishes. Environ Biol Fishes 94:549–558
Kitano J, Ross JA, Mori S et al (2009) A role for a neo-sex chromosome in stickleback speciation. Nature 461:1079–1083
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359
Losos JB (2009) Lizards in an evolutionary tree: ecology and adaptive radiation of anoles. University of California Press, California
Makunin A, Kichigin I, Larkin D et al (2016) Contrasting origin of B chromosomes in two cervids (Siberian roe deer and grey brocket deer) unravelled by chromosome-specific DNA sequencing. BMC Genomics (in press)
Mank JE, Vicoso B, Berlin S, Charlesworth B (2010) Effective population size and the faster-X effect: empirical results and their interpretation. Evolution 64:663–674
Martin M (2011) Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J 17:10–12
Matsubara K, Tarui H, Toriba M (2006) Evidence for different origin of sex chromosomes in snakes, birds, and mammals and step-wise differentiation of snake sex chromosomes. Proc Natl Acad Sci 103:18190–18195. doi:10.1073/pnas.0605274103
McKenna A, Hanna M, Banks E et al (2010) The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 20:1297–1303
Nicholson K, Crother B, Guyer C, Savage J (2012) It is time for a new classification of anoles (Squamata: Dactyloidae). Zootaxa 3477:1–108
O’Meally D, Ezaz T, Georges A et al (2012) Are some chromosomes particularly good at sex? Insights from amniotes. Chromosome Res 20:7–19
Poe S (2013) 1986 Redux: new genera of anoles (Squamata: Dactyloidae) are unwarranted. Zootaxa 3626(2):295–299
Presgraves DC (2008) Sex chromosomes and speciation in Drosophila. Trends Genet 24:336–343
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26:841–842
Quinn AE, Georges A, Sarre SD et al (2007) Temperature sex reversal implies sex gene dosage in a reptile. Science 316:411
Quinn AE, Sarre SD, Ezaz T et al (2011) Evolutionary transitions between mechanisms of sex determination in vertebrates. Biol Lett 7:443–448
Qvarnström A, Bailey RI (2008) Speciation through evolution of sex-linked genes. Heredity 102:4–15
Rovatsos M, Altmanová M, Pokorná M, Kratochvíl L (2014a) Conserved sex chromosomes across adaptively radiated anolis lizards. Evolution 68:2079–2085
Rovatsos M, Pokorná M, Altmanová M, Kratochvíl L (2014b) Cretaceous park of sex determination: sex chromosomes are conserved across iguanas. Biol Lett 10:20131093
Sarre SD, Ezaz T, Georges A (2011) Transitions between sex-determining systems in reptiles and amphibians. Annu Rev Genom Hum Genet 12:391–406
Smith CA, Roeszler KN, Ohnesorg T et al (2009) The avian Z-linked gene DMRT1 is required for male sex determination in the chicken. Nature 461:267–271
Solari AJ, Rahn MI (2005) Fine structure and meiotic behaviour of the male multiple sex chromosomes in the genus Alouatta. Cytogenet Genome Res 108:262–267
Swain A, Lovell-Badge R (1999) Mammalian sex determination: a molecular drama. Genes Dev 13:755–767
Telenius H, Carter NP, Bebb CE et al (1992) Degenerate oligonucleotide-primed PCR: general amplification of target DNA by a single degenerate primer. Genomics 13:718–725
Veyrunes F, Waters PD, Miethke P et al (2008) Bird-like sex chromosomes of platypus imply recent origin of mammal sex chromosomes. Genome Res 18:965–973
Vicoso B, Bachtrog D (2013) Reversal of an ancient sex chromosome to an autosome in Drosophila. Nature 499:332–335
Webster TP, Hall WP, Williams EE (1972) Fission in the evolution of a lizard karyotype. Science 177:611–613
Yang F, Trifonov V, Ng BL et al (2009) generation of paint probes by flow-sorted and microdissected chromosomes. In: Liehr T (ed) Fluorescence in situ hybridization (FISH)—application guide. Springer, Berlin, pp 35–52
Zatloukalová P, Hřibová E, Kubaláková M et al (2011) Integration of genetic and physical maps of the chickpea (Cicer arietinum L.) genome using flow-sorted chromosomes. Chromosome Res 19:729–739
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The work was supported by the Budget Projects 0310-2014-0003, 0310-2014-0008, 0310-2014-0009 provided to Vladimir A. Trifonov, partly by RSF Grant No. 16-14-10009 provided to A. Graphodatsky, and by the funds provided by Ministry of Education, University and Research (Italy) (“Ricerche di citogenetica molecolare sui sistemi di determinazione del sesso nei rettili squamati, sottordine Sauria”; Grant number: PRIN2009/20093HYH97) to Vincenzo Caputo Barucchi.
Conflict of interest
The authors declare they have no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by S. Xu.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kichigin, I.G., Giovannotti, M., Makunin, A.I. et al. Evolutionary dynamics of Anolis sex chromosomes revealed by sequencing of flow sorting-derived microchromosome-specific DNA. Mol Genet Genomics 291, 1955–1966 (2016). https://doi.org/10.1007/s00438-016-1230-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-016-1230-z