Abstract
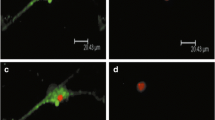
Schistosomiasis is still prevalent and seriously endangering the health of people and livestock in many countries. There have been great efforts to develop vaccines against schistosomiasis for prolonged protection in epidemic areas. Molecules from lung-stage schistosomula have been regarded as potential vaccine candidates against schistosomiasis. Our previous work has shown that cathepsin L3 from Schistosoma japonicum (SjCL3) is expressed in lung-stage schistosomula, but its role is not well known. In the present study, we characterized SjCL3 and detected its effect as a possible vaccine in vivo and in vitro. From the results of quantitative PCR (qPCR) and western blot, SjCL3 was present throughout the lifecycle of the worm, and its relative expressed level was higher in the liver eggs and adult worms than other stages. Additionally, immunofluorescence assay showed that SjCL3 was mainly concentrated in the eggshell, alimentary canal, and musculature of worms. Compared with the adjuvant group, the immunization of SjCL3 in mice resulted in a 28.9% decrease in worm burden and a 29.2% reduction in egg number in the host liver. In antibody-dependent cell-mediated cytotoxicity (ADCC) insecticidal experiments in vitro, the existence of SjCL3 could in part suppress adherence between macrophages and worm. The above results indicated that the immunization of SjCL3 could induce limited immune protection against S. japonicum infection in mice, and this protease played a role in breaking the process of ADCC, which was beneficial to the survival of worms.






Similar content being viewed by others
References
Basch PF (1981) Cultivation of Schistosoma mansoni in vitro. I. Establishment of cultures from cercariae and development until pairing. J Parasitol 67(2):179–185
Bogitsh BJ (1978) Schistosoma mansoni: uptake of exogenous hemeproteins by schistosomules grown in vitro. Exp Parasitol 45(2):247–254
Bozzola JJ (2014) Conventional specimen preparation techniques for scanning electron microscopy of biological specimens. Methods Mol Biol (Clifton, NJ) 1117:133–150
Brady CP, Brindley PJ, Dowd AJ, Dalton JP (2000) Schistosoma mansoni: differential expression of cathepsins L1 and L2 suggests discrete biological functions for each enzyme. Exp Parasitol 94(2):75–83
Brady CP, Dowd AJ, Brindley PJ, Ryan T, Day SR, Dalton JP (1999) Recombinant expression and localization of Schistosoma mansoni cathepsin L1 support its role in the degradation of host hemoglobin. Infect Immun 67(1):368–374
Caffrey CR, Rheinberg CE, Mone H, Jourdane J, Li YL, Ruppel A (1997) Schistosoma japonicum, S. mansoni, S. haematobium, S. intercalatum, and S. rodhaini: cysteine-class cathepsin activities in the vomitus of adult worms. Parasitol Res 83(1):37–41
Carmona C, Dowd AJ, Smith AM, Dalton JP (1993) Cathepsin L proteinase secreted by Fasciola hepatica in vitro prevents antibody-mediated eosinophil attachment to newly excysted juveniles. Mol Biochem Parasitol 62(1):9–17
Chung PR, Asch HL, Bruce JI (1982) Antibody-dependent murine macrophage-mediated damage to Schistosoma mansoni schistosomula in vitro. Cell Immunol 74(2):243–259
Dean DA, Mangold BL (1992) Evidence that both normal and immune elimination of Schistosoma mansoni take place at the lung stage of migration prior to parasite death. Am J Trop Med Hyg 47(2):238–248
Dewalick S, Bexkens ML, van Balkom BW, Wu YP, Smit CH, Hokke CH, de Groot PG, Heck AJ, Tielens AG, van Hellemond JJ (2011) The proteome of the insoluble Schistosoma mansoni eggshell skeleton. Int J Parasitol 41(5):523–532
deWalick S, Tielens AG, van Hellemond JJ (2012) Schistosoma mansoni: the egg, biosynthesis of the shell and interaction with the host. Exp Parasitol 132(1):7–13
Dvorak J, Mashiyama ST, Sajid M, Braschi S, Delcroix M, Schneider EL, McKerrow WH, Bahgat M, Hansell E, Babbitt PC, Craik CS, McKerrow JH, Caffrey CR (2009) SmCL3, a gastrodermal cysteine protease of the human blood fluke Schistosoma mansoni. PLoS Negl Trop Dis 3(6):e449
El Ridi R, Tallima H (2009) Schistosoma mansoni ex vivo lung-stage larvae excretory-secretory antigens as vaccine candidates against schistosomiasis. Vaccine 27(5):666–673
Eraky MA, Aly NS, Selem RF, El-Kholy AA, Rashed GA (2016) In vitro schistosomicidal activity of phytol and tegumental alterations induced in juvenile and adult stages of Schistosoma haematobium. Korean J Parasitol 54(4):477–484
Gao Y, Zhou X, Wang H, Liu R, Ye Q, Zhao Q, Ming Z, Dong H (2017) Immunization with recombinant schistosome adenylate kinase 1 partially protects mice against Schistosoma japonicum infection. Parasitol Res 116(6):1665–1674
Hong Y, Huang L, Yang J, Cao X, Han Q, Zhang M, Han Y, Fu Z, Zhu C, Lu K, Li X, Lin J (2015) Cloning, expression and enzymatic characterization of 3-phosphoglycerate kinase from Schistosoma japonicum. Exp Parasitol 159:37–45
Kassim OO, Dean DA, Mangold BL, Von Lichtenberg F (1992) Combined microautoradiographic and histopathologic analysis of the fate of challenge Schistosoma mansoni schistosomula in mice immunized with irradiated cercariae. Am J Trop Med Hyg 47(2):231–237
Kominami E, Tsukahara T, Hara K, Katunuma N (1988) Biosyntheses and processing of lysosomal cysteine proteinases in rat macrophages. FEBS Lett 231(1):225–228
Lawrence JD (1973) The ingestion of red blood cells by Schistosoma mansoni. J Parasitol 59(1):60–63
Liu R, Zhao QP, Ye Q, Xiong T, Tang CL, Dong HF, Jiang MS (2013) Cloning and characterization of a bone morphogenetic protein homologue of Schistosoma japonicum. Exp Parasitol 135(1):64–71
Merrifield M, Hotez PJ, Beaumier CM, Gillespie P, Strych U, Hayward T, Bottazzi ME (2016) Advancing a vaccine to prevent human schistosomiasis. Vaccine 34(26):2988–2991
Moloney NA, Doenhoff MJ, Webbe G, Hinchcliffe P (1982) Studies on the host-parasite relationship of Schistosoma japonicum in normal and immunosuppressed mice. Parasite Immunol 4(6):431–440
Ou Y, Zeng N, Feng Z, Xu P (2010) Determination of papain’s enzyme activity in meat tenderizer by UV spectrophotometry. China Food Additives 2(02):214–216 [in Chinese]
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9):e45
Smyth MJ, Cretney E, Kelly JM, Westwood JA, Street SE, Yagita H, Takeda K, van Dommelen SL, Degli-Esposti MA, Hayakawa Y (2005) Activation of NK cell cytotoxicity. Mol Immunol 42(4):501–510
Su J, Zhang N, Ho PC (2004) Determination of tributyrin and its metabolite butyrate in Wistar rat plasma samples by gas chromatography/mass spectrometry. Rapid communications in mass spectrometry : RCM 18(19):2217–2222
Tallima H, Dvorak J, Kareem S, Abou El Dahab M, Abdel Aziz N, Dalton JP, El Ridi R (2017) Protective immune responses against Schistosoma mansoni infection by immunization with functionally active gut-derived cysteine peptidases alone and in combination with glyceraldehyde 3-phosphate dehydrogenase. PLoS Negl Trop Dis 11(3):e0005443
Tang CLSZ, Cheng LF, Jiang MS, Liu XH (2016) The function of glyceraldehyde phosphate dehydrogenase in mice infected with Schistosoma japonicum. J Pathogen Biol 11(7):638–642 [in Chinese]
Torben W, Ahmad G, Zhang W, Nash S, Le L, Karmakar S, Siddiqui AA (2012) Role of antibody dependent cell mediated cytotoxicity (ADCC) in Sm-p80-mediated protection against Schistosoma mansoni. Vaccine 30(48):6753–6758
van Erp EA, Luytjes W, Ferwerda G, van Kasteren PB (2019) Fc-mediated antibody effector functions during respiratory syncytial virus infection and disease. Front Immunol 10:548
Wesolowska A, Basalaj K, Norbury LJ, Sielicka A, Wedrychowicz H, Zawistowska-Deniziak A (2018) Vaccination against Fasciola hepatica using cathepsin L3 and B3 proteases delivered alone or in combination. Vet Parasitol 250:15–21
WHO (2019) schistosomiasis. https://www.who.int/news-room/fact-sheets/detail/schistosomiasis?tdsourcetag=s_pcqq_aiomsg
Yang DHLY, Xiao R, Li QW (2012) The structure and function of cathepsin L. Chin J Biochem Mol Biol 28(12):1093–1099 [in Chinese]
Ye Q, Zhu JY, Ming ZP, Zhao QP, Grevelding CG, Liu R, Zhong QP, Jiang MS, Dong HF (2012) Studies on the establishment of a co-culture system of lung stage Schistosoma japonicum with host cells. Parasitol Res 111(2):735–748
Zhou S, Ling J, Jiang M (2001) Schistosoma. Science Press, Bei Jing [in Chinese]
Funding
This study was funded by the National Natural Science Foundation of China (No. 81273010) and the Scientific Research Subject of the Health and Family Planning Commission of Hubei Province (No. XF2012–18).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict interest.
Ethical approval
All animal experimentations followed the guidelines of the Association for Assessment and Accreditation of Laboratory Animal Care International and were approved by the Centre for Animal Experiment of Wuhan University (approval no. 2019103).
Additional information
Section Editor: Ramaswamy Kalyanasundaram
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Huang, W., Gu, M., Cheng, W. et al. Characteristics and function of cathepsin L3 from Schistosoma japonicum. Parasitol Res 119, 1619–1628 (2020). https://doi.org/10.1007/s00436-020-06647-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06647-x