Abstract
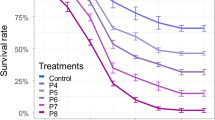
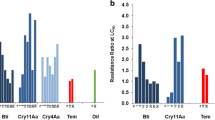
Mosquitoes are important vectors of many infectious diseases. Bacillus sphaericus (Bs) is an ideal larvicide and has attracted more and more attention, recently. However, the fundamental research of its application is very limited, especially on the subsequent impact of Bs exposure on mosquito’s fecundity and resistance emergence. Through bioassay, LC50 and LC95 of Bs in killing Anopheles dirus larvae were determined as 9.793 ± 1.878 IU/L and 62.4 ± 6.438 IU/L at 48 h posttreatment, 7.128 ± 0.913 IU/L and 34.385 ± 12.547 IU/L at 72 h post treatment, respectively. After being treated with a sub-lethal dose of Bs, gravidity, oviposition, hatch, pupation, and eclosion of the surviving mosquitoes were counted and analyzed to elucidate the subsequent effects of Bs exposure on the reproductive capacity of A. dirus. The result interestingly showed that the exposure of Bs significantly reduced the oviposition ability of the surviving A. dirus, without effect on egg formation/gravidity, hatch, pupation, and eclosion. The surviving mosquitoes were also maintained routinely for generations to test the sustained effect of Bs exposure on the fecundity of the offsprings. After conventional breeding for generations, the capacity of egg laying totally recovered. To explore the rules of resistance development, bioassays were performed after treatment twice with a sub-lethal dose of Bs on two continuous generations of A. dirus larvae. The killing efficacies between the Bs treated group and control group were compared. The results showed that LC50 and LC95 increased by 4.35- and 7.37-folds after treatment with the sub-lethal dose of Bs on two consecutive generations, respectively. The results indicated that A. dirus was sensitive to Bs, which could reduce oviposition of the surviving A. dirus. The subsequent effect might help to further decrease the mosquito population. However, a sub-lethal dose of Bs exposure could easily cause resistance development. Our study provides a dose standard and reference for the rational use of Bs, which will be helpful for mosquito control.





Similar content being viewed by others
References
Attoui H, Mohd Jaafar F, Belhouchet M, Aldrovandi N, Tao S, Chen B, Liang G, Tesh RB, de Micco P, de Lamballerie X (2005) Yunnan orbivirus, a new orbivirus species isolated from Culex tritaeniorhynchus mosquitoes in China. J Gen Virol 86(Pt 12):3409–3417. doi:10.1099/vir.0.81258-0
Berry C (2012) The bacterium, Lysinibacillus sphaericus, as an insect pathogen. J Invertebr Pathol 109(1):1–10. doi:10.1016/j.jip.2011.11.008
Corbel V, Akogbeto M, Damien GB, Djenontin A, Chandre F, Rogier C, Moiroux N, Chabi J, Banganna B, Padonou GG, Henry MC (2012) Combination of malaria vector control interventions in pyrethroid resistance area in Benin: a cluster randomised controlled trial. Lancet Infect Dis 12(8):617–626. doi:10.1016/S1473-3099(12)70081-6
el-Bendary MA (2006) Bacillus thuringiensis and Bacillus sphaericus biopesticides production. J Basic Microb 46(2):158–170. doi:10.1002/jobm.200510585
Gravitz L (2012) Vector control: the last bite. Nature 484(7395):S26–S27. doi:10.1038/484S26a
Huang JX, Xia ZG, Zhou SS, Pu XJ, Hu MG, Huang DC, Ren ZP, Zhang SS, Yang MN, Wang DQ, Wang JF (2015) Spatio-temporal analysis of malaria vectors in national malaria surveillance sites in China. Parasite Vectors 8:146. doi:10.1186/s13071-015-0741-5
Joshi D, Park MH, Saeung A, Choochote W, Min GS (2010) Multiplex assay to identify Korean vectors of malaria. Mol Ecol Resour 10(4):748–750. doi:10.1111/j.1755-0998.2010.02835.x
Kellen WR, Clark TB, Lindegren JE, Ho BC, Rogoff MH, Singer S (1965) Bacillus sphaericus Neide as a pathogen of mosquitoes. J Invertebr Pathol 7(4):442–448. doi:10.1016/0022-2011(65)90120-5
Majambere S, Lindsay SW, Green C, Kandeh B, Fillinger U (2007) Microbial larvicides for malaria control in The Gambia. Malar J 6:76. doi:10.1186/1475-2875-6-76
Morrison AC, Zielinski-Gutierrez E, Scott TW, Rosenberg R (2008) Defining challenges and proposing solutions for control of the virus vector Aedes aegypti. PLoS Med 5(3):e68. doi:10.1371/journal.pmed.0050068
Mwangangi JM, Kahindi SC, Kibe LW, Nzovu JG, Luethy P, Githure JI, Mbogo CM (2011) Wide-scale application of Bti/Bs biolarvicide in different aquatic habitat types in urban and peri-urban Malindi, Kenya. Parasitol Res 108(6):1355–1363. doi:10.1007/s00436-010-2029-1
Rodcharoen J, Mulla MS (1997) Biological fitness of Culex quinquefasciatus (Diptera:Culicidae) susceptible and resistant to Bacillus sphaericus. J Med Entomol 34(1):5–10
Shi L, Fu S, Wang L, Li X, Gu D, Liu C, Zhao C, He J, Liang G (2016) Surveillance of mosquito-borne infectious diseases in febrile travelers entering China via Shenzhen ports, China, 2013. Travel Med Infect Dis 14(2):123–130. doi:10.1016/j.tmaid.2016.02.002
Small ST, Reimer LJ, Tisch DJ, King CL, Christensen BM, Siba PM, Kazura JW, Serre D, Zimmerman PA (2016) Population genomics of the filarial nematode parasite Wuchereria bancrofti from mosquitoes. Mol Ecol 25(7):1465–1477. doi:10.1111/mec.13574
Suryadi BF, Yanuwiadi B, Ardyati T, Suharjono (2015) Isolation of Bacillus sphaericus from Lombok Island, Indonesia, and their toxicity against Anopheles aconitus. Int J Microbiol 2015:854709. doi:10.1155/2015/854709
Tikar SN, Mendki MJ, Sharma AK, Sukumaran D, Veer V, Prakash S, Parashar BD (2011) Resistance status of the malaria vector mosquitoes, Anopheles stephensi and Anopheles subpictus towards adulticides and larvicides in arid and semi-arid areas of India. J Insect Sci 11:85. doi:10.1673/031.011.8501
Tomori O (2002) Yellow fever in Africa: public health impact and prospects for control in the 21st century. Biomedica : revista del Instituto Nacional de Salud 22(2):178–210
Tripathi A, Hadapad AB, Hire RS, Melo JS, D’Souza SF (2013) Polymeric macroporous formulations for the control release of mosquitocidal Bacillus sphaericus ISPC-8. Enzym Microb Technol 53(6–7):398–405. doi:10.1016/j.enzmictec.2013.08.006
Wang J, Zhang H, Sun X, Fu S, Wang H, Feng Y, Wang H, Tang Q, Liang GD (2011) Distribution of mosquitoes and mosquito-borne arboviruses in Yunnan Province near the China-Myanmar-Laos border. AmJTrop Med Hyg 84(5):738–746. doi:10.4269/ajtmh.2011.10-0294
Wang Y, Zhong D, Cui L, Lee MC, Yang Z, Yan G, Zhou G (2015) Population dynamics and community structure of Anopheles mosquitoes along the China-Myanmar border. Parasit Vectors 8:445. doi:10.1186/s13071-015-1057-1
Yu G, Yan G, Zhang N, Zhong D, Wang Y, He Z, Yan Z, Fu W, Yang F, Chen B (2013) The Anopheles community and the role of Anopheles minimus on malaria transmission on the China-Myanmar border. Parasit Vectors 6(1):264. doi:10.1186/1756-3305-6-264
Acknowledgements
We would like to thank the staffs in our lab for their contribution in mosquitoes maintaining. And thanks to Prof. Jiannong Xu from New Mexico State University and Prof. Xiangyu Ma from Third Military Medical University for polishing the English expression. This study was supported by the National Natural Science Foundation of China (#81271875), the Military Scientific Research Project of China (CWS12J017), and National Institutes of Health, USA (U19AI089672).
Author information
Authors and Affiliations
Corresponding author
Additional information
Shasha Yu, Chen Ji, Xiaobo Zhu, and Jinwei Xue are authors who contributed equally to the work.
Rights and permissions
About this article
Cite this article
Yu, S., Ji, C., Zhu, X. et al. Impact of Bacillus sphaericus exposure on Anopheles dirus’s fecundity and resistance development. Parasitol Res 116, 859–864 (2017). https://doi.org/10.1007/s00436-016-5358-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-016-5358-x