Abstract
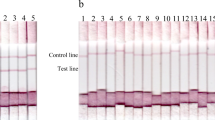
Animal trypanosomosis is a disease that is distributed worldwide which results in huge economic losses due to reduced animal productivity. Endemic regions are often located in the countryside where laboratory diagnosis is costly or inaccessible. The establishment of simple, effective, and accurate field tests is therefore of great interest to the farming and veterinary sectors. Our study aimed to develop a simple, rapid, and sensitive immunochromatographic test (ICT) for animal trypanosomosis utilizing the recombinant tandem repeat antigen TeGM6-4r, which is conserved amongst salivarian trypanosome species. In the specificity analysis, TeGM6-4r/ICT detected all of Trypanosoma evansi-positive controls from experimentally infected water buffaloes. As expected, uninfected controls tested negative. All sera samples collected from Tanzanian and Ugandan cattle that were Trypanosoma congolense- and/or Trypanosoma vivax-positive by microscopic examination of the buffy coat were found to be positive by the newly developed TeGM6-4r/ICT, which was comparable to results from TeGM6-4r/ELISA (kappa coefficient [κ] = 0.78). TeGM6/ICT also showed substantial agreement with ELISA using Trypanosoma brucei brucei (κ = 0.64) and T. congolense (κ = 0.72) crude antigen, suggesting the high potential of TeGM6-4r/ICT as a field diagnostic test, both for research purposes and on-site diagnosis of animal trypanosomosis.



Similar content being viewed by others
References
Barrett MP et al (2003) The trypanosomiases. Lancet 362(9394):1469–1480. doi:10.1016/s0140-6736(03)14694-6
Desquesnes M et al (2008) First outbreak of Trypanosoma evansi in camels in metropolitan France. Vet Rec 162(23):750–752
Fu E et al (2011) Enhanced sensitivity of lateral flow tests using a two-dimensional paper network format. Anal Chem 83(20):7941–7946. doi:10.1021/ac201950g
Gillingwater K, Mamabolo MV, Majiwa PAO (2010) Prevalence of mixed Trypanosoma congolense infections in livestock and tsetse in KwaZulu-Natal, South Africa. J S Afr Vet Assoc-Tydskr Suid-Afr Vet Ver 81(4):219–223
Goto Y et al (2011) Serological characterizations of tandem repeat proteins for detection of African trypanosome infection in cattle. Parasitol Int 60(4):538–540. doi:10.1016/j.parint.2011.09.003
Hirumi H, Hirumi K, Doyle JJ, Cross GA (1980) In vitro cloning of animal-infective bloodstream forms of Trypanosoma brucei. Parasitology 80(2):371–382
Kristjanson PM, Swallow BM, Rowlands GJ, Kruska RL, de Leeuw PN (1999) Measuring the costs of African animal trypanosomosis, the potential benefits of control and returns to research. Agric Syst 59(1):79–98. doi:10.1016/s0308-521x(98)00086-9
Laohasinnarong D et al (2011) Prevalence of Trypanosoma sp in cattle from Tanzania estimated by conventional PCR and loop-mediated isothermal amplification (LAMP). Parasitol Res 109(6):1735–1739. doi:10.1007/s00436-011-2513-2
Nguyen TT et al (2014) Diagnostic value of the recombinant tandem repeat antigen TeGM6-4r for surra in water buffaloes. Vet Parasitol 201(1–2):18–23. doi:10.1016/j.vetpar.2014.01.009
Nguyen TT et al (2015) Application of crude and recombinant ELISAs and immunochromatographic test for serodiagnosis of animal trypanosomosis in the Umkhanyakude District of KwaZulu-Natal Province, South Africa. J Vet Med Sci 77(2):217–220. doi:10.1292/jvms.14-0330
Njiru ZK, Ouma JO, Enyaru JC, Dargantes AP (2010) Loop-mediated isothermal amplification (LAMP) test for detection of Trypanosoma evansi strain B. Exp Parasitol 125(3):196–201. doi:10.1016/j.exppara.2010.01.017
OIE (2012) Trypanosoma evansi infection (surra). In: OIE (ed) Manual of diagnostic tests and vaccines for terrestrial animal, vol 1, 7th edn. OIE, Paris, pp 314–328
Paek SH, Lee SH, Cho JH, Kim YS (2000) Development of rapid one-step immunochromatographic assay. Methods 22(1):53–60. doi:10.1006/meth.2000.1036
Pillay D et al (2013) Trypanosoma vivax GM6 antigen: a candidate antigen for diagnosis of African animal trypanosomosis in cattle. PLoS One 8(10):10. doi:10.1371/journal.pone.0078565
Posthuma-Trumpie GA, Korf J, van Amerongen A (2009) Lateral flow (immuno) assay: its strengths, weaknesses, opportunities and threats. A literature survey. Anal Bioanal Chem 393(2):569–582. doi:10.1007/s00216-008-2287-2
Shaw APM (2009) Assessing the economics of animal trypanosomosis in Africa-history and current perspectives. Onderstepoort J Vet Res 76(1):27–32
Sullivan L, Wall SJ, Carrington M, Ferguson MAJ (2013) Proteomic selection of immunodiagnostic antigens for human African trypanosomiasis and generation of a prototype lateral flow immunodiagnostic device. Plos Neglect Trop Dis 7(2):14. doi:10.1371/journal.pntd.0002087
Tamarit A, Tejedor-Junco MT, Gonzalez M, Alberola J, Gutierrez C (2011) Morphological and biometrical features of Trypanosoma evansi isolates from an outbreak in mainland Spain. Vet Parasitol 177(1–2):152–156. doi:10.1016/j.vetpar.2010.11.044
Tanaka R et al (2006) A novel enhancement assay for immunochromatographic test strips using gold nanoparticles. Anal Bioanal Chem 385(8):1414–1420. doi:10.1007/s00216-006-0549-4
Thuy N, Goto Y, Lun ZR, Kawazu SI, Inoue N (2012) Tandem repeat protein as potential diagnostic antigen for Trypanosoma evansi infection. Parasitol Res 110(2):733–739. doi:10.1007/s00436-011-2632-9
Valiente-Gabioud AA, Veaute C, Perrig M, Galan-Romano FS, Sferco SJ, Marcipar IS (2011) Effect of repetitiveness on the immunogenicity and antigenicity of Trypanosoma cruzi FRA protein. Exp Parasitol 127(3):672–679. doi:10.1016/j.exppara.2010.11.011
Viera AJ, Garrett JM (2005) Understanding interobserver agreement: the kappa statistic. Fam Med 37(5):360–363
Acknowledgments
We thank Dr. Zhao-Rong Lun, Dr. Charles P. Otim, and Dr. Heriel A. Mbwambo for providing serum samples. This study was financially supported by the Ministry of Education, Cultural, Sports, Science, and Technology (MEXT), the Japan Society for the Promotion of Science (JSPS), and JST/JICA SATREPS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nguyen, TT., Ruttayaporn, N., Goto, Y. et al. A TeGM6-4r antigen-based immunochromatographic test (ICT) for animal trypanosomosis. Parasitol Res 114, 4319–4325 (2015). https://doi.org/10.1007/s00436-015-4672-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4672-z