Abstract
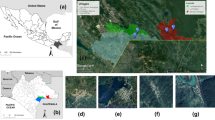
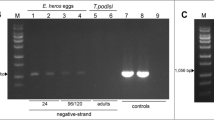
Since 2010, the number of cases of both human visceral leishmaniasis and cutaneous leishmaniasis in southwestern Madrid region (Spain) and more specifically in the town of Fuenlabrada has increased. Direct xenodiagnosis of leishmaniasis proved that hares (Lepus granatensis) from this focus are able to infect with Leishmania infantum colonized Phlebotomus perniciosus. To a better understanding of this focus of leishmaniasis, we conducted an entomological survey using CDC light traps, at the end of the seasonal transmission period of 2011 before the beginning of control measures of the disease, to study the phlebotomine sand flies species involved. Detection of Leishmania DNA in the sand flies captured was studied by kDNA-PCR and cpb-PCR. In addition, blood fed and gravid female P. perniciosus were analysed by a PCR based in vertebrate cytochrome b (cyt b) gene. Taxonomic identification of captured sand flies (n = 174) as P. perniciosus (n = 171) and Sergentomyia minuta (n = 3) together with the analysis of blood feeding in ten sand flies that shows a high preference for hares (n = 6), followed by humans (n = 3), and cats (n = 1) confirm a strong association between P. perniciosus hares and humans in the focus. Moreover, 79 out of 135 (58.5 %) P. perniciosus were positive to L. infantum by PCR approaches. These data support the increase of human leishmaniasis cases in the area and the existence of an unusual sylvatic cycle alternative to the classical domestic one, where the dog is the main reservoir of L. infantum.


Similar content being viewed by others
References
Abassi I, Cunio R, Warburg A (2009) Identification of bloodmeals imbibed by phlebotomine sand flies using cytochrome b PCR and reverse line blotting. Vector-Borne Zoonotic Dis 9(1):79–86
Alcover MM, Gramiccia M, Di Muccio T, Ballart C, Castillejo S, Picado A, Portús M, Gállego M (2012) Application of molecular techniques in the study of natural infection of Leishmania infantum vectors and utility of sandfly blood meal digestion for epidemiological surveys of leishmaniasis. Parasitol Res 111(2):515–523
Alves PC, Ferrand N, Suchentrunk F, Harrisa DJ (2003) Ancient introgression of Lepus timidus mtDNA into L. granatensis and L. europaeus in the Iberian Peninsula. Mol Phylogenet Evol 27:70–80
Alves-Pires C, Ribeiro H (1991) The phlebotomine sandflies of Portugal. V. Observations on the ecology of the vectors of leishmaniasis in the Alto Douro region. Parassitologia 33:63–68
Aransay AM, Scoulica E, Tselentis Y (2000) Detection and identification of Leishmania DNA within naturally infected sand flies by seminested PCR on minicircle kinetoplastic DNA. Appl Environ Microbiol 66:1933–1938
Azizi K, Abedi F, Moemenbellah-Fard MD (2012) Identification and frequency distribution of Leishmania (L.) major infections in sand flies from a new endemic ZCL focus in southeast Iran. Parasitol Res 111:1821–1826
Bongiorno G, Habluetzel A, Khoury C, Maroli M (2003) Host preferences of phlebotomine sand flies at a hypoendemic focus of canine leishmaniasis in central Italy. Acta Trop 88:119–123
Branco S, Alves-Pires C, Maia C, Cortes S, Cristovão JMS, Gonçalves L, Campino L, Afonso MO (2013) Entomological and ecological studies in a new potential zoonotic leishmaniasis focus in Torres Novas municipality, Central Region, Portugal. Acta Trop 125:339–348
Colmenares M, Portús M, Botet J, Dobaño C, Gállego M, Woff M, Seguí G (1995) Identification of blood meals of Phlebotomus perniciosus (Diptera:Psychodidae) in Spain by a competitive enzyme-linked immunosorbent assay biotin/avidin method. J Med Entomol 3:229–233
Conesa Gallego E, Romera Lozano E, Martínez Ortega E (1999) Estudio de las poblaciones de flebotomos (Diptera, Psychodidae) de la Comunidad de Madrid (España). Anales de Biología. 22 (Biología animal, 11) (1997): 43–50
Cruz I (2012) Re-Emerging visceral leishmaniasis in Europe and an outbreak in Spain. XVIII International Congress for Tropical Medicine and Malaria 1:73
El Tai NO, Osman OF, El Far M, Presber W, Schönian G (2000) Genetic heterogeneity of ribosomal internal transcribed spacer in clinical samples of Leishmania donovani spotted on filter paper as revealed by single-strand conformation polymorphisms and sequencing. Trans R Soc Trop Med Hyg 94:575–579
Gálvez R, Descalzo MA, Miró G, Jiménez MI, Martín O, Dos Santos-Brandao F, Guerrero I, Cubero E, Molina R (2010) Seasonal trends and spatial relations between environmental/meteorological factors and leishmaniosis sand fly vector abundances in Central Spain. Acta Trop 115:95–202
Guy MW, Killick-Kendrick R, Gill GS, Rioux JA, Bray RS (1984) Ecology of leishmaniasis in the south of France. 19. Determination of the hosts of Phlebotomus ariasi Tonnoir, 1921 in the Cévennes by bloodmeal analyses. Ann Parasitol Hum Comp 59:449–458
Haouas N, Pesson B, Boudabous R, Dedet J, Badda H, Ravel C (2007) Development of a molecular tool for the identification of Leishmania reservoir by bloodmeal analysis in the insect vectors. AmJTrop Med Hyg 77(6):1054–1059
Hide M, Bañuls A (2006) Species-specific PCR assay for L. infantum/L. donovani discrimination. Acta Trop 100:241–245
Kent RJ (2009) Molecular methods for arthropod bloodmeal identification and applications to ecological and vector-borne disease studies. Mol Ecol Resour 9:4–18
Killick-Kendrick R, Rioux JA (2002) Mark-release-recapture sand flies fed on leishmania dogs: the natural life-cycle of Leishmania infantum in Phlebotomus ariasi. Parassitologia 44:67–71
Kobets T, Badalova J, Grekov I, Havelková H, Svobodová M, Lipoldová M (2010) Leishmania parasite detection and quantification using PCR-ELISA. Nat Protoc 5:1074–1080
Kuhls K, Mauricio IL, Pratlong F, Presber W, Schönian G (2005) Analysis of ribosomal DNA internal transcribed spacer sequences of the Leishmania donovani complex. Microbes Infect 7:1224–1234
Maia C, Afonso MO, Neto L, Dionísio L, Campino L (2009) Molecular detection of Leishmania infantum in naturally infected hlebotomus perniciosus from Algarve Region, Portugal. J Vector Borne Dis 46:268–272
Martín-Sánchez J, Gállego M, Barón S, Castillejo S, Morillas-Marquez F (2006) Pool screen PCR for estimating the prevalence of Leishmania infantum infection in sandflies (Diptera: nematocera, Phlebotomidae). Trans R Soc Trop Med Hyg 100:527–532
Melo-Ferreira J, Boursot P, Carneiro M, Esteves PJ, Farelo L, Alves PC (2012) Recurrent introgression of mitochondrial DNA among hares (Lepus spp.) revealed by species-tree inference and coalescent simulations. Syst Biol 61:1–15
Michalsky EM, Fortes-Dias CI, Pimenta PFM, Secundino NFC, Dias ES (2002) Assessment of PCR in the detection of Leishmania spp in experimentally infected individual phlebotomine sandflies (Diptera: psychodidae: phlebotominae). Rev Inst Med Trop S Paulo 44:255–259
Molina R, Jiménez MI, Cruz I, Iriso A, Martín-Martín I, Sevillano O, Melero S, Bernal J (2012) The hare (Lepus granatensis) as potential sylvatic reservoir of Leishmania infantum in Spain. Vet Parasitol 190:268–271
Morillas F, Sanchíz-Marín MC, Martín-Sánchez J, Acedo Sánchez C (1991) On Phlebotomus perniciosus Newstead, 1911 (Diptera, Phlebotomidae) in the province of Almería in southeastern Spain. Parassitologia 33:437–444
Morillas F, Sanchez Rabasco F, Ocaña J, Martin-Sanchez J, Ocaña-Wihelmi J, Acedo C, Sanchiz-Marin MC (1996) Leishmaniosis in the focus of the Axarquía region, Malaga province, southern Spain: a survey of the human, dog, and vector. Parasitol Res 82:569–570
Oshaghi MA, Ravasan NM, Javadian E, Mohebali M, Hajjaran H, Zare Z, Mohtarami F, Rassi Y (2009) Vector incrimination of sand flies in the most important visceral leishmaniasis focus in Iran. Am J Trop Med Hyg 81:572–577
Quaresma PF, de Lima Carvalho GM, das Neves Farah Ramos MC, Andrade Filho JD (2012) Natural Leishmania spp. reservoirs and phlebotomine sandfly food source identification in Ibitipoca State Park, Minas Gerais, Brazil. Mem Inst Oswaldo Cruz 107(4):480–485
Rioux JA, Guilvard E, Gállego J, Moreno G, Pratlong F, Portús M, Rispail P, Gállego M, Bastien P (1986) Phlebotomus ariasi Tonnoir, 1921 et Phlebotomus perniciosus Newstead, 1911 vecteurs du complexe Leishmania infantum dans un même foyer: Infestations par deux zymodèmes syntopiques. A propos d´une enquête en Catalogne (Espagne). Leishmania: Taxonomie et Phylogenèse. IMEEE (Ed.) Montpellier, France, pp 439–444
Rossi E, Bongiorno G, Ciolli E, Di Muccio T, Scalone A, Gramiccia M, Gradoni L, Maroli M (2008) Seasonal phenology, host-blood feeding preferences and natural Leishmania infection of Phlebotomus perniciosus (Diptera, Psychodidae) in a high-endemic focus of canine leishmaniasis in Rome province, Italy. Acta Trop 105:158–165
Sant’Anna MRV, Jones NG, Hindley JA, Mesndes-Sousa AF, Dillon RJ, Cavalcante RR, Alexander B, Bates PA (2008) Bloodmeal identification and parasite detection in laboratory-fed and field-captured Lutzomya longipalpis by PCR using FTA databasing paper. Acta Trop 107:230–237
Steuber S, Abdel-Rady A, Clausen PH (2005) PCR-RFLP analysis: a promising technique for host species identification of blood meals from tsetse flies (Diptera: glossinidae). Parasitol Res 97:247–254
Suárez B, Isidoro B, Santos S, Sierra MJ, Molina R, Astray J, Amela C (2012) Situación epidemiológica y de los factores de riesgo de transmisión de Leishmania infantum en España. Rev Esp Salud Pública 86:555–564
Tiwananthagorn S, Bhutto AM, Baloch JH, Soomro FR, Kawamura Y, Nakao R, Aoshima K, Nonaka N, Oku Y, Katakura K (2012) Zoophilic feeding behavior of phlebotomine sand flies in the endemic areas of cutaneous leishmaniasis of Sindh Province, Pakistan. Parasitol Res 111:125–133
Torina A, Sole M, Reale S, Vitale F, Caracappa S (2008) Use of Phlebotomine Sand Flies as indicator of Leishmania prevalence in an endemic area. Animal biodiversity and emerging diseases. Ann NY Acad Sci 1149:355–357
World Health Organization (2010) Control of Leishmaniases. WHO Technical Report Series no. 949. Report of a meeting of the WHO Expert Committee on the Control of Leishmaniases, Geneva, 22–26 March 2010
Acknowledgments
We would like to thank Ángeles Vázquez and Ana Tello from the Departamento de Zoología y Antropología Física, Facultad de Ciencias Biológicas, Universidad Complutense, Madrid, Spain for their support. The authors are grateful to the Fuenlabrada and Leganés Councils. This study was partially sponsored and funded by the Subdirección General de Sanidad Ambiental y Epidemiología, the Dirección General de Ordenación e Inspección de la Consejería de Sanidad (CM), the Colegio de Veterinarios de Madrid, and the Colegio de Biólogos de Madrid. This study also was partially funded by EU grant GOCE-2003-010284 EDENext and is cataloged by the EDENext Steering Committee as EDENext 092 (http://www.edenext.eu). The contents of this publication are the sole responsibility of the authors and don’t necessarily reflect the views of the European Commission.
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Jiménez, M., González, E., Iriso, A. et al. Detection of Leishmania infantum and identification of blood meals in Phlebotomus perniciosus from a focus of human leishmaniasis in Madrid, Spain. Parasitol Res 112, 2453–2459 (2013). https://doi.org/10.1007/s00436-013-3406-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-013-3406-3