Abstract
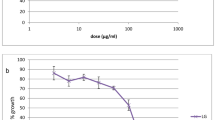
The absence of a vaccine and the rampant resistance to almost all antimalarial drugs have accentuated the urgent need for new antimalarial drugs and drug targets for both prophylaxis and chemotherapy. The aim of the study was to discover effective plant extracts against Plasmodium falciparum. In the present study, the hexane, chloroform, ethyl acetate, acetone, and methanol extracts of Citrus sinensis (peel), Leucas aspera, Ocimum sanctum, Phyllanthus acidus (leaf), Terminalia chebula (seed) were tested for their antimalarial activity against chloroquine (CQ)-sensitive (3D7) strain of P. falciparum which was cultured following the candle-jar method. Antimalarial evaluations of daily replacement of culture medium containing CQ and different plant crude extracts were performed on 96-well plates at 37°C for 24 and 48 h. Parasitemia was determined microscopically on thin-film Giemsa-stained preparations. Plant extracts were tested for their cytotoxicity using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay on human laryngeal cancer cell line (HEp-2) and normal cell line (Vero). Out of the 25 extracts tested, six showed good (IC50 4.76–22.76 μg/mL), 15 exhibited moderate (IC50 31.42–88.03 μg/mL), while four displayed mild (IC50 > 100 μg/mL) antiplasmodial activity. The leaf ethyl acetate and methanol extracts of L. aspera; ethyl acetate, acetone, and methanol extracts of P. acidus; and seed acetone extract of T. chebula had good antiplasmodial activity (IC50 = 7.81, 22.76, 9.37, 14.65, 12.68, and 4.76 μg/mL) with selectivity indices 5.43, 2.04, 4.88, 3.35, 3.42, and 9.97 for HEp-2 and >5.79, >2.20, >11.75, >3.41, >3.94, and >7.38 for Vero cells, respectively. These analyses have revealed for the first time that the components present in the solvent extracts of L. aspera, P. acidus, and T. chebula have antiplasmodial activity. The high antiplasmodial activity observed make these plants good candidates for isolation of anti-protozoal compounds which could serve as new lead structures for drug development.
Similar content being viewed by others
References
Abiodun O, Gbotosho G, Ajaiyeoba E, Happi T, Falade M, Wittlin S, Sowunmi A, Brun R, Oduola A (2010) In vitro antiplasmodial activity and toxicity assessment of some plants from Nigerian ethnomedicine. Pharm Biol. doi:10.3109/13880209.2010.490224
Adamu M, Nwosu CO, Agbede RI (2009) Anti-trypanosomal effects of aqueous extract of Ocimum gratissimum (Lamiaceae) leaf in rats infected with Trypanosoma brucei brucei. Afr J Tradit Complement Altern Med 6(3):262–267
Aderbauer B, Clausen PH, Kershaw O, Melzig MF (2008) In vitro and in vivo trypanocidal effect of lipophilic extracts of medicinal plants from Mali and Burkina Faso. J Ethnopharmacol 119(2):225–231
Adewunmi CO, Agbedahunsi JM, Adebajo AC, Aladesanmi AJ, Murphy N, Wando J (2001) Ethno-veterinary medicine: screening of Nigerian medicinal plants for trypanocidal properties. J Ethnopharmacol 77(1):19–24
Adjobimey T, Edayé I, Lagnika L, Gbenou J, Moudachirou M, Sanni A (2004) Activités antiplasmodiales in vitro de quelques plantes antipaludiques de la pharmacopée béninoise. CR Chim 7:1023–1027
Ahn MJ, Kim CY, Lee JS, Kim TG, Kim SH, Lee CK, Lee BB, Shin CG, Huh H, Kim J (2002) Inhibition of HIV-1 integrase by galloyl glucoses from Terminalia chebula and flavonol glycoside gallates from Euphorbia pekinensis. Planta Med 68(5):457–459
Albrecht P, Enterline JC, Boone EJ, Klutch MJ (1983) Poliovirus and polio antibody assay in HEp-2 and Vero cell cultures. J Biol Stand 11(2):91–97
Ali H, König GM, Khalid SA, Wright AD, Kaminsky R (2002) Evaluation of selected Sudanese medicinal plants for their in vitro activity against hemoflagellates, selected bacteria, HIV-1-RT and tyrosine kinase inhibitory, and for cytotoxicity. J Ethnopharmacol 83(3):219–228
Alves TMD, Nagem TJ, de Carvallo LH, Krefft AU, Zani CL (1997) Antiplasmodial triterpenes from Vernonia brasiliana. Planta Med 63:554–555
Anees AM (2008) Larvicidal activity of Ocimum sanctum Linn. (Labiatae) against Aedes aegypti (L.) and Culex quinquefasciatus (Say). Parasitol Res 103(6):1451–1453
Bagavan A, Rahuman AA, Kaushik NK, Sahal D (2010) In vitro antimalarial activity of medicinal plant extracts against Plasmodium falciparum. Parasitol Res. doi:10.1007/s00436-010-2034-4
Banzouzi JT, Prado R, Mena H, Valentin A, Roumetan C, Mallie M, Pelissier Y, Blache Y (2002) In vitro antiplasmodial activity of extracts of Alchornea cordifolia and identification of an active constituent: ellagic acid. J Ethnopharmacol 81:399–401
Bastos JK, Albuquerque S, Silva MLA (1999) Evaluation of the trypanocidal activity of lignans isolated from the leaves of Zanthoxylum naranjillo. Planta Med 65:541–544
Bhat GP, Surolia N (2001) In vitro antimalarial activity of extracts of three plants used in the traditional medicine of India. Am J Trop Med Hyg 65(4):304–308
Bizimana N, Tietjen U, Zessin KH, Diallo D, Djibril C, Melzig MF, Clausen PH (2006) Evaluation of medicinal plants from Mali for their in vitro and in vivo trypanocidal activity. J Ethnopharmacol 103(3):350–356
Breman JG (2001) The ears of the hippopotamus: manifestations, determinants, and estimates of the malaria burden. Am J Trop Med Hyg 64(1–2):1–11
Calixto JB, Santos ARS, Filho VC, Yunes RA (1998) A review of the plants of the genus Phyllanthus: their chemistry, pharmacology, and therapeutic potential. Med Res Rev 18(4):225–258
Chan KL, Choo CY, Abdullah NR, Ismail Z (2004) Antiplasmodial studies of Eurycoma longifolia Jack using the lactate dehydrogenase assay of Plasmodium falciparum. J Ethnopharmacol 92(2–3):223–227
Chandel S, Bagai U (2010) Antiplasmodial activity of Ajuga bracteosa against Plasmodium berghei infected BALB/c mice. Indian J Med Res 131:440–444
Christensen SB, Kharazmi A (2001) Antimalarial natural products. Isolation, characterisation and biological properties. In: Tringali C (ed) Bioactive compounds from natural sources. Taylor & Francis, London, pp 379–432
Chukwujekwu JC, Smith P, Coombes PH, Mulholland DA, van Staden J (2005) Antiplasmodial diterpenoid from the leaves of Hyptis suaveolens. J Ethnopharmacol 102(2):295–297
Cimanga RK, Tona L, Luyindula N, Mesia K, Lusakibanza M, Musuamba CT, Apers S, De Bruyne T, Van Miert S, Hermans N, Totté J, Pieters L, Vlietinck AJ (2004) In vitro antiplasmodial activity of callus culture extracts and fractions from fresh apical stems of Phyllanthus niruri L. (Euphorbiaceae): part 2. J Ethnopharmacol 95(2–3):399–404
de Almeida I, Alviano DS, Vieira DP, Alves PB, Blank AF, Lopes AH, Alviano CS, Rosa Mdo S (2007) Antigiardial activity of Ocimum basilicum essential oil. Parasitol Res 101(2):443–452
de Sá MS, Costa JF, Krettli AU, Zalis MG, Maia GL, Sette IM, de Câmara CA, Filho JM, Giulietti-Harley AM, RibeiroDosSantos R, Soares MB (2009) Antimalarial activity of betulinic acid and derivatives in vitro against Plasmodium falciparum and in vivo in P. berghei-infected mice. Parasitol Res 105(1):275–279
Dhar R, Zhang K, Talwar GP, Garg S, Kumar N (1998) Inhibition of the growth and development of asexual and sexual stages of drug-sensitive and resistant strains of the human malaria parasite Plasmodium falciparum by Neem (Azadirachta indica) fractions. J Ethnopharmacol 61(1):31–39
Dolabela MF, Oliveira SG, Nascimento JM, Peres JM, Wagner H, Póvoa MM, de Oliveira AB (2008) In vitro antiplasmodial activity of extract and constituents from Esenbeckia febrifuga, a plant traditionally used to treat malaria in the Brazilian Amazon. Phytomedicine 15(5):367–372
Fokialakis N, Kalpoutzakis E, Tekwani BL, Khan SI, Kobaisy M, Skaltsounis AL, Duke SO (2007) Evaluation of the antimalarial and antileishmanial activity of plants from the Greek island of Crete. J Nat Med 61:38–45
Fournet A, Barrios AA, Munoz V, Hocquemiller R, Roblot F, Cavé A, Richomme P, Bruneton J (1994) Antiprotozoal activity of quinoline alkaloids isolated from Galipea longiflora, a Bolivian plant used as a treatment for cutaneous leishmaniasis. Phytother Res 8(3):174–178
Gathirwa JW, Rukunga GM, Njagi EN, Omar SA, Guantai AN, Muthaura CN, Mwitari PG, Kimani CW, Kirira PG, Tolo FM, Ndunda TN, Ndiege IO (2007) In vitro anti-plasmodial and in vivo anti-malarial activity of some plants traditionally used for the treatment of malaria by the Meru community in Kenya. J Nat Med 61:261–268
Habila N, Agbaji AS, Ladan Z, Bello IA, Haruna E, Dakare MA, Atolagbe TO (2010) Evaluation of in vitro activity of essential oils against Trypanosoma brucei brucei and Trypanosoma evansi. J Parasitol Res. doi:10.1155/2010/534601
Hout S, Chea A, Bun SS, Elias R, Gasquet M, Timon-David P, Balansard G, Azas N (2006) Screening of selected indigenous plants of Cambodia for antiplasmodial activity. J Ethnopharmacol 107(1):12–18
Jonville MC, Kodja H, Humeau L, Fournel J, De Mol P, Cao M, Angenot L, Frédérich M (2008) Screening of medicinal plants from Reunion Island for antimalarial and cytotoxic activity. J Ethnopharmacol 120(3):382–386
Kamaraj C, Bagavan A, Rahuman AA, Zahir AA, Elango G, Pandiyan G (2009) Larvicidal potential of medicinal plant extracts against Anopheles subpictus Grassi and Culex tritaeniorhynchus Giles (Diptera: Culicidae). Parasitol Res 104(5):1163–1171
Kamaraj C, Rahuman AA, Bagavan A, Elango G, Rajakumar G, Zahir AA, Marimuthu S, Santhoshkumar T, Jayaseelan C (2010) Evaluation of medicinal plant extracts against blood-sucking parasites. Parasitol Res 106(6):1403–1412
Kassim OO, Loyevsky M, Elliott B, Geall A, Amonoo H, Gordeuk VR (2005) Effects of root extracts of Fagara zanthoxyloides on the in vitro growth and stage distribution of Plasmodium falciparum. Antimicrob Agents Chemother 49(1):264–268
Khan A, Ahmad A, Manzoor N, Khan LA (2010) Antifungal activities of Ocimum sanctum essential oil and its lead molecules. Nat Prod Commun 5(2):345–349
Khonkarn R, Okonogi S, Ampasavate C, Anuchapreeda S (2010) Investigation of fruit peel extracts as sources for compounds with antioxidant and antiproliferative activities against human cell lines. Food Chem Toxicol 48(8–9):2122–2129
Kirira P, Rukunga G, Wanyonyi A, Muregi F, Gathirwa J, Muthaura C, Omar S, Tolo F, Mungai G, Ndiege I (2006) Anti-plasmodial activity and toxicity of extracts of plants used in traditional malaria therapy in Meru and Kilifi Districts of Kenya. J Ethnopharmacol 106(3):403–407
Kuria KA, De Coster S, Muriuki G, Masengo W, Kibwage I, Hoogmartens J, Laekeman GM (2001) Antimalarial activity of Ajuga remota Benth (Labiatae) and Caesalpinia volkensii Harms (Caesalpiniaceae): in vitro confirmation of ethnopharmacological use. J Ethnopharmacol 74(2):141–148
Li S, Pan MH, Lai CS, Lo CY, Dushenkov S, Ho CT (2007) Isolation and syntheses of polymethoxyflavones and hydroxylated polymethoxyflavones as inhibitors of HL-60 cell lines. Bioorg Med Chem 15(10):3381–3389
Mahidol C, Prawat H, Prachyawarakorn V, Ruchirawat S (2002) Investigation of some bioactive Thai medicinal plants. Phytochem Rev 1:287–297
Mangathayaru K, Lakshmikant J, Shyam Sundar N, Swapna R, Grace XF, Vasantha J (2005) Antimicrobial activity of Leucas aspera flowers. Fitoterapia 76(7–8):752–754
Meléndez PA, Capriles VA (2006) Antibacterial properties of tropical plants from Puerto Rico. Phytomedicine 13(4):272–276
Moein MR, Pawar RS, Khan SI, Tekwani BL, Khan IA (2008) Antileishmanial, antiplasmodial and cytotoxic activities of 12, 16-dideoxy aegyptinone B from Zhumeria majdae Rech.f. & Wendelbo. Phytother Res 22(3):283–285
Morita H, Kishi E, Takeya K, Itokawa H, Tanaka O (1990) New quassinoids from the roots of Eurycoma longifolia. Chem Lett 5:749–752
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Muganga R, Angenot L, Tits M, Frédérich M (2010) Antiplasmodial and cytotoxic activities of Rwandan medicinal plants used in the treatment of malaria. J Ethnopharmacol 128(1):52–57
Murebwayire S, Frédérich M, Hannaert V, Jonville MC, Duez P (2008) Antiplasmodial and antitrypanosomal activity of Triclisia sacleuxii (Pierre) Diels. Phytomedicine 15(9):728–733
Mustofa, Valentin A, Benoit-Vical F, Pélissier Y, Koné-Bamba D, Mallié M (2000) Antiplasmodial activity of plant extracts used in west African traditional medicine. J Ethnopharmacol 73(1–2):145–151
Mustofa, Sholikhah EN, Wahyuono S (2007) In vitro and in vivo antiplasmodial activity and cytotoxicity of extracts of Phyllanthus niruri L. herbs traditionally used to treat malaria in Indonesia. Southeast Asian J Trop Med Public Health 38(4):609–615
Oliveira VC, Moura DM, Lopes JA, de Andrade PP, da Silva NH, Figueiredo RC (2009) Effects of essential oils from Cymbopogon citratus (DC) Stapf., Lippia sidoides Cham., and Ocimum gratissimum L. on growth and ultrastructure of Leishmania chagasi promastigotes. Parasitol Res 104(5):1053–1059
Omulokoli E, Khan B, Chhabra SC (1997) Antiplasmodial activity of four Kenyan medicinal plants. J Ethnopharmacol 56(2):133–137
Pessoa LM, Morais SM, Bevilaqua CM, Luciano JH (2002) Anthelmintic activity of essential oil of Ocimum gratissimum Linn. and eugenol against Haemonchus contortus. Vet Parasitol 109(1–2):59–63
Rahman MS, Sadhu SK, Hasan CM (2007) Preliminary antinociceptive, antioxidant and cytotoxic activities of Leucas aspera root. Fitoterapia 78(7–8):552–555
Ramazani A, Sardari S, Zakeri S, Vaziri B (2010) In vitro antiplasmodial and phytochemical study of five Artemisia species from Iran and in vivo activity of two species. Parasitol Res 107:593–599
Ratsimamanga-Urverg S, Rasoanaivo P, Rakoto-Ratsimamanga A, Le Bras J, Ramiliarisoa O, Savel J, Coulaud JP (1991) Antimalarial activity and cytotoxicity of Evodia fatraina stem bark extracts. J Ethnopharmacol 33(3):231–236
Sanon S, Ollivier E, Azas N, Mahiou V, Gasquet M, Ouattara CT, Nebie I, Traore AS, Esposito F, Balansard G, Timon-David P, Fumoux F (2003) Ethnobotanical survey and in vitro antiplasmodial activity of plants used in traditional medicine in Burkina Faso. J Ethnopharmacol 86(2–3):143–147
Santoro GF, Cardoso MG, Guimarães LG, Mendonça LZ, Soares MJ (2007) Trypanosoma cruzi: activity of essential oils from Achillea millefolium L., Syzygium aromaticum L. and Ocimum basilicum L. on epimastigotes and trypomastigotes. Exp Parasitol 116(3):283–290
Severino VG, Cazal Cde M, Forim MR, da Silva MF, Rodrigues-Filho E, Fernandes JB, Vieira PC (2009) Isolation of secondary metabolites from Hortia oreadica (Rutaceae) leaves through high-speed counter-current chromatography. J Chromatogr A 1216(19):4275–4281
Sharma VP (1999) Current scenario of malaria in India. Parassitologia 41:349–353
Shokeen P, Bala M, Singh M, Tandon V (2008) In vitro activity of eugenol, an active component from Ocimum sanctum, against multiresistant and susceptible strains of Neisseria gonorrhoeae. Int J Antimicrob Agents 32(2):174–179
Shuaibu MN, Wuyep PA, Yanagi T, Hirayama K, Tanaka T, Kouno I (2008) The use of microfluorometric method for activity-guided isolation of antiplasmodial compound from plant extracts. Parasitol Res 102(6):1119–1127
Staerk D, Kesting JR, Sairafianpour M, Witt M, Asili J, Emami SA, Jaroszewski JW (2009) Accelerated dereplication of crude extracts using HPLC-PDA-MS-SPE-NMR: quinolinone alkaloids of Haplophyllum acutifolium. Phytochemistry 70(8):1055–1061
Suzuki A, Shirota O, Mori K, Sekita S, Fuchino H, Takano A, Kuroyanagi M (2009) Leishmanicidal active constituents from Nepalese medicinal plant Tulsi (Ocimum sanctum L.). Chem Pharm Bull (Tokyo) 57(3):245–251
Tona L, Ngimbi NP, Tsakala M, Mesia K, Cimanga K, Apers S, De Bruyne T, Pieters L, Totté J, Vlietinck AJ (1999) Antimalarial activity of 20 crude extracts from nine African medicinal plants used in Kinshasa, Congo. J Ethnopharmacol 68(1–3):193–203
Tona L, Cimanga RK, Mesia K, Musuamba CT, De Bruyne T, Apers S, Hernans N, Van Miert S, Pieters L, Totté J, Vlietinck AJ (2004) In vitro antiplasmodial activity of extracts and fractions from seven medicinal plants used in the Democratic Republic of Congo. J Ethnopharmacol 93(1):27–32
Trager W, Jensen JB (1976) Human malaria parasites in continuous culture. Science 193:673–675
Unander DW, Webster GL, Blumberg BS (1995) Usage and bioassays in Phyllanthus (Euphorbiaceae). IV. Clustering of antiviral uses and other effects. J Ethnopharmacol 45(1):1–18
Valentin A, Pélissier Y, Benoit F, Marion C, Kone D, Mallié M, Bastide JM, Bessiére JM (1995) Composition and antimalarial activity in vitro of volatile components of Lippia multiflora. Phytochemistry 40:1439–1442
Valsaraj R, Pushpangadan P, Smitt UW, Adsersen A, Christensen SB, Sittie A, Nyman U, Nielsen C, Olsen CE (1997) New anti-HIV-1, antimalarial, and antifungal compounds from Terminalia bellerica. Nat Prod 60(7):739–742
Wansi JD, Hussain H, Tcho AT, Kouam SF, Specht S, Sarite SR, Hoerauf A, Krohn K (2010) Antiplasmodial activities of furoquinoline alkaloids from Teclea afzelii. Phytother Res 24(5):775–777
Were PS, Kinyanjui P, Gicheru MM, Mwangi E, Ozwara HS (2010) Prophylactic and curative activities of extracts from Warburgia ugandensis Sprague (Canellaceae) and Zanthoxylum usambarense (Engl.) Kokwaro (Rutaceae) against Plasmodium knowlesi and Plasmodium berghei. J Ethnopharmacol 130(1):158–162
Wernsdorfer WH, Trigg PI (1988) Recent progress of malaria research: chemotherapy. In: Wernsdorfer WH, McGregor I (eds) Malaria principles and practice of malariology, vol. 2. Churchill Livingstone, Edinburgh, pp 1569–1674
WHO (2002) World Health Organisation Fact Sheet No. 271. World Health Organisation, Geneva
Winstanley P (2001) Modern chemotherapeutic options for malaria. Lancet Infect Dis 1(4):242–250
Wright CW, Phillipson JD (1990) Natural products and the development of selective antiprotozoal drugs. Phytother Res 4:127–139
Yao QQ, Zuo CX (1993) Chemical studies on the constituents of Phyllanthus urinaria L. Yaoxue Xuebao 28:829–835
Acknowledgements
The authors are grateful to C. Abdul Hakeem of the College Management; Dr. S. Mohammed Yousuff, Principal; and Dr. K. Abdul Subhan, HOD of Zoology Department for providing the facilities to carry out this work. We are grateful to the Malaria Research Laboratory, International Centre for Genetic Engineering and Biotechnology, New Delhi, India, for providing P. falciparum. This investigation received financial support from the University Grants Commission (F.No.35-71/2008 SR), Government of India, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bagavan, A., Rahuman, A.A., Kamaraj, C. et al. Antiplasmodial activity of botanical extracts against Plasmodium falciparum . Parasitol Res 108, 1099–1109 (2011). https://doi.org/10.1007/s00436-010-2151-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-010-2151-0