Abstract
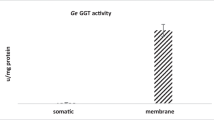
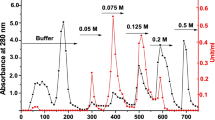
Glutathione (GSH) metabolism represents a potential target for antiparasitic drug design. Glutathione-S-transferase (GST), an important enzyme of the GSH cycle, is considered to be an essential detoxification enzyme in parasitic species. Soluble GST from rodent malarial parasites Plasmodium yoelii was purified to homogeneity using a combination of salt precipitation, affinity chromatography on GSH–sepharose 6B and ultrafiltration. Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) analysis revealed a single band and activity staining was also detected on PAGE gels. Kinetic studies on the purified enzyme revealed significant differences between the parasitic and mammalian enzymes. The purified enzyme exhibited an optimum pH of 8.2 and K m values of 0.2±0.213 and 3.3±0.056 mM with respect to co-substrate GSH and substrate 1-chloro-2, 4-dinitrobenzene (CDNB), respectively. Hemin, the known mammalian GST inhibitor was found to be a potent inhibitor of P. yoelii GST, with a K i of 4.0 μM.








Similar content being viewed by others
References
Anders MW, Dekant W (1998) Glutathione-dependent bioactivation of haloalkenes. Annu Rev Pharmacol Toxicol 38:501–537
Becker K, Rahlfs S, Nickel C, Schirmer RH (2003) Glutathione: function and metabolism in the malarial parasite Plasmodium falciparum. Biol Chem 348:551–566
Becker K, Tilley L, Vennerstrom JL, Roberts D, Rogerson S, Ginsburg H (2004) Oxidative stress in malaria parasite-infected erythrocytes: host–parasite interactions. Int J Parasitol 34:163–189
Board PG, Coggan M, Chelvanayagam G, Easteal S, Jermin LS, Schulte GK, Danley DE, Hoth LR, Griffor MC, Kamath AV (2000) Identification, characterization and crystal structure of the omega class glutathione transferases. J Biol Chem 275:24798–24806
Dekant W, Vamvakas S, Anders MW (1994) Formation and fate of nephrotoxic and cytotoxic cysteine conjugates: cysteine conjugate β-lyase pathway. In: Anders MW, Dekant W (eds) Advances in pharmacology. Academic Press, London, pp 115–162
Denton H, McGregor JC, Coombs GH (2004) Reduction of antileishmanial pentavalent antimonial drugs by a parasite-specific thiol dependent reductase TDR1. Biochem J 381:405–412
Deponte M, Becker K (2005) Glutathione-S-transferase from malarial parasites: structural and functional aspects. Methods Enzymol 404:241–253
Dubois VL, Platel DF, Pauly G, Tribouley-Duret J (1995) Plasmodium berghei: implication of intracellular glutathione and its related enzyme in chloroquine resistance in vivo. Exp Parasitol 81:117–124
Ferone R, Burchall JJ, Hitchings GH (1969) Plasmodium berghei dihydrofolate reductase: isolation, properties and inhibition by antifolates. Mol Pharmacol 5:49–59
Fritz-Wolf K, Becker A, Rahlfs S, Harwaldt P, Schirmer RH, Kabsch W, Becker K (2003) X-ray structure of glutathione-S-transferase from the malarial parasite Plasmodium falciparum. Proc Natl Acad Sci USA 100:13821–13826
Habig HW, Pabst MJ, Jakoby WB (1974) Glutathione-S-transferase(s): the first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Harwaldt P, Rahlfs S, Becker K (2002) Glutathione-S-transferase of the malarial parasite Plasmodium falciparum: characterization of a potential drug target. Biol Chem 383:821–830
Hemingway J (2000) The molecular basis of two contrasting metabolic mechanisms of insecticide resistance. Insect Biochem Mol Biol 30:1009–1015
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature (London) 227:680–685
Liebau E, Bergmann B, Campbell AM, Teesdale-Spittle P, Brophy PM, Lüersen K, Walter RD (2002) The glutathione-S-transferase from Plasmodium falciparum. Mol Biochem Parasitol 124:85–90
Listowsky I (2005) A subclass of mu glutathione-S-transferases selectively expressed in testis and brain. Methods Enzymol 401:278–287
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–276
Morgenstein R, DePierre W (1988) Membrane-bound glutathione transferases. In: Sies H, Ketterer B (eds) Glutahione conjugation. Academic press, London, pp 157–175
Müller S, Liebau E, Walter RD, Krauth-Siegel RL (2003) Thiol-based redox metabolism of protozoan parasites. Trends Parasitol 19:320–328
Murphy SC, Breman JG (2001) Gaps in the childhood malaria burden in Africa: cerebral malaria, neurological sequelae, anemia, respiratory distress, hypoglycemia, and complications of pregnancy. Am J Trop Med Hyg 64:57–67
Ouaissi MA, Dubremetz JF, Schöneck R, Fernandez-Gomez R, Gomez-Corvera R, Billaut-Mulot O et al (1995) Trypanosoma cruzi: a 52 kDa protein sharing sequence homology with glutathione-S-transferase is localized in parasite organelles morphologically resembling reservosomes. Exp Parasitol 81:453–461
Perbandt M, Burmeister C, Walter RD, Betzel C, Liebau E (2004) Native and inhibited structure of a Mu class-related glutathione S-transferase from Plasmodium falciparum. J Biol Chem 279:1336–1342
Platel DF, Mangou F, Tribouley-Duret J (1999) Role of glutathione in the detoxification of ferriprotoporphyrin IX in chloroquine resistant Plasmodium berghei. Mol Biochem Parasitol 98:215–223
Rahlfs S, Schirmer RH, Becker K (2002) The thioredoxin system of Plasmodium falciparum and other parasites. Cell Mol Life Sci 59:1024–1041
Ricci G, Bello ML, Caccuri AM, Galiazzo F, Federici G (1984) Detection of glutathione-S-transferase activity on polyacrylamide gels. Anal Biochem 143:226–230
Richards WH, Williams SG (1973) The removal of leucocytes from malaria infected blood. Ann Trop Med Parasitol 67:249–250
Salinas AE, Wong MG (1999) Glutathione-S-transferase(s)—a review. Curr Med Chem 6:279–309
Sheehan D, Meade G, Foley VM, Dowd CA (2001) Structure, function and evolution of glutathione-S-transferase(s): implications for classification of non-mammalian members of an ancient enzyme superfamily. Biochem J 360:1–16
Simons PC, Vander Jagt DL (1977) Purification of GST(s) from human liver by glutathione affinity chromatography. Anal Biochem 82:334–341
Srivastava P, Puri SK, Kamboj KK, Pandey VC (1999) Glutathione-S-transferase activity in malarial parasites. Trop Med Int Health 4:251–254
Sztajer H, Gamain B, Aumann KD, Slomianny C, Becker K, Brigelius-Flohé R, Flohé L (2001) The putative glutathione peroxidase gene of Plasmodium falciparum codes for a thioredoxin peroxidase. J Biol Chem 276:7397–7403
Townsend D, Tew K (2003) Cancer drugs, genetic variation and the glutathione-S-transferase gene family. Am J Pharmacogenomics 3:157–172
Vickers TJ, Fairlamb AH (2004) Trypanothione S-transferase activity in a trypanosomatid ribosomal elongation factor 1B. J Biol Chem 279:27246–27256
Acknowledgements
This investigation received financial support from Council of Scientific and Industrial Research, New Delhi (India), in the form of a Senior Research Fellowship to R. Ahmad and Volkswagen Stiftung, Hannover (Germany) in the form of ad-hoc research grant to A.K. Srivastava. We are also grateful to Dr. S.K. Puri, Head, Parasitology Division, Central Drug Research Institute, Lucknow, India, for providing the P. yoelii nigeriensis-infected albino mice for maintenance of P. yoelii infection in the present work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmad, R., Srivastava, A.K. Purification and biochemical characterization of cytosolic glutathione-S-transferase from malarial parasites Plasmodium yoelii . Parasitol Res 100, 581–588 (2007). https://doi.org/10.1007/s00436-006-0295-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-006-0295-8