Abstract
Purpose
The aim of the present study was to investigate whether the gene expression levels of LKB1 and LGR5 correlated with clinical outcome in patients with locally advanced rectal cancer treated with preoperative chemoradiotherapy (CRT).
Methods
Residual cancer cells were obtained from 52 patients with locally advanced rectal cancer treated with preoperative CRT. Total RNA was then isolated from formalin-fixed, paraffin-embedded specimens using microdissection. The expression levels of LKB1 and LGR5 genes were measured using real-time reverse-transcription polymerase chain reaction and by immunohistochemistry. In addition, in vitro studies were performed using colon cancer cell lines to study the serial changes of LKB1, LGR5 and PRKAA1 (AMPK) gene expression levels after irradiation.
Results
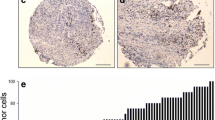
Our data demonstrate that specimens obtained from patients with poor pathological response and tumor recurrence had significantly higher gene expression levels of LKB1 and LGR5 than those without them (P < 0.05), and there was a significant positive correlation between LKB1 and LGR5 gene expression after CRT (Spearman’s ρ: 0.429, P = 0.0023). The patients with high expression levels of both LKB1 and LGR5 had a significantly lower recurrence-free survival compared with the other group (P = 0.0055, 95 % confidence interval: 1.39–11.08). Lastly, in vitro studies demonstrated a similar pattern of serial gene expression among LKB1, LGR5 and PRKAA1 after irradiation.
Conclusions
Our results suggest that LKB1 and LGR5 expression may be implicated in resistance to CRT, therefore contributing to tumor relapse in patients with locally advanced rectal cancer treated with preoperative CRT.




Similar content being viewed by others
References
Alexander A, Walker CL (2011) The role of LKB1 and AMPK in cellular responses to stress and damage. FEBS Lett 585:952–957
Alexander A, Cai SL, Kim J, Nanez A, Sahin M, MacLean KH, Inoki K, Guan KL, Shen J, Person MD, Kusewitt D, Mills GB, Kastan MB, Walker CL (2010) ATM signals to TSC2 in the cytoplasm to regulate mTORC1 in response to ROS. Proc Natl Acad Sci U S A 107:4153–4158
Barcellos-Hoff MH, Park C, Wright EG (2005) Radiation and the microenvironment—tumorigenesis and therapy. Nat Rev Cancer 5:867–875
Barker N, van Es JH, Kuipers J, Kujala P, van den Born M, Cozijnsen M, Haegebarth A, Korving J, Begthel H, Peters PJ, Clevers H (2007) Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449:1003–1007
Barker N, Ridgway RA, van Es JH, van de Wetering M, Begthel H, van den Born M, Danenberg E, Clarke AR, Sansom OJ, Clevers H (2009) Crypt stem cells as the cells-of-origin of intestinal cancer. Nature 457:608–611
Bateman AC, Jaynes E, Bateman AR (2009) Rectal cancer staging post neoadjuvant therapy–how should the changes be assessed? Histopathology 54:713–721
Carretero J, Shimamura T, Rikova K, Jackson AL, Wilkerson MD, Borgman CL, Buttarazzi MS, Sanofsky BA, McNamara KL, Brandstetter KA, Walton ZE, Gu TL, Silva JC, Crosby K, Shapiro GI, Maira SM, Ji H, Castrillon DH, Kim CF, Garcia-Echeverria C, Bardeesy N, Sharpless NE, Hayes ND, Kim WY, Engelman JA, Wong KK (2010) Integrative genomic and proteomic analyses identify targets for Lkb1-deficient metastatic lung tumors. Cancer Cell 17:547–559
Clevers H (2011) The cancer stem cell: premises, promises and challenges. Nat Med 17:313–319
Guillem JG, Chessin DB, Cohen AM, Shia J, Mazumdar M, Enker W, Paty PB, Weiser MR, Klimstra D, Saltz L, Minsky BD, Wong WD (2005) Long-term oncologic outcome following preoperative combined modality therapy and total mesorectal excision of locally advanced rectal cancer. Ann Surg 241:829–836 (discussion 836–838)
Gurumurthy S, Xie SZ, Alagesan B, Kim J, Yusuf RZ, Saez B, Tzatsos A, Ozsolak F, Milos P, Ferrari F, Park PJ, Shirihai OS, Scadden DT, Bardeesy N (2011) The Lkb1 metabolic sensor maintains haematopoietic stem cell survival. Nature 468:659–663
Haegebarth A, Clevers H (2009) Wnt signaling, lgr5, and stem cells in the intestine and skin. Am J Pathol 174:715–721
Herrmann JL, Byekova Y, Elmets CA, Athar M (2011) Liver kinase B1 (LKB1) in the pathogenesis of epithelial cancers. Cancer Lett 306:1–9
Jansen M, de Leng WW, Baas AF, Myoshi H, Mathus-Vliegen L, Taketo MM, Clevers H, Giardiello FM, Offerhaus GJ (2006) Mucosal prolapse in the pathogenesis of Peutz-Jeghers polyposis. Gut 55:1–5
Jansen M, Ten Klooster JP, Offerhaus GJ, Clevers H (2009) LKB1 and AMPK family signaling: the intimate link between cell polarity and energy metabolism. Physiol Rev 89:777–798
Lai C, Robinson J, Clark S, Stamp G, Poulsom R, Silver A (2011) Elevation of WNT5A expression in polyp formation in Lkb1 ± mice and Peutz-Jeghers syndrome. J Pathol 223:584–592
Ma Y, Zhang G, Fu X, Xia O, Zhan C, Li L, Wang Z, Wu B (2010) Wnt signaling may be activated in a subset of Peutz-Jeghers syndrome polyps closely correlating to LKB1 expression. Oncol Rep 23:1569–1576
Merlos-Suarez A, Barriga FM, Jung P, Iglesias M, Cespedes MV, Rossell D, Sevillano M, Hernando-Momblona X, da Silva-Diz V, Munoz P, Clevers H, Sancho E, Mangues R, Batlle E (2011) The intestinal stem cell signature identifies colorectal cancer stem cells and predicts disease relapse. Cell Stem Cell 8:511–524
Nakada D, Saunders TL, Morrison SJ (2010) Lkb1 regulates cell cycle and energy metabolism in haematopoietic stem cells. Nature 468:653–658
Nguyen DH, Oketch-Rabah HA, Illa-Bochaca I, Geyer FC, Reis-Filho JS, Mao JH, Ravani SA, Zavadil J, Borowsky AD, Jerry DJ, Dunphy KA, Seo JH, Haslam S, Medina D, Barcellos-Hoff MH (2011) Radiation acts on the microenvironment to affect breast carcinogenesis by distinct mechanisms that decrease cancer latency and affect tumor type. Cancer Cell 19:640–651
Ryan R, Gibbons D, Hyland JM, Treanor D, White A, Mulcahy HE, O’Donoghue DP, Moriarty M, Fennelly D, Sheahan K (2005) Pathological response following long-course neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Histopathology 47:141–146
Saigusa S, Tanaka K, Toiyama Y, Yokoe T, Okugawa Y, Ioue Y, Miki C, Kusunoki M (2009) Correlation of CD133, OCT4, and SOX2 in rectal cancer and their association with distant recurrence after chemoradiotherapy. Ann Surg Oncol 16:3488–3498
Sanchez-Cespedes M, Parrella P, Esteller M, Nomoto S, Trink B, Engles JM, Westra WH, Herman JG, Sidransky D (2002) Inactivation of LKB1/STK11 is a common event in adenocarcinomas of the lung. Cancer Res 62:3659–3662
Sapkota GP, Deak M, Kieloch A, Morrice N, Goodarzi AA, Smythe C, Shiloh Y, Lees-Miller SP, Alessi DR (2002) Ionizing radiation induces ataxia telangiectasia mutated kinase (ATM)-mediated phosphorylation of LKB1/STK11 at Thr-366. Biochem J 368:507–516
Sauer R, Becker H, Hohenberger W, Rodel C, Wittekind C, Fietkau R, Martus P, Tschmelitsch J, Hager E, Hess CF, Karstens JH, Liersch T, Schmidberger H, Raab R (2004) Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med 351:1731–1740
Shaw RJ (2009) LKB1 and AMP-activated protein kinase control of mTOR signalling and growth. Acta Physiol (Oxf) 196:65–80
Shaw RJ, Kosmatka M, Bardeesy N, Hurley RL, Witters LA, DePinho RA, Cantley LC (2004) The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc Natl Acad Sci U S A 101:3329–3335
Shen Z, Wen XF, Lan F, Shen ZZ, Shao ZM (2002) The tumor suppressor gene LKB1 is associated with prognosis in human breast carcinoma. Clin Cancer Res 8:2085–2090
Spicer J, Ashworth A (2004) LKB1 kinase: master and commander of metabolism and polarity. Curr Biol 14:R383–R385
Uchida H, Yamazaki K, Fukuma M, Yamada T, Hayashida T, Hasegawa H, Kitajima M, Kitagawa Y, Sakamoto M (2010) Overexpression of leucine-rich repeat-containing G protein-coupled receptor 5 in colorectal cancer. Cancer Sci 101:1731–1737
Udd L, Makela TP (2011) LKB1 signaling in advancing cell differentiation. Fam Cancer 10:425–435
van den Brink M, Stiggelbout AM, van den Hout WB, Kievit J, Klein Kranenbarg E, Marijnen CA, Nagtegaal ID, Rutten HJ, Wiggers T, van de Velde CJ (2004) Clinical nature and prognosis of locally recurrent rectal cancer after total mesorectal excision with or without preoperative radiotherapy. J Clin Oncol 22:3958–3964
van der Flier LG, Clevers H (2009) Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu Rev Physiol 71:241–260
Wingo SN, Gallardo TD, Akbay EA, Liang MC, Contreras CM, Boren T, Shimamura T, Miller DS, Sharpless NE, Bardeesy N, Kwiatkowski DJ, Schorge JO, Wong KK, Castrillon DH (2009) Somatic LKB1 mutations promote cervical cancer progression. PLoS ONE 4:e5137
Yan D, Avtanski D, Saxena NK, Sharma D (2012) Leptin-induced epithelial-mesenchymal transition in breast cancer cells requires beta-catenin activation via Akt/GSK3- and MTA1/Wnt1 protein-dependent pathways. J Biol Chem 287:8598–8612
Yoshikawa R, Kusunoki M, Yanagi H, Noda M, Furuyama JI, Yamamura T, Hashimoto-Tamaoki T (2001) Dual antitumor effects of 5-fluorouracil on the cell cycle in colorectal carcinoma cells: a novel target mechanism concept for pharmacokinetic modulating chemotherapy. Cancer Res 61:1029–1037
Zeki SS, Graham TA, Wright NA (2011) Stem cells and their implications for colorectal cancer. Nat Rev Gastroenterol Hepatol 8:90–100
Zhong D, Liu X, Khuri FR, Sun SY, Vertino PM, Zhou W (2008) LKB1 is necessary for Akt-mediated phosphorylation of proapoptotic proteins. Cancer Res 68:7270–7277
Acknowledgments
The authors would like to thank Motoko Ueeda and Yuka Kato for providing excellent technical assistance.
Conflict of interest
Susumu Saigusa and other authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saigusa, S., Inoue, Y., Tanaka, K. et al. Significant correlation between LKB1 and LGR5 gene expression and the association with poor recurrence-free survival in rectal cancer after preoperative chemoradiotherapy. J Cancer Res Clin Oncol 139, 131–138 (2013). https://doi.org/10.1007/s00432-012-1308-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-012-1308-x