Abstract
Purpose
Nine previously reported associations between single nucleotide polymorphisms (SNPs) and breast cancer outcomes from the Shanghai Breast Cancer Study (Stage 1) were further evaluated in relation to disease-free survival (DFS) and overall survival (OS) among 5,192 additional breast cancer patients (Stage 2).
Methods
Hazard ratios (HR) and 95% confidence intervals (CI) were calculated by proportional hazards regression in models adjusted for age, disease stage, estrogen and progesterone receptor status, and treatment regimens.
Results
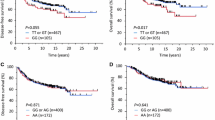
Two SNPs had generally consistent results and significant associations with OS in combined analyses. Compared to women with MMP7 rs11225297 AA genotypes, OS was moderately better for women with AT genotypes (HR: 0.8, 95% CI: 0.7–1.0) and much better for women with TT genotypes (HR: 0.4, 95% CI: 0.2–0.8). Compared to women with MMP8 rs11225395 CC genotypes, OS was slightly better for women with CT genotypes (HR: 0.9, 95% CI: 0.7–1.1) and moderately better for women with TT genotypes (HR: 0.6, 95% CI: 0.4–0.9). Joint analysis showed significant dose–response relationships with increasing numbers of rare alleles for both OS (p < 0.001) and DFS (p = 0.001).
Conclusions
A functional variant in MMP8 and a SNP in high linkage disequilibrium with a functional variant in MMP7 were significantly associated with breast cancer survival in a large two-stage survival study among Chinese women. This supports the hypothesis that SNPs in matrix metalloproteinase genes may influence breast cancer prognosis; additional research on these and other SNPs in genes important in metastasis, angiogenesis, and the regulation of the tumor microenvironment is warranted.

Similar content being viewed by others
References
Abraham JE, Harrington P, Driver KE, Tyrer J, Easton DF, Dunning AM et al (2009) Common polymorphisms in the prostaglandin pathway genes and their association with breast cancer susceptibility and survival. Clin Cancer Res 15:2181–2191
Ambrosone CB, Ahn J, Singh KK, Rezaishiraz H, Furberg H, Sweeney C et al (2005) Polymorphisms in genes related to oxidative stress (MPO, MnSOD, CAT) and survival after treatment for breast cancer. Cancer Res 65:1105–1111
Azzato E, Driver K, Lesueur F, Shah M, Greenberg D, Easton D et al (2008) Effects of common germline genetic variation in cell cycle control genes on breast cancer survival: results from a population-based cohort. Breast Cancer Res 10:R47
Beeghly-Fadiel A, Long JR, Gao YT, Li C, Qu S, Cai Q et al (2008) Common MMP-7 polymorphisms and breast cancer susceptibility: a multistage study of association and functionality. Cancer Res 68:6453–6459
Beeghly-Fadiel A, Shu XO, Long J, Li C, Cai Q, Cai H et al (2009) Genetic polymorphisms in the MMP-7 gene and breast cancer survival. Int J Cancer 124:208–214
Decock J, Long JR, Laxton RC, Shu XO, Hodgkinson C, Hendrickx W et al (2007) Association of matrix metalloproteinase-8 gene variation with breast cancer prognosis. Cancer Res 67:10214–10221
Decock J, Hendrickx W, Vanleeuw U, Van Belle V, Van Huffel S, Christiaens MR et al (2008) Plasma MMP1 and MMP8 expression in breast cancer: protective role of MMP8 against lymph node metastasis. BMC Cancer 8:77
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174
Hojilla CV, Mohammed FF, Khokha R (2003) Matrix metalloproteinases and their tissue inhibitors direct cell fate during cancer development. Br J Cancer 89:1817–1821
Jiang WG, Davies G, Martin TA, Parr C, Watkins G, Mason MD et al (2005) Targeting matrilysin and its impact on tumor growth in vivo: the potential implications in breast cancer therapy. Clin Cancer Res 11:6012–6019
Knechtel G, Hofmann G, Gerger A, Renner W, Langsenlehner T, Szkandera J et al (2010) Analysis of common germline polymorphisms as prognostic factors in patients with lymph node-positive breast cancer. J Cancer Res Clin Oncol 136:1813–1819
Long JR, Cai Q, Shu XO, Cai H, Gao YT, Zheng W (2007) Genetic polymorphisms in estrogen-metabolizing genes and breast cancer survival. Pharmacogenet Genomics 17:331–338
Lu H, Shu XO, Cui Y, Kataoka N, Wen W, Cai Q et al (2005) Association of genetic polymorphisms in the VEGF gene with breast cancer survival. Cancer Res 65:5015–5019
Martin M, Matrisian L (2007) The other side of MMPs: protective roles in tumor progression. Cancer Metastasis Rev 26:717–724
Saarialho-Kere UK, Crouch EC, Parks WC (1995) Matrix metalloproteinase matrilysin is constitutively expressed in adult human exocrine epithelium. J Invest Dermatol 105:190–196
Shin A, Cai Q, Shu XO, Gao YT, Zheng W (2005) Genetic polymorphisms in the matrix metalloproteinase 12 gene (MMP12) and breast cancer risk and survival: the Shanghai breast cancer study. Breast Cancer Res 7:R506–R512
Shu XO, Gao YT, Cai Q, Pierce L, Cai H, Ruan ZX et al (2004) Genetic polymorphisms in the TGF-beta 1 gene and breast cancer survival: a report from the Shanghai breast cancer study. Cancer Res 64:836–839
Shu XO, Moore DB, Cai Q, Cheng J, Wen W, Pierce L et al (2005) Association of Cyclin D1 genotype with breast cancer risk and survival. Cancer Epidemiol Biomarkers Prev 14:91–97
Shu XO, Zheng Y, Cai H, Gu K, Chen Z, Zheng W et al (2009) Soy food intake and breast cancer survival. JAMA J Am Med Assoc 302:2437–2443
Skol AD, Scott LJ, Abecasis GR, Boehnke M (2006) Joint analysis is more efficient than replication-based analysis for two-stage genome-wide association studies. Nat Genet 38:209–213
Udler MS, Azzato EM, Healey CS, Ahmed S, Pooley KA, Greenberg D et al (2009) Common germline polymorphisms in COMT, CYP19A1, ESR1, PGR, SULT1E1 and STS and survival after a diagnosis of breast cancer. Int J Cancer 125:2687–2696
Wilson CL, Matrisian LM (1996) Matrilysin: an epithelial matrix metalloproteinase with potentially novel functions. Int J Biochem Cell Biol 28:123–136
Zhang X, Shu XO, Cai Q, Ruan Z, Gao YT, Zheng W (2006) Functional plasminogen activator inhibitor-1 gene variants and breast cancer survival. Clin Cancer Res 12:6037–6042
Zheng W, Long J, Gao YT, Li C, Zheng Y, Xiang YB et al (2009) Genome-wide association study identifies a new breast cancer susceptibility locus at 6q25.1. Nat Genet 41:324–328
Acknowledgments
The authors wish to thank Dr. Fan Jin for her contributions to data collection and study implementation, Dr. Wanqing Wen for assistance with Kaplan–Meier analyses, and Ms. Bethanie Rammer and Ms. Jacqueline Stern for manuscript editing and submission. We also gratefully acknowledge the participants and research staff of the Shanghai Breast Cancer Study and Shanghai Breast Cancer Survival Study. This research was supported by grants from the National Institute of Health, National Cancer Institute (R01 CA064277 and R01 CA124558, PI: W. Zheng and R01 CA118229, PI: XO Shu), and the Department of Defense Breast Cancer Research Program (DAMD 17-02-1-0607, PI: XO Shu). Dr. Beeghly-Fadiel is supported in part by a grant from the National Institutes of Health, National Institute of Child Health and Human Development (5K12 HD043483-09; PI: N. Brown).
Conflict of Interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beeghly-Fadiel, A., Zheng, W., Lu, W. et al. Replication study for reported SNP associations with breast cancer survival. J Cancer Res Clin Oncol 138, 1019–1026 (2012). https://doi.org/10.1007/s00432-012-1174-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-012-1174-6