Abstract
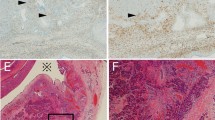
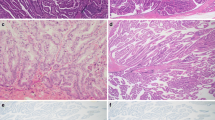
A thickened, enhanced cyst wall on imaging examinations is one of the “worrisome features” described in the consensus guidelines for management of intraductal papillary mucinous neoplasm of the pancreas (IPMN). Podoplanin (PDPN) expression by cancer-associated fibroblasts is known to be an indicator of poor prognosis in some types of cancer. We performed immunohistochemical staining for alpha-smooth muscle actin (α-SMA) in IPMN lesions and determined the pathological wall thickness by measuring the thinnest and thickest α-SMA-positive parts of the wall of the largest cyst in each case, and the mean of these two values was recorded as the wall thickness. The thickness of the pathological wall increased with progression from IPMN with low-grade dysplasia to IPMN with an invasive carcinoma. The pathological wall was thicker in IPMN with main duct involvement, nongastric-type IPMN, and IPMN with mural nodules. We also stained for PDPN and assessed the thickness of cyst wall staining as for α-SMA. The thickness of the PDPN-positive cyst wall varied in a pattern similar to the thickness of the α-SMA-positive pathological cyst wall. PDPN-positive stromal fibroblasts in the invasive component of IPMN-IC were evaluated as a ratio to α-SMA-positive fibroblasts. A high ratio (>50 %) of PDPN-positive stromal fibroblasts was a predictor of poor outcome. PDPN expression in the cyst wall correlates with the progression of IPMN. PDPN may be a significant prognostic marker of IPMN-IC.







Similar content being viewed by others
Abbreviations
- PDPN:
-
Podoplanin
- IPMN:
-
Intraductal papillary mucinous neoplasm of the pancreas
- IPMN-L:
-
IPMN with low-grade dysplasia
- IPMN-I:
-
IPMN with intermediate-grade dysplasia
- IPMN-H:
-
IPMN with high-grade dysplasia
- IPMN-IC:
-
IPMN with an associated invasive carcinoma
- PB:
-
Pancreatobiliary
- HE:
-
Hematoxylin and eosin
References
Adsay NV, Fukushima N, Furukawa T et al (2010) Intraductal neoplasms of the pancreas. In: Bosman FT, Carneiro F, Hruban RH et al (eds) WHO classification of tumours of the digestive system, 4th edn. IARC Press, Lyon, pp 304–313
Takaori K (2007) Current understanding of precursors to pancreatic cancer. J Hepatobiliary Pancreat Surg 14:217–223
Farrell JJ, Fernandez-del Castillo C (2013) Pancreatic cystic neoplasms: management and unanswered questions. Gastroenterology 144:1303–1315
Distler M, Kersting S, Niedergethmann M, Aust DE, Franz M, Ruckert F, Ehehalt F, Pilarsky C, Post S, Saeger HD, Grutzmann R (2013) Pathohistological subtype predicts survival in patients with intraductal papillary mucinous neoplasm (IPMN) of the pancreas. Ann Surg 258:324–330
Mino-Kenudson M, Fernandez-del Castillo C, Baba Y et al (2011) Prognosis of invasive intraductal papillary mucinous neoplasm depends on histological and precursor epithelial subtypes. Gut 60:1712–1720
Tanaka M, Fernandez-del Castillo C, Adsay V et al (2012) International consensus guidelines 2012 for the management of IPMN and MCN of the pancreas. Pancreatology 12:183–197
Kalluri R, Zeisberg M (2006) Fibroblasts in cancer. Nat Rev Cancer 6:392–401
Polanska UM, Orimo A (2013) Carcinoma-associated fibroblasts: non-neoplastic tumour-promoting mesenchymal cells. J Cell Physiol 228:1651–1657
Cirri P, Chiarugi P (2011) Cancer associated fibroblasts: the dark side of the coin. Am J Cancer Res 1:482–1497
Kawase A, Ishii G, Nagai K, Ito T, Nagano T, Murata Y, Hishida T, Nishimura M, Yoshida J, Suzuki K, Ochiai A (2008) Podoplanin expression by cancer associated fibroblasts predicts poor prognosis of lung adenocarcinoma. Int J Cancer 123:1053–1059
Kitano H, Kageyama S, Hewitt SM, Hayashi R, Doki Y, Ozaki Y, Fujino S, Takikita M, Kubo H, Fukuoka J (2010) Podoplanin expression in cancerous stroma induces lymphangiogenesis and predicts lymphatic spread and patient survival. Arch Pathol Lab Med 134:1520–1527
Pula B, Jethon A, Piotrowska A, Gomulkiewicz A, Owczarek T, Calik J, Wojnar A, Witkiewicz W, Rys J, Ugorski M, Dziegiel P, Podhorska-Okolow M (2011) Podoplanin expression by cancer-associated fibroblasts predicts poor outcome in invasive ductal breast carcinoma. Histopathology 59:1249–1260
Wicki A, Lehembre F, Wick N, Hantusch B, Kerjaschki D, Christofori G (2006) Tumor invasion in the absence of epithelial-mesenchymal transition: podoplanin-mediated remodeling of the actin cytoskeleton. Cancer Cell 9:261–272
Honma M, Minami-Hori M, Takahashi H, Iizuka H (2012) Podoplanin expression in wound and hyperproliferative psoriatic epidermis: regulation by TGF-beta and STAT-3 activating cytokines, IFN-gamma, IL-6, and IL-22. J Dermatol Sci 65:134–140
Morimatsu K, Aishima S, Yamamoto H, Hayashi A, Nakata K, Oda Y, Shindo K, Fujino M, Tanaka M, Oda Y (2013) Insulin-like growth factor II messenger RNA-binding protein-3 is a valuable diagnostic and prognostic marker of intraductal papillary mucinous neoplasm. Hum Pathol 44:1714–1721
Furukawa T, Kloppel G, Volkan Adsay N, Albores-Saavedra J, Fukushima N, Horii A, Hruban RH, Kato Y, Klimstra DS, Longnecker DS, Luttges J, Offerhaus GJ, Shimizu M, Sunamura M, Suriawinata A, Takaori K, Yonezawa S (2005) Classification of types of intraductal papillary-mucinous neoplasm of the pancreas: a consensus study. Virchows Arch 447:794–799
Aishima S, Nishihara Y, Iguchi T, Taguchi K, Taketomi A, Maehara Y, Tsuneyoshi M (2008) Lymphatic spread is related to VEGF-C expression and D2-40-positive myofibroblasts in intrahepatic cholangiocarcinoma. Mod Pathol 21:256–264
Cuzick J (1985) A Wilcoxon-type test for trend. Stat Med 4:87–90
Shields MA, Dangi-Garimella S, Redig AJ, Munshi HG (2012) Biochemical role of the collagen-rich tumour microenvironment in pancreatic cancer progression. Biochem J 441:541–552
Rodriguez JR, Salvia R, Crippa S, Warshaw AL, Bassi C, Falconi M, Thayer SP, Lauwers GY, Capelli P, Mino-Kenudson M, Razo O, McGrath D, Pederzoli P, Fernández-Del Castillo C (2007) Branch-duct intraductal papillary mucinous neoplasms: observations in 145 patients who underwent resection. Gastroenterology 133:72–79, quiz 309-310
Bassi C, Crippa S, Salvia R (2008) Intraductal papillary mucinous neoplasms (IPMNs): is it time to (sometimes) spare the knife? Gut 57:287–289
Hwang DW, Jang JY, Lee SE, Lim CS, Lee KU, Kim SW (2012) Clinicopathologic analysis of surgically proven intraductal papillary mucinous neoplasms of the pancreas in SNUH: a 15-year experience at a single academic institution. Langenbecks Arch Surg 397:93–102
Kim KW, Park SH, Pyo J, Yoon SH, Byun JH, Lee MG, Krajewski KM, Ramaiya NH (2014) Imaging features to distinguish malignant and benign branch-duct type intraductal papillary mucinous neoplasms of the pancreas: a meta-analysis. Ann Surg 259:72–81
Rautou PE, Levy P, Vullierme MP, O’Toole D, Couvelard A, Cazals-Hatem D, Palazzo L, Aubert A, Sauvanet A, Hammel P, Hentic O, Rebours V, Pelletier AL, Maire F, Ruszniewski P (2008) Morphologic changes in branch duct intraductal papillary mucinous neoplasms of the pancreas: a midterm follow-up study. Clin Gastroenterol Hepatol 6:807–814
Ideno N, Ohtsuka T, Kono H, Fujiwara K, Oda Y, Aishima S, Ito T, Ishigami K, Tokunaga S, Ohuchida K, Takahata S, Nakamura M, Mizumoto K, Tanaka M (2013) Intraductal papillary mucinous neoplasms of the pancreas with distinct pancreatic ductal adenocarcinomas are frequently of gastric subtype. Ann Surg 258:141–151
Nakamura A, Horinouchi M, Goto M, Nagata K, Sakoda K, Takao S, Imai K, Kim YS, Sato E, Yonezawa S (2002) New classification of pancreatic intraductal papillary-mucinous tumour by mucin expression: its relationship with potential for malignancy. J Pathol 197:201–210
Adsay NV, Merati K, Andea A, Sarkar F, Hruban RH, Wilentz RE, Goggins M, Iocobuzio-Donahue C, Longnecker DS, Klimstra DS (2002) The dichotomy in the preinvasive neoplasia to invasive carcinoma sequence in the pancreas: differential expression of MUC1 and MUC2 supports the existence of two separate pathways of carcinogenesis. Mod Pathol 15:1087–1095
Okabayashi T, Shima Y, Kosaki T, Sumiyoshi T, Kozuki A, Iiyama T, Takezaki Y, Kobayashi M, Nishimori I, Ogawa Y, Hanazaki K (2013) Invasive carcinoma derived from branch duct-type IPMN may be a more aggressive neoplasm than that derived from main duct-type IPMN. Oncol Lett 5:1819–1825
Van Laethem JL, Resibois A, Rickaert F et al (1997) Different expression of transforming growth factor beta 1 in pancreatic ductal adenocarcinoma and cystic neoplasms. Pancreas 15:41–47
Shindo K, Aishima S, Ohuchida K, Fujiwara K, Fujino M, Mizuuchi Y, Hattori M, Mizumoto K, Tanaka M, Oda Y (2013) Podoplanin expression in cancer-associated fibroblasts enhances tumor progression of invasive ductal carcinoma of the pancreas. Mol Cancer 12:168
Hoshino A, Ishii G, Ito T, Aoyagi K, Ohtaki Y, Nagai K, Sasaki H, Ochiai A (2011) Podoplanin-positive fibroblasts enhance lung adenocarcinoma tumor formation: podoplanin in fibroblast functions for tumor progression. Cancer Res 71:4769–4779
Okada K, Hirabayashi K, Imaizumi T, Matsuyama M, Yazawa N, Dowaki S, Tobita K, Ohtani Y, Tanaka M, Inokuchi S, Makuuchi H (2010) Stromal thrombospondin-1 expression is a prognostic indicator and a new marker of invasiveness in intraductal papillary-mucinous neoplasm of the pancreas. Biomed Res 31:13–19
Acknowledgements
This work was supported in part by a Grant-in-Aid from the Japan Society for the Promotion of Science (JSPS) fellows. We thank Edanz Group Japan for English language editing.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource Fig. 1
Immunohistochemical staining of normal pancreatic duct. (a) Normal pancreatic ducts had α-SMA-positive cells sparsely. (b) PDPN was negative in the normal pancreatic ducts. (c) Lymphatic vessels were positive for PDPN without exception, and used as a positive control. Original magnification: (a,b) × 40 (c) × 100 (GIF 698 kb)
ESM 1
(TIFF 1233 kb)
Rights and permissions
About this article
Cite this article
Shindo, K., Aishima, S., Ohuchida, K. et al. Podoplanin expression in the cyst wall correlates with the progression of intraductal papillary mucinous neoplasm. Virchows Arch 465, 265–273 (2014). https://doi.org/10.1007/s00428-014-1610-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-014-1610-x