Abstract
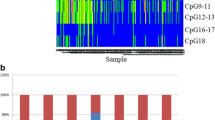
Gastric carcinoma (GC) is one of the human cancers in which promoter CpG island hypermethylation is frequently found. CpG island methylator phenotype (CIMP) refers to a subset of GCs which harbor concordant methylation of multiple promoter CpG island loci. However, little is known regarding clinicopathological features of CIMP-positive (CIMP-high) GC. Our study aimed to characterize clinicopathological features of CIMP-high GC. We analyzed 196 cases of GCs for their methylation status in 16 cancer-specific CpG island loci using MethyLight assay and arbitrarily defined CIMP-high GC as those with methylation at 13 or more CpG island loci. With exclusion of microsatellite instability-positive GC and EBV-positive GC from the analysis, CIMP-high GC (n = 10, 6.7%) demonstrated tendency toward higher cancer stage, infiltrative growth type, poor differentiation, and diffuse or mixed type of Lauren classification. CIMP-high GC showed significantly shortened survival compared with that of CIMP-negative GC. When CIMP-negative GC (methylation at 12 or less) was divided into CIMP-intermediate and CIMP-low (methylation at one or none), CIMP-low exhibited better clinical outcome than CIMP-intermediate. Hypermethylation at 14 CpG island loci or more was closely associated with poor clinical outcome and found to be an independent prognostic factor. Our findings that CIMP-high GCs were featured with characteristic clinicopathological parameters, including poor prognosis are distinct from previous studies. More extensive, large-scaled study is necessary to validate the findings of the present study.



Similar content being viewed by others
References
Toyota M, Ahuja N, Ohe-Toyota M et al (1999) CpG island methylator phenotype in colorectal cancer. Proc Natl Acad Sci USA 96:8681–8686
Weisenberger DJ, Siegmund KD, Campan M et al (2006) CpG island methylator phenotype underlies sporadic microsatellite instability and is tightly associated with BRAF mutation in colorectal cancer. Nat Genet 38:787–793
Ogino S, Goel A (2008) Molecular classification and correlates in colorectal cancer. J Mol Diagn 10:13–27
Kim JH, Shin SH, Kwon HJ, Cho NY, Kang GH (2009) Prognostic implications of CpG island hypermethylator phenotype in colorectal cancers. Virchows Arch 455:485–494
Toyota M, Ahuja N, Suzuki H et al (1999) Aberrant methylation in gastric cancer associated with the CpG island methylator phenotype. Cancer Res 59:5438–5442
Enomoto S, Maekita T, Tsukamoto T et al (2007) Lack of association between CpG island methylator phenotype in human gastric cancers and methylation in their background non-cancerous gastric mucosae. Cancer Sci 98:1853–1861
Kang GH, Lee S, Kim WH et al (2002) Epstein-barr virus-positive gastric carcinoma demonstrates frequent aberrant methylation of multiple genes and constitutes CpG island methylator phenotype-positive gastric carcinoma. Am J Pathol 160:787–794
Chang MS, Uozaki H, Chong JM et al (2006) CpG island methylation status in gastric carcinoma with and without infection of Epstein-Barr virus. Clin Cancer Res 12:2995–3002
Oue N, Oshimo Y, Nakayama H et al (2003) DNA methylation of multiple genes in gastric carcinoma: association with histological type and CpG island methylator phenotype. Cancer Sci 94:901–905
An C, Choi IS, Yao JC et al (2005) Prognostic significance of CpG island methylator phenotype and microsatellite instability in gastric carcinoma. Clin Cancer Res 11:656–663
Kusano M, Toyota M, Suzuki H et al (2006) Genetic, epigenetic, and clinicopathologic features of gastric carcinomas with the CpG island methylator phenotype and an association with Epstein-Barr virus. Cancer 106:1467–1479
Chan AO, Peng JZ, Lam SK et al (2006) Eradication of Helicobacter pylori infection reverses E-cadherin promoter hypermethylation. Gut 55:463–468
Chan AO, Lam SK, Wong BC et al (2003) Promoter methylation of E-cadherin gene in gastric mucosa associated with Helicobacter pylori infection and in gastric cancer. Gut 52:502–506
Boland CR, Thibodeau SN, Hamilton SR et al (1998) A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res 58:5248–5257
Kang GH, Lee S, Cho NY et al (2008) DNA methylation profiles of gastric carcinoma characterized by quantitative DNA methylation analysis. Lab Invest 88:161–170
Yoo EJ, Park SY, Cho NY et al (2008) Helicobacter pylori-infection-associated CpG island hypermethylation in the stomach and its possible association with polycomb repressive marks. Virchows Arch 452:515–524
Park SY, Yoo EJ, Cho NY, Kim N, Kang GH (2009) Comparison of CpG island hypermethylation and repetitive DNA hypomethylation in premalignant stages of gastric cancer, stratified for Helicobacter pylori infection. J Pathol 219:410–416
Park SY, Kook MC, Kim YW et al (2010) Mixed-type gastric cancer and its association with high-frequency CpG island hypermethylation. Virchows Arch 456(6):625–633
Ogino S, Cantor M, Kawasaki T et al (2006) CpG island methylator phenotype (CIMP) of colorectal cancer is best characterised by quantitative DNA methylation analysis and prospective cohort studies. Gut 55:1000–1006
Ogino S, Kawasaki T, Brahmandam M et al (2006) Precision and performance characteristics of bisulfite conversion and real-time PCR (MethyLight) for quantitative DNA methylation analysis. J Mol Diagn 8:209–217
Goel A, Nagasaka T, Arnold CN et al (2007) The CpG island methylator phenotype and chromosomal instability are inversely correlated in sporadic colorectal cancer. Gastroenterology 132:127–138
Acknowledgement
Supported by a grant of the Korea Healthcare Technology R&D Project, Ministry for Health, Welfare and Family Affairs, Republic of Korea (A091081).
Disclosure
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, SY., Kook, M.C., Kim, Y.W. et al. CpG island hypermethylator phenotype in gastric carcinoma and its clinicopathological features. Virchows Arch 457, 415–422 (2010). https://doi.org/10.1007/s00428-010-0962-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-010-0962-0