Abstract
Main conclusion
In this report, we demonstrate that Brachypodium distachyon could serve as a relatively high throughput in planta functional assay system for Triticeae anther-specific gene promoters.
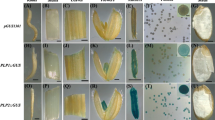
There remains a vast gap in our knowledge of the promoter cis-acting elements responsible for the transcriptional regulation of Triticeae anther-specific genes. In an attempt to identify conserved cis-elements, 14 pollen-specific and 8 tapetum-specific Triticeae putative promoter sequences were analyzed using different promoter sequence analysis tools. Several cis-elements were found to be enriched in these sequences and their possible role in gene expression regulation in the anther is discussed. Despite the fact that potential cis-acting elements can be identified within putative promoter sequence datasets, determining whether particular promoter sequences can in fact direct proper tissue-specific and developmental gene expression still needs to be confirmed via functional assays preferably performed in closely related plants. Transgenic functional assays with Triticeae species remain challenging and Brachypodium distachyon may represent a suitable alternative. The promoters of the triticale pollen-specific genes group 3 pollen allergen (PAL3) and group 4 pollen allergen (PAL4), as well as the tapetum-specific genes chalcone synthase-like 1 (CHSL1), from wheat and cysteine-rich protein 1 (CRP1) from triticale were fused to the green fluorescent protein gene (GFP) and analyzed in transgenic Brachypodium. This report demonstrates that this model species could serve to accelerate the functional analysis of Triticeae anther-specific gene promoters.




Similar content being viewed by others
References
Abe H, Urao T, Ito T, Seki M, Shinozaki K, Yamaguchi-Shinozaki K (2003) Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell 15:63–78
Azria D, Bhalla PL (2011) Agrobacterium-mediated transformation of Australian rice varieties and promoter analysis of major pollen allergen gene, Ory s 1. Plant Cell Rep 30:1673–1681
Badea A, Eudes F, Laroche A, Graf R, Doshi K, Amundsen E, Nilsson D, Puchalski B (2013) Antimicrobial peptides expressed in wheat reduce susceptibility to Fusarium head blight and powdery mildew. Can J Plant Sci 93:199–208
Bailey TL, Bodén M, Buske FA, Frith M, Grant CE, Clementi L, Ren J, Li WW, Noble WS (2009) MEME SUITE; tools for motif discovery and searching. Nucleic Acids Res 37:W202–W208
Berckmans B, Lammens T, Van Den Daele H, Magyar Z, Bögre L, De Veyder L (2011) Light-dependent regulation of DEL1 is determined by the antagonistic action of E2Fb and E2fc. Plant Physiol 157:1440–1451
Buck MJ, Lieb JD (2004) ChIP-chip: considerations for the design, analysis, and application of genome-wide chromatin immunoprecipitation experiments. Genomics 83:349–360
Chen L, Tu Z, Hussain J, Cong L, Yan Y, Jin L, Yang G, He G (2010) Isolation and heterologous transformation analysis of a pollen-specific promoter from wheat (Triticum aestivum L.). Mol Biol Rep 37:737–744
Chen L, Miao Y, Wang C, Su P, Li T, Wang R, Hao X, Yang G, He G, Gao C (2012) Characterization of a novel pollen-specific promoter from wheat (Triticum aestivum L.). Plant Mol Biol Rep 30:1426–1432
Chhun T, Aya K, Asano K, Yamamoto E, Morinaka Y, Watanabe M, Kitano H, Ashikari M, Matsuoka M, Ueguchi-Tanaka M (2007) Gibberellin regulates pollen viability and pollen tube growth in rice. Plant Cell 19:3876–3888
De Wilde C, Van Houdt H, De Buck S, Angenon G, De Jaeger G, Depicker A (2000) Plants as bioreactors for protein production: avoiding the problem of transgene silencing. Plant Mol Biol 43:347–359
Draper J, Mur LAJ, Jenkins G, Ghosh-Biswas GC, Bablak P, Hasterok R, Routledge APM (2001) Brachypodium distachyon. A new model system for functional genomics in grasses. Plant Physiol 127:1539–1555
Ericson ML, Murén E, Gustavsson H, Josefsson L, Rask L (1991) Analysis of the promoter region of napin genes from Brassica napus demonstrates binding of nuclear protein in vitro to a conserved sequence motif. Eur J Biochem 197:741–746
Ezcurra I, Wycliffe P, Nehlin L, Ellerström M, Rask L (2000) Transactivation of the Brassica napus napin promoter by ABI3 requires interaction of the conserved B2 and B3 domains of ABI3 with different cis-elements: B2 mediates activation through an ABRE, whereas B3 interacts with an RY/G-box. Plant J 24:57–66
Fujiwara T, Beachy RN (1994) Tissue-specific and temporal regulation of a β-conglycinin gene: roles of the RY repeat and other cis-acting elements. Plant Mol Biol 24:261–272
Grotewold E, Drummond BJ, Bowen B, Peterson T (1994) The myb-homologous P gene controls phlobaphen pigmentation in maize floral organs by directly activating a flavonoid biosynthetic gene subset. Cell 76:543–553
Gubler F, Jacobsen JV (1992) Gibberellin-responsive elements in the promoter of a barley high-pI α-amylase gene. Plant Cell 4:1435–1441
Gupta V, Khurana R, Tyagi AK (2007) Promoters of two anther-specific genes confer organ-specific gene expression in a stage-specific manner in transgenic systems. Plant Cell Rep 26:1919–1931
Hamilton DA, Schwarz YH, Mascarenhas JP (1998) A monocot pollen-specific promoter contains separable pollen-specific and quantitative elements. Plant Mol Biol 38:663–669
Hensel G, Himmelbach A, Chen W, Douchkov DK, Kumlehn J (2011) Transgene expression systems in the Triticeae cereals. J Plant Physiol 168:30–44
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucl Acids Res 27:297–300
Hishiguro S, Kawai-Oda A, Ueda J, Nishida I, Okada K (2001) The DEFECTIVE IN ANTHER DEHISCENCE1 gene encodes a novel phospholipase A1 catalyzing the initial step of jasmonic acid biosynthesis, which synchronizes pollen maturation, anther dehiscence, and flower opening in Arabidopsis. Plant Cell 13:2191–2209
Huang Z, Gan Z, He Y, Li Y, Liu X, Mu H (2011) Functional analysis of a rice late pollen-abundant UDP-glucose pyrophosphorylase (OsUgp2) promoter. Mol Biol Rep 38:4291–4302
Hwang Y-S, Karrer EE, Thomas BR, Chen L, Rodriguez RL (1998) Three cis-elements required for rice α-amylase Amy3D expression during sugar starvation. Plant Mol Biol 36:331–341
Jefferson RA (1987) Assaying chimeric genes in plants: The GUS gene fusion system. Plant Mol Biol Rep 5:387–405
Jiang SY, Cai M, Ramachandran S (2005) The Oryza sativa no pollen (Osnop) gene plays a role in male gametophyte development and most likely encodes a C2-GRAM domain-containing protein. Plant Mol Biol 57:835–853
Jin Y, Bian T (2004) Isolation and partial characterization of a novel pollen-specific cDNA with multiple polyadenylation sites from wheat. Acta Biochim Biophys Sin (Shanghai) 36:467–476
Joshi CP (1987) Putative polyadenylation signals in nuclear genes of higher plants: a compilation and analysis. Nucleic Acids Res 15:9627–9640
Kempe K, Rubtsova M, Berger C, Kumlehn J, Schollmeier C, Gils M (2010) Transgene excision from wheat chromosomes by phage phiC31 integrase. Plant Mol Biol 72:673–687
Khurana R, Kapoor S, Tyagi AK (2012) Anthology of anther/pollen-specific promoters and transcription factors. Crit Rev Plant Sci 31:359–390
Khurana R, Kapoor S, Tyagi AK (2013) Spatial and temporal activity of upstream regulatory regions of rice anther-specific genes in transgenic rice and Arabidopsis. Transgenic Res 22:31–46
Kim SY, Chung H, Thomas TL (1997) Isolation of a novel class of bZIP transcription factors that interact with ABA-responsive and embryo-specification elements in the Dc3 promoter using a modified yeast one-hybrid system. Plant J 11:1237–1251
Kim DW, Lee SH, Choi S, Won S, Heo Y, Cho M, Park Y, Cho H (2006) Functional conservation of a root hair cell-specific cis-element in angiosperms with different root hair distribution patterns. Plant Cell 18:2958–2970
Kuriakose B, Arun V, Gnanamanickam SS, Thomas G (2009) Tissue-specific expression in transgenic rice and Arabidopsis thaliana plants of GUS gene driven by the 5′ regulatory sequences of an anther specific rice gene YY2. Plant Sci 177:390–397
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y, Rouzé P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucl Acids Res 30:325–327
Li H, Pinot F, Sauveplane V, Werck-Reichhart D, Diehl P, Schreiber L, Franke R, Zhang P, Chen L, Gao Y, Liang W, Zhang D (2010) Cytochrome P450 family member CYP704B2 catalyzes the ω-hydroxylation of fatty acids and is required for anther cutin biosynthesis and pollen exine formation in rice. Plant Cell 22:173–190
Li H, Yuan Z, Vizcay-Barrena G, Yang C, Liang W, Zong J, Wilson ZA, Zhang D (2011) PERSISTENT TAPETAL CELL1 encodes a PHD-finger protein that is required for tapetal cell death and pollen development in rice. Plant Physiol 156:615–630
Luo H, Lee J, Hu Q, Nelson-Vasilchik K, Eitas TK, Lickwar C, Kausch AP, Chandlee JM, Hodges TK (2006) RTS, a rice anther-specific gene is required for male fertility and its promoter sequence directs tissue-specific gene expression in different plant species. Plant Mol Biol 62:397–408
Maruyama K, Todaka D, Mizoi J, Yoshida T, Kidokoro S, Matsukura S, Takasaki H, Sakurai T, Yamamot YY, Yoshiwara K, Kojima M, Sakakibara H, Shinozaki K, Yamaguchi-Shinozaki K (2012) Identification of cis-acting promoter elements in cold- and dehydration-induced transcriptional pathways in Arabidopsis, rice and soybean. DNA Res 19:37–49
Mizelle MB, Sethi R, Ashton ME, Jensen WD (1989) Development of the pollen grain and tapetum of wheat (Triticum aestivum) in untreated plants and plants treated with chemical hybridizing agent RH0007. Sex Plant Reprod 2:231–253
Murmu J, Bush MJ, DeLong C, Li S, Xu M, Khan M, Malcolmson C, Fobert PR, Zachgo S, Hepworth SR (2010) Arabidopsis basic leucine-zipper transcription factors TGA9 and TGA10 interact with floral glutaredoxins ROXY1 and ROXY2 and are redundantly required for anther development. Plant Physiol 154:1492–1504
Pacini E (2012) Pollen and seed analogies. Plant Biosyst 146:738–748
Parish RW, Phan HA, Iacuone S, Li SF (2012) Tapetal development and abiotic stress: a centre of vulnerability. Funct Plant Biol 39:553–559
Park PJ (2009) ChIP-seq: advantages and challenges of a maturing technology. Nat Rev Genet 10:669–680
Park JI, Hakozaki H, Endo M, Takada Y, Ito H, Uchida M, Okabe T, Watanabe M (2006) Molecular characterization of mature pollen-specific genes encoding novel small cysteine-rich proteins in rice (Oryza sativa L.). Plant Cell Rep 25:466–474
Phan HA, Iacuone S, Li SF, Parish RW (2011) The MYB80 transcription factor is required for pollen development and the regulation of tapetal programmed cell death in Arabidopsis thaliana. Plant Cell 23:2209–2224
Phan HA, Li SF, Parish RW (2012) MYB80, a regulator of tapetal and pollen development, is functionally conserved in crops. Plant Mol Biol 78:171–183
Pressman E, Shaked R, Shen S, Altahan L, Firon N (2012) Variations in carbohydrate content and sucrose-metabolizing enzymes in tomato (Solanum lycopersicum L.) stamen parts during pollen maturation. Amer J Plant Sci 3:252–260
Preston J, Wheeler J, Heazlewood J, Li SF, Parish RW (2004) AtMYB32 is required for normal pollen development in Arabidopsis thaliana. Plant J 40:979–995
Rouster J, Leah R, Mundy J, Cameron-Mills V (1997) Identification of a methyl jasmonate-responsive region in the promoter of a lipoxygenase 1 gene expressed in barley grain. Plant J 11:513–523
Šamaj J, Müller J, Beck M, Böhm N, Menzel D (2006) Vesicular trafficking, cytoskeleton and signalling in root hairs and pollen tubes. Trends Plant Sci 11:594–600
Song S, Qi T, Huang H, Xie D (2013) Coordinated actions of jasmonate, auxin, and gibberellin in Arabidopsis. Mol Plant 6:1065–1073
Swapna L, Khurana R, Vijaya Kumar S, Tyagi AK, Rao KV (2011) Pollen-specific expression of Oryza sativa Indica pollen allergen gene (OSIPA) promoter in rice and Arabidopsis transgenic systems. Mol Biotechnol 48:49–59
Tamagnone L, Merida A, Parr A, Mackay S, Culianez-Macia FA, Roberts K, Martin C (1998) The AmMYB308 and AmMYB330 transcription factors from Antirrhinum regulate phenylpropanoid and lignin biosynthesis in transgenic tobacco. Plant Cell 10:135–154
Thilmony R, Guttman ME, Lin JW, Blechl AE (2014) The wheat HMW-glutenin 1Dy10 gene promoter controls endosperm expression in Brachypodium distachyon. GM Crops Food Biotechnol Agric Food Chain 5:36–43
Thum KE, Kim M, Morishige DT, Eibl C, Koop H, Mullet JE (2001) Analysis of barley chloroplast psbD light-responsive promoter elements in transplastomic tobacco. Plant Mol Biol 47:353–366
Tran F, Penniket C, Patel RV, Provart NJ, Laroche A, Rowland O, Robert LS (2013) Developmental transcriptional profiling reveals key insights into Triticeae reproductive development. Plant J 74:971–988
Tsuchiya T, Toriyama K, Ejiri S, Hinata K (1994) Molecular characterization of rice genes specifically expressed in the anther tapetum. Plant Mol Biol 26:1737–1746
Vandepoele K, Vlieghe K, Florquin K, Hennig L, Beemster GTS, Gruissem W, Van de Peer Y, Inzé D, De Veylder L (2005) Genome-wide identification of potential plant E2F target genes. Plant Physiol 139:316–328
Vergne P, Delvallee I, Dumas C (1987) Rapid assessment of microspore and pollen development stage in wheat and maize using DAPI and membrane permeabilization. Stain Technol 62:299–304
Vogel J, Hill T (2008) High-efficiency Agrobacterium-mediated transformation of Brachypodium distachyon inbred line Bd21-3. Plant Cell Rep 27:471–478
Wang M, Hoekstra S, van Bergen S, Lamers GEM, Oppedijk BJ, van der Heijden MW, de Priester W, Schilperoort RA (1999) Apoptosis in developing anthers and the role of ABA in this process during androgenesis in Hordeum vulgare L. Plant Mol Biol 39:489–501
Wang C, Hsu S, Hsu Y (2013) New insights into dessication-associated gene regulation by Lilium longiflorum ASR during pollen maturation and in transgenic Arabidopsis. Int Rev Cell Mol Biol 301:37–94
Wu S, O’Leary SJB, Gleddie S, Eudes F, Laroche A, Robert LS (2008) A chalcone synthase-like gene is highly expressed in the tapetum of both wheat (Triticum aestivum L.) and triticale (x Triticosecale Wittmack). Plant Cell Rep 27:1441–1449
Yang S, Sweetman JP, Amirsadeghi S, Barghchi M, Huttly AK, Chung W, Twell D (2001) Novel anther-specific myb genes from tobacco as putative regulators of phenylalanine ammonia-lyase expression. Plant Physiol 126:1738–1753
Yokoi S, Tsuchiya T, Toriyama K, Hinata K (1997) Tapetum-specific expression of the Osg6B promoter-β-glucuronidase gene in transgenic rice. Plant Cell Rep 16:363–367
Zaidi MA, O’Leary S, Wu S, Gleddie S, Eudes F, Laroche A, Robert LS (2012) A molecular and proteomic investigation of proteins rapidly released from triticale pollen upon hydration. Plant Mol Biol 79:101–121
Zou J, Zhan X, Wu H, Wang H, Cheung AY (1994) Characterization of a rice pollen-specific gene and its expression. Amer J Bot 81:552–561
Acknowledgements
We wish to thank Dr. John Vogel (USDA-ARS, Albany, NY, USA) for pOL001. We also acknowledge the support of the AAFC Agricultural Bioproducts Innovation Program and AAFC/NSERC Visiting Fellowships program (MAZ, SJBO, and SW).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zaidi, M.A., O’Leary, S.J.B., Wu, S. et al. Investigating Triticeae anther gene promoter activity in transgenic Brachypodium distachyon . Planta 245, 385–396 (2017). https://doi.org/10.1007/s00425-016-2612-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-016-2612-5