Abstracts
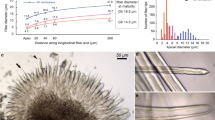
A unique fiber spinning was found in protoplasts from white birch (Betula platyphylla) leaves under an acidic medium containing high concentration of Ca2+. After expanding from 10 to 100 μm in diameter under the culture condition, the protoplast started secreting a gigantic fiber while moving in a spiral way. Real time video analyses elucidated that the orientation, rate and pattern of the motion were directed due to the inverse force of the fiber spinning. Moreover, observation using several microscopic methods accompanied with histochemical staining and nuclear magnetic resonance (NMR) analysis indicated that the fiber was composed of 400–500 nm wide (1→3)-β-glucan hollow sub-fibrils. This entire phenomenon may be a response against the stress imposed. The observation presented provides an understanding of the unique relationship between fiber spinning and the bottom-up fiber fabrication from nano to micro scales.











Similar content being viewed by others
Abbreviations
- NMR:
-
Nuclear magnetic resonance
- IBA:
-
Indol-3-butyric acid
- NAA:
-
α-Naphthaleneacetic acid
- FDA:
-
Fluorescein diacetate
References
Brown RM Jr, Willison JHM, Richardson CL (1976) Cellulose biosynthesis in Acetobacter xylinum: visualization of the site of synthesis and direct measurement of the in vivo process. Proc Nat Acad Sci USA 73(12):4565–4569
Colombani A, Djerbi S, Bessueille L, Blomqvist K, Ohlsson A, Berglund T, Teeri TT, Bulone V (2004) In vitro synthesis of (1→3)-β-D-glucan (callose) and cellulose by detergent extracts of membranes from cell suspension cultures of hybrid aspen. Cellulose 11:313–327
Deslandes Y, Marchessault RH, Sarko A (1980) Triple-Helical structure of(1→3)-β-D-Glucan. Macromolecules 13:1466–1471
Fink J, Jeblick W, Blaschek W, Kauss H (1987) Calcium ions and polyamines activate the plasma membrane-located 1,3-β-glucan synthase. Planta 171:130–135
Hanley SJ, Giasson J, Revol J-F, Gray DG (1992) Atomic force microscopy of cellulose microfibrils: comparison with transmission electron microscopy. Polymer 33:4639–4642
Him JLK, Pelosi L, Chanzy H, Putaux J-L, Bulone V (2001) Biosynthesis of (1→3)-β-D-glucan (callose) by detergent extracts of microsomal fraction from Arabidopsis thaliana. Eur J Biochem 268:4628–4638
Hirai N, Sonobe S, Hayashi T (1998) In situ synthesis of β-glucan microfibrils on tobacco plasma membrane sheets. Proc Natl Acad Sci USA 95:15102–15106
Kataoka Y, Kondo T (1996) Changing cellulose crystalline structure in forming wood cell walls. Macromolecules 29:6356–6358
Kataoka Y, Kondo T (1998) FT-IR microscopic analysis of changing cellulose crystalline structure during wood cell wall formation. Macromolecules 31:760–764
Kataoka Y, Kondo T (1999) Quantitative analysis for the cellulose Iα crystalline phase in developing wood cell walls. Int J Biol Macromol 24:37–41
Kauss H (1987) Some aspects of calcium-dependent regulation in plant metabolism. Annu Rev Plant Physiol 38:47–72
Kauss H (1996) Callose synthesis. In: Smallwood M, Knox JP, Bowles DJ (eds) Membranes: specialized functions in plants. BIOS Scientific Publishers, Oxford, pp 77–92
Kondo T, Nojiri M, Hishikawa Y, Togawa E, Romanovicz D, Brown RM Jr (2002) Biodirected epitaxial nanodeposition of polymers on oriented macromolecular templates. Proc Natl Acad Sci USA 99:14008–14013
Kondo T, Magoshi J, Abe H, Sasamoto H (2000) Method for producing of non cellulosic callose fibers from protoplasts and the callose fiber. Japan Patent No.3936522
Meikle PJ, Bonig I, Hoogenraad NJ, Clarke AE, Stone AB (1991) The location of (1→3)-β-glucans in the walls of pollen tubes of Nicotiana alata using a (1→3)-β-glucan-specific monoclonal antibody. Planta 185:1–8
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Pelosi L, Imai T, Chanzy H, Heux L, Buhler E, Bulone V (2003) Structural and Morphological diversity of (1→3)-D-Glucans synthesized in Vitro by enzymes from Saprolegnia monoïca. Comparison with a corresponding in vitro product from blackberry (Rubus fruticosus). Biochemistry 42:6264–6274
Wakita Y, Sasamoto H, Yokota S, Yoshizawa N (1996) Plantlet regeneration from mesophyll protoplasts of Betula platyphylla var. japonica. Plant Cell Reports 16:50–53
Waldmann T, Jeblick W, Kauss H (1988) Induced net Ca2+ uptake and callose biosynthesis in suspension-cultured plant cells. Planta 173:88–95
Zuiderweg ERP, Hallenga K, Olejniczak ET (1986) Improvement of 2D NOE spectra of biomacromolecules in H2O solution by coherent suppression of the solvent resonance. J Magn Reson 70:336–343
Acknowledgments
This research was supported partly by a Grant-in-Aid for Scientific Research (No. 14360101), Japan Society for the Promotion of Science (JSPS). The authors are grateful to Dr. Adya P. Singh at ENSIS (The Joint Forces of CSIRO & SION) for the editing of the text, and to Dr. Kazuaki Shikii for the NMR measurement (Nissan Chemical Industries, Ltd.). SEKI TECHNOTRON CORP. is also acknowledged by the assistance for observation using sectioning microscopy.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00425-008-0769-2
Rights and permissions
About this article
Cite this article
Seyama, T., Kimura, S., Sasamoto, H. et al. Spinning of a gigantic bundle of hollow fibrils by a spirally moving higher plant protoplast. Planta 227, 1187–1197 (2008). https://doi.org/10.1007/s00425-008-0689-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-008-0689-1