Abstract
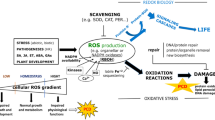
Recent studies have suggested that ultraviolet-C (UV-C) overexposure induces programmed cell death (PCD) in Arabidopsis thaliana (L.) Heynh, and this process includes participation of caspase-like proteases, DNA laddering as well as fragmentation of the nucleus. To investigate possible early signal events, we used microscopic observations to monitor in vivo the behaviour of mitochondria, as well as the production and localization of reactive oxygen species (ROS) during protoplast PCD induced by UV-C. A quick burst of ROS was detected when the protoplasts were kept in continuous light after UV-C exposure, which was restricted in chloroplasts and the adjacent mitochondria. Pre-incubation with ascorbic acid (AsA, antioxidant molecule) or 3-(3, 4-dichlorophenyl)-1, 1-dimethylurea (DCMU, an inhibitor of photosynthetic electron transport) decreased the ROS production and partially protected protoplasts from PCD. A mitochondrial transmembrane potential (MTP) loss occurred prior to cell death; thereafter, the mitochondria irregularly clumped around chloroplasts or aggregated in other places within the cytoplasm, and the movement of mitochondria was concomitantly blocked. Pre-treatment with an inhibitor of mitochondrial permeability transition pores (MPTP), cyclosporine (CsA), effectively retarded the decrease of MTP and reduced the percentage of protoplasts undergoing PCD after UV-C overexposure. Our results suggest that the MTP loss and the changes in distribution and mobility of mitochondria, as well as the production of ROS play important roles during UV-induced plant PCD, which is in good accordance with what has been reported in many types of apoptotic cell death, both in animals and plants.








Similar content being viewed by others
Abbreviations
- AsA:
-
Ascorbic acid
- CsA:
-
Cyclosporine A
- DCMU:
-
3-(3, 4-dichlorophenyl)-1, 1-dimethylurea
- FDA:
-
Fluorescein diacetate
- H2DCFDA:
-
2′, 7′-Dichlorodihydrofluorescein diacetate
- HR:
-
Hypersensitive response
- MPTP:
-
Mitochondrial permeability transitions pore
- MTP:
-
Mitochondrial transmembrane potential
- PCD:
-
Programmed cell death
- Rh123:
-
Rhodamine 123
- ROS:
-
Reactive oxygen species
References
Allan AC, Fluhr R (1997) Two distinct sources of elicited reactive oxygen species in tobacco epidermal cells. Plant Cell 9:1559–1572
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Asai T, Stone JM, Heard JE, Kovtun Y, Yorgey P, Sheen J, Ausubel FM (2000) Fumonisin B1-induced cell death in Arabidopsis protoplasts requires jasmonate-, ethylene-, and salicylate-dependent signaling pathways. Plant Cell 12:1823–1836
Babu TS, Akhtar TA, Lampi MA, Tripuranthakam S, Dixon DG, Greenberg BM (2003) Similar stress responses are elicited by copper and ultraviolet radiation in the aquatic plant Lemna gibba: implication of reactive oxygen species as common signals. Plant Cell Physiol 44:1320–1329
Bereiter-Hahn J, Vöth M (1994) Dynamics of mitochondria in living cells: shape changes, dislocations, fusion, and fission of mitochondria. Microsc Res Tech 27:198–219
Chandra-Shekara AC, Gupte M, Navarre D, Raina S, Raina R, Klessig D, Kachroo P (2006) Light-dependent hypersensitive response and resistance signaling against Turnip Crinkle Virus in Arabidopsis. Plant J 45:320–334
Chen SR, Dickman MB (2004) Bcl-2 family localize to tobacco chloroplasts and inhibit programmed cell death induced by chloroplast-targeted herbicides. J Exp Bot 55:2617–2623
Danon A, Gallois P (1998) UV-C radiation induces apoptotic-like changes in Arabidopsis thaliana. FEBS Lett 437:131–136
Danon A, Delorme V, Mailhac N, Gallois P (2000) Plant programmed cell death: a common way to die. Plant Physiol Biochem 38:647–655
Danon A, Rotari VI, Gordon A, Mailhac N, Gallois P (2004) Ultraviolet-C overexposure induces programmed cell death in Arabidopsis, which is mediated by caspase-like activities and which can be suppressed by caspase inhibitors, p35 and Defender against Apoptotic Death. J Biol Chem 279:779–787
Dat J, Vandenabeele S, Vranová E, Van Montagu M, Inzé D, Van Breusegem F (2000) Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci 57:779–795
Dutilleul C, Garmier M, Noctor G, Mathieu C, Chétrit P, Foyer CH, Paepe DR (2003) Leaf mitochondria modulate whole cell redox homeostasis, set antioxidant capacity, and determine stress resistance through altered signaling and diurnal regulation. Plant Cell 15:1212–1226
Edreva A (2005) Generation and scavenging of reactive oxygen species in chloroplasts: a submolecular approach. Agric Ecosyst Environ 106:119–133
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875
Foyer CH, Lelandais M, Kunert KJ (1994) Photooxidative stress in plants. Physiol Plant 92:696–717
Gestel KV, Köhler RH, Verbelen JP (2002) Plant mitochondria move on F-actin, but their positioning in the cortical cytoplasm depends on both F-actin and microtubules. J Exp Bot 53:659–667
Hauser BA, Sun K, Oppenheimer DG, Sage TL (2006) Changes in mitochondrial membrane potential and accumulation of reactive oxygen species precede ultrastructural changes during ovule abortion. Planta 223:492–499
He YY, Häder DP (2002) Involvement of reactive oxygen species in the UV-B damage to the cyanobacterium Anabaena sp. J Photochem Photobiol B Biol 66:73–80
He P, Shan L, Sheen J (2006) The use of protoplasts to study innate immune responses. Methods Mol Biol 354:1–10
Jakubowski W, Bartosz G (2000) 2, 7-dichlorofluorescein oxidation and reactive oxygen species: what does it measure? Cell Biol Int 24:757–760
Karpinski S, Gabrys H, Mateo A, Karpinska B, Mullineaux PM (2003) Light perception in plant disease defence signalling. Curr Opin Plant Biol 6:390–396
Kikuyama M, Tazawa M (1982) Ca2+ ion reversibly inhibits the cytoplasmic streaming of Nitella. Protoplasma 113:241–243
Kluk RM, Bossy-Wetzel E, Green DR, Newmeyer DD (1997) The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275:1132–1136
Kroemer G (1999) Mitochondrial control of apoptosis: an overview. Biochem Soc Symp 66:1–15
Kroemer G, Dallaporta B, Resche-Rigon M (1998) The mitochondrial death/life regulation in apoptosis and necrosis. Annu Rev Physiol 60:619–642
Kulms D, Schwarz T (2002) Molecular mechanisms involved in UV-induced apoptotic cell death. Skin Pharmacol Appl Skin Physiol 15:342–347
Levine A, Tenhaken R, Dixon RA, Lamb C (1994) H2O2 from the oxidative burst orchestrates the plant hypersensitive disease resistance response. Cell 79:583–593
Logan DC (2006) Plant mitochondrial dynamics. Biochim Biophys Acta 1763:430–441
Logan DC, Leaver CJ (2000) Mitochondria-targeted GFP highlights the heterogeneity of mitochondrial shape, size and movement within living plant cells. J Exp Bot 51:865–871
Navrot N, Rouhier N, Gelhaye E, Jacquot JP (2007) Reactive oxygen species generation and antioxidant systems in plant mitochondria. Physiol Plant 129:185–195
Orrenius S, Gogvadze V, Zhivotovsky B (2007) Mitochondrial oxidative stress: implications for cell death. Annu Rev Pharmacol Toxicol 47:143–183
Pontier D, Balague C, Roby D (1998) The hypersensitive response: a programmed cell death associated with plant resistance. C R Acad Sci III 321:721–734
Roger RI, Lamb C (1997) Programmed cell death in plants. Plant Cell 9:1157–1168
Sakamoto M, Tada Y, Nakayashiki H, Tosa Y, Mayama S (2005) Two phases of intracellular reactive oxygen species production during victorin-induced cell death in oats. J Gen Plant Pathol 71:387–394
Samadi L, Shahsavan Behboodi B (2006) Fusaric acid induces apoptosis in saffron root-tip cells: roles of caspase-like activity, cytochrome c, and H2O2. Planta 225:223–234
Samuilov VD, Lagunova EM, Kiselevsky DB, Dzyubinskaya EV, Makarova YV, Gusev MV (2003) Participation of chloroplasts in plant apoptosis. Biosci Rep 23:103–117
Sazanov LA, Burrows PA, Nixon PJ (1998) The plastid ndh genes code for a NAD-specific dehydrogenase: purification and characterization of a mitochondrial-like complex I from pea thylakoid membranes. Proc Natl Acad Sci USA 95:1319–1324
Simon HU, Haj-Yehia A, Levi-Schaffer F (2000) Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 5:415–418
Sinha RP, Häder DP (2002) UV-induced DNA damage and repair: a review. Photochem Photobiol Sci 1:225–236
Vacca RA, Valenti D, Bobba A, Merafina RS, Passarella S, Marra E (2006) Cytochrome c is released in a reactive oxygen species-dependent manner and is degraded via caspase-like proteases in tobacco Bright-Yellow 2 cells en route to heat shock-induced cell death. Plant Physiol 141:208–219
Van Breusegem F, Dat JF (2006) Reactive oxygen species in plant cell death. Plant Physiol 141:384–390
van Doorn WG, Woltering EJ (2005) Many ways to exit? Cell death categories in plants. Trends Plant Sci 10:117–122
Wang X (2001) The expanding role of mitochondria in apoptosis. Genes Dev 15:2922–2933
Wu YY, Xing D, Liu L, Chen TS, Chen WR (2007) Fluorescence resonance energy transfer analysis of bid activation in living cells during Ultraviolet-induced apoptosis. Acta Biochim Biophys Sin 39:37–45
Yamamoto Y, Kobayashi Y, Devi SR, Rikiishi S, Matsumoto H (2002) Aluminum toxicity is associated with mitochondrial dysfunction and the production of reactive oxygen species in plant cells. Plant Physiol 128:63–72
Yao N, Greenberg JT (2006) Arabidopsis ACCELERATED CELL DEATH2 modulates programmed cell death. Plant Cell 18:397–411
Yao N, Eisfelder BJ, Marvin J, Greenberg JT (2004) The mitochondrion––an organelle commonly involved in programmed cell death in Arabidopsis thaliana. Plant J 40:596–610
Yoshinaga K, Arimura SI, Niwa Y, Tsutsumi N, Uchimiya H, Kawai-Yamada M (2005) Mitochondrial behaviour in the early stages of ROS stress leading to cell death in Arabidopsis thaliana. Ann Bot 96:337–342
Zapata JM, Guéra A, Esteban-Carrasco A, Martín M, Sabater B (2005) Chloroplasts regulate leaf senescence: delayed senescence in transgenic ndhF-defective tobacco. Cell Death Differ 12:1277–1284
Acknowledgments
We are grateful to Dr. David C. Logan (St Andrews, UK) for kindly providing transgenic Arabidopsis seeds harboring GFP-labeled mitochondria. We thank Yonghong Tang for her help in using laser scanning confocal microscope in our research group, anonymous reviewers for their constructive comments, Prof. Wei R. Chen for critical reading of the manuscript, and members of our group for helpful discussions. This research is supported by the National Natural Science Foundation of China (30670507; 30600128; 30470494) and the Natural Science Foundation of Guangdong Province (015012).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary movie (AVI 2280 kb)
Rights and permissions
About this article
Cite this article
Gao, C., Xing, D., Li, L. et al. Implication of reactive oxygen species and mitochondrial dysfunction in the early stages of plant programmed cell death induced by ultraviolet-C overexposure. Planta 227, 755–767 (2008). https://doi.org/10.1007/s00425-007-0654-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-007-0654-4