Abstract
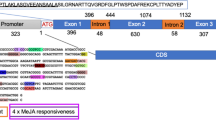
Chitinases are ubiquitous proteins that occur in all plants in multiple isoforms. We have isolated the ChtC2 gene encoding an unusual, basic (class I) chitinase from potato (Solanum tuberosum L.). In contrast to other chitinase genes, ChtC2 is not activated by infection, but rather constitutively expressed in leaves and stems where it is restricted to epidermal cells. Sequence analysis revealed a number of potential regulatory elements in the promoter, but most striking was the presence of a 319-bp direct repeat located between −333 and −968 upstream of the transcription start site. For a functional analysis, a 1,322-bp promoter fragment and two 5′ deletions of 782 bp and 162 bp in length were translationally fused to the β-glucuronidase (GUS) reporter gene and used for transient expression studies by particle bombardment. All promoter constructs conferred expression of GUS activity in different epidermal cell types of potato leaves. Expression in parenchyma cells of the leaf mesophyll was not detectable with any of the ChtC2 gene promoter constructs, in contrast to the pattern observed with the 35S promoter from cauliflower mosaic virus. The epidermis-specific expression of the reporter gene was confirmed using transgenic potato plants containing the fusion of the entire ChtC2 promoter with the GUS reporter. Histochemical analysis indicated that the promoter was only active in epidermal cells of leaves.







Similar content being viewed by others
Abbreviations
- ChtC:
-
chitinase C
- GUS:
-
β-glucuronidase
- RACE:
-
rapid amplification of cDNA ends
- X-gluc:
-
5-bromo-4-chloro-3-indolyl-β-d-glucuronide
References
Ancillo G, Witte B, Schmelzer E, Kombrink E (1999) A distinct member of the basic (class I) chitinase gene family in potato is specifically expressed in epidermal cells. Plant Mol Biol 39:1137–1151
Antikainen M, Griffith M, Zhang J, Hon W-C, Yang DSC, Pihakaski-Maunsbach K (1996) Immunolocalization of antifreeze proteins in winter rye leaves, crowns, and roots by tissue printing. Plant Physiol 110:845–857
Baureithel K, Felix G, Boller T (1994) Specific, high affinity binding of chitin fragments to tomato cells and membranes. J Biol Chem 269:17931–17938
Becker-André M, Schulze-Lefert P, Hahlbrock K (1991) Structural comparison, modes of expression, and putative cis-acting elements of the two 4-coumarate:CoA ligase genes in potato. J Biol Chem 266:8551–8559
Beerhues L, Kombrink E (1994) Primary structure and expression of mRNAs encoding basic chitinase and 1,3-β-glucanase in potato. Plant Mol Biol 24:353–367
Büchter R, Strömberg A, Schmelzer E, Kombrink E (1997) Primary structure and expression of acidic (class II) chitinase in potato. Plant Mol Biol 35:749–761
Chen EY, Seeburg PH (1985) Supercoil sequencing: a fast and simple method for sequencing plasmid DNA. DNA 4:165–170
de Jong AJ, Cordewener J, Lo Schiavo F, Terzi M, Vandekerckhove J, van Kammen A, de Vries SC (1992) A carrot somatic embryo mutant is rescued by chitinase. Plant Cell 4:425–433
de Jong AJ, Heidstra R, Spaink HP, Hartog MV, Meijer EA, Hendriks T, Lo Schiava F, Terzi M, Bisseling T, van Kammen A, de Vries SC (1993) Rhizobium lipooligosaccharides rescue a carrot somatic embryo mutant. Plant Cell 5:615–620
Devereux J, Haeberli P, Smithies O (1984) A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res 12:387–395
Ebel J, Scheel D (1997) Signals in host-parasite interactions. In: Carroll G, Tudzynski P (eds) The Mycota, vol 5, part A, Plant relationships. Springer, Berlin Heidelberg New York, pp 85–105
Felix G, Regenass M, Boller T (1993) Specific perception of subnanomolar concentrations of chitin fragments by tomato cells: induction of extracellular alkalinization, changes in protein phosphorylation, and establishment of a refractory state. Plant J 4:307–316
Foster R, Izawa T, Chua N-H (1994) Plant bZIP proteins gather at ACGT elements. FASEB J 8:192–200
Garcia-Garcia F, Schmelzer E, Hahlbrock K, Roxby R (1994) Differential expression of chitinase and β-1,3-glucanase genes in various tissues of potato plants. Z Naturforsch C 49:195–203
Hart CM, Nagy F, Meins F Jr (1993) A 61 bp enhancer element of the tobacco β-1,3-glucanase B gene interacts with one or more regulated nuclear proteins. Plant Mol Biol 21:121–131
Hon W-C, Griffith M, Mlynarz A, Kwok YC, Yang DSC (1995) Antifreeze proteins in winter rye are similar to pathogenesis-related proteins. Plant Physiol 109:879–889
Hu W-J, Kawaoka A, Tsai C-J, Lung J, Osakabe K, Ebinuma H, Chiang VL (1998) Compartmentalized expression of two structurally and functionally distinct 4-coumarate:CoA ligase genes in aspen (Populus tremuloides). Proc Natl Acad Sci USA 95:5407–5412
Huang N, Sutliff TD, Litts JC, Rodriguez RL (1990) Classification and characterization of the rice α-amylase multigene family. Plant Mol Biol 14:655–668
Hunhold R, Bronner R, Hahne G (1994) Early events in microprojectile bombardment: cell viability and particle location. Plant J 5:593–604
Izawa T, Foster R, Chua N-H (1993) Plant bZIP protein DNA binding specificity. J Mol Biol 230:1131–1144
Jach G, Görnhardt B, Mundy J, Logemann J, Pinsdorf E, Leah R, Schell J, Maas C (1995) Enhanced quantitative resistance against fungal disease by combinatorial expression of different barley antifungal proteins in transgenic tobacco. Plant J 8:97–109
Jongedijk E, Tigelaar H, van Roekel JSC, Bres-Vloemans SA, Dekker I, van den Elzen PJM, Cornelissen BJC, Melchers LS (1995) Synergistic activity of chitinases and β-1,3-glucanases enhances fungal resistance in transgenic tomato plants. Euphytica 85:173–180
Keefe D, Hinz U, Meins F Jr (1990) The effect of ethylene on the cell-type-specific and intracellular localization of β-1,3-glucanase and chitinase in tobacco leaves. Planta 182:43–51
Kombrink E, Somssich IE (1997) Pathogenesis-related proteins and plant defense. In: Carroll G, Tudzynski P (eds) The Mycota, vol 5, part A, Plant relationships. Springer, Berlin Heidelberg New York, pp 107–128
Kombrink E, Schröder M, Hahlbrock K (1988) Several "pathogenesis-related" proteins in potato are 1,3-β-glucanases and chitinases. Proc Natl Acad Sci USA 85:782–786
Kombrink E, Beerhues L, Garcia-Garcia F, Hahlbrock K, Müller M, Schröder M, Witte B, Schmelzer E (1993) Expression patterns of defense-related genes in infected and uninfected plants. In: Fritig B, Legrand M (eds) Mechanisms of plant defense responses, vol 2. Kluwer, Dordrecht, pp 236–249
Koncz C, Schell J (1986) The promoter of TL-DNA gene 5 controls the tissue-specific expression of chimaeric genes carried by a novel type of Agrobacterium binary vector. Mol Gen Genet 204:383–396
Kragh KM, Hendriks T, de Jong AJ, Lo Schiavo F, Bucherna N, Højrup P, Mikkelsen JD, de Vries SC (1996) Characterization of chitinases able to rescue somatic embryos of the temperature-sensitive carrot variant ts11. Plant Mol Biol 31:631–645
Leubner-Metzger G, Meins F Jr (1999) Functions and regulation of plant β-1,3-glucanases (PR-2). In: Datta SK, Muthukrishnan S (eds) Pathogenesis-related proteins in plants. CRC Press, Boca Raton, pp 49–76
Mauch F, Mauch-Mani B, Boller T (1988) Antifungal hydrolases in pea tissue. II. Inhibition of fungal growth by combinations of chitinase and 1,3-β-glucanase. Plant Physiol 88:936–942
Mauch F, Meehl JB, Staehelin LA (1992) Ethylene-induced chitinase and β-1,3-glucanase accumulate specifically in the lower epidermis and along vascular strands of bean leaves. Planta 186:367–375
Neuhaus J-M (1999) Plant chitinases (PR-3, PR-4, PR-8, PR-11). In: Datta SK, Muthukrishnan S (eds) Pathogenesis-related proteins in plants. CRC Press, Boca Raton, pp 77–105
Neuhaus J-M, Ahl-Goy P, Hinz U, Flores S, Meins F Jr (1991) High-level expression of a tobacco chitinase gene in Nicotiana sylvestris. Susceptibility of transgenic plants to Cercospora nicotianae infection. Plant Mol Biol 16:141–151
Nielsen KK, Mikkelsen JD, Kragh KM, Bojsen K (1993) An acidic class III chitinase in sugar beet: induction by Cercospora beticola, characterization, and expression in transgenic tobacco plants. Mol Plant Microbe Interact 6:495–506
Ponath Y, Vollberg H, Hahlbrock K, Kombrink E (2000) Two differentially regulated class II chitinases from parsley. Biol Chem 381:667–678
Ren Y-Y, West CA (1992) Elicitation of diterpene biosynthesis in rice (Oryza sativa L.) by chitin. Plant Physiol 99:1169–1178
Reuber S, Bornmann JF, Weissenböck G (1996) Phenylpropanoid compounds in primary leaf tissues of rye (Secale cereale). Light response of their metabolism and the possible role in UV-B protection. Physiol Plant 97:160–168
Roby D, Gadelle A, Toppan A (1987) Chitin oligosaccharides as elicitors of chitinase activity in melon plants. Biochem Biophys Res Commun 143:885–892
Rushton PJ, Somssich IE (1999) Transcriptional regulation of plant genes responsive to pathogens and elicitors. In: Stacey G, Keen NT (eds) Plant–microbe interactions, vol 4. APS Press, St. Paul, MN, pp 251–274
Rushton PJ, Tovar Torres J, Parniske M, Wernert P, Hahlbrock K, Somssich IE (1996) Interaction of elicitor-induced DNA-binding proteins with elicitor response elements in the promoters of parsley PR1 genes. EMBO J 15:5690–5700
Samac DA, Shah DM (1991) Developmental and pathogen-induced activation of the Arabidopsis acidic chitinase promoter. Plant Cell 3:1063–1072
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning. A laboratory manual. Cold Spring Harbor Laboratory Press, Plainview, NY
Schmelzer E, Jahnen W, Hahlbrock K (1988) In situ localization of light-induced chalcone synthase mRNA, chalcone synthase, and flavonoid end products in epidermal cells of parsley leaves. Proc Natl Acad Sci USA 85:2989–2993
Schmid J, Doerner PW, Clouse SD, Dixon RA, Lamb CJ (1990) Developmental and environmental regulation of a bean chalcone synthase promoter in transgenic tobacco. Plant Cell 2:619–631
Sela-Buurlage MB, Ponstein AS, Bres-Vloemans SA, Melchers LS, van den Elzen PJM, Cornelissen BJC (1993) Only specific tobacco (Nicotiana tabacum) chitinases and β-1,3-glucanases exhibit antifungal activity. Plant Physiol 101:857–863
Sessions A, Weigel D, Yanofsky MF (1999) The Arabidopsis thaliana MERISTEM LAYER 1 promoter specifies epidermal expression in meristems and young primordia. Plant J 20:259–263
Shirsat A, Wilford N, Croy R, Boulter D (1989) Sequences responsible for the tissue specific promoter activity of a pea legumin gene in tobacco. Mol Gen Genet 215:326–331
Sokal R, Rohlf FJ (1969) Biometry. Freeman, San Francisco
van de Löcht U, Meier I, Hahlbrock K, Somssich IE (1990) A 125 bp promoter fragment is sufficient for strong elicitor-mediated gene activation in parsley. EMBO J 9:2945–2950
Wemmer T, Kaufmann H, Kirch H-H, Schneider K, Lottspeich F, Thompson RD (1994) The most abundant soluble basic protein of the stylar transmitting tract in potato (Solanum tuberosum L.) is an endochitinase. Planta 194:264–273
Wu S-C, Hahlbrock K (1992) In situ localization of phenylpropanoid-related gene expression in different tissues of light- and dark-grown parsley seedlings. Z Naturforsch C 47: 591–600
Xia Y, Nikolau BJ, Schnable PS (1996) Cloning and characterization of CER2, an Arabidopsis gene that affects cuticular wax accumulation. Plant Cell 8:1291–1304
Zhu Q, Doerner PW, Lamb CJ (1993) Stress induction and developmental regulation of a rice chitinase promoter in transgenic tobacco. Plant J 3:203–212
Zhu Y, Maher EA, Masoud S, Dixon RA, Lamb CJ (1994) Enhanced protection against fungal attack by constitutive co-expression of chitinase and glucanase genes in transgenic tobacco. Bio/Technology 12:807–812
Acknowledgements
We thank Dr. S. Elena (University of Valencia, Spain) for his help and advice on the statistical analysis of the bombardment data, Drs. I.E. Somssich, J.L. Carrasco for valuable discussions and critical reading of the manuscript and Professor K. Hahlbrock for continuous support of this work. G.A. was the recipient of a postdoctoral fellowship of the Instituto Nacional de Investigaciones Agrarias of the Spanish Ministry of Agriculture. The GenBank accession number of the sequence reported in this paper is AF153195 (ChtC gene).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ancillo, G., Hoegen, E. & Kombrink, E. The promoter of the potato chitinase C gene directs expression to epidermal cells. Planta 217, 566–576 (2003). https://doi.org/10.1007/s00425-003-1029-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-003-1029-0