Abstract
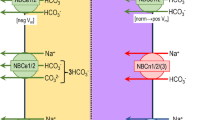
The expression cloning some 25 years ago of the first member of SLC34 solute carrier family, the renal sodium-coupled inorganic phosphate cotransporter (NaPi-IIa) from rat and human tissue, heralded a new era of research into renal phosphate handling by focussing on the carrier proteins that mediate phosphate transport. The cloning of NaPi-IIa was followed by that of the intestinal NaPi-IIb and renal NaPi-IIc isoforms. These three proteins constitute the main secondary-active Na+-driven pathways for apical entry of inorganic phosphate (Pi) across renal and intestinal epithelial, as well as other epithelial-like organs. The key role these proteins play in mammalian Pi homeostasis was revealed in the intervening decades by numerous in vitro and animal studies, including the development of knockout animals for each gene and the detection of naturally occurring mutations that can lead to Pi-handling dysfunction in humans. In addition to characterising their physiological regulation, research has also focused on understanding the underlying transport mechanism and identifying structure-function relationships. Over the past two decades, this research effort has used real-time electrophysiological and fluorometric assays together with novel computational biology strategies to develop a detailed, but still incomplete, understanding of the transport mechanism of SLC34 proteins at the molecular level. This review will focus on how our present understanding of their molecular mechanism has evolved in this period by highlighting the key experimental findings.








Similar content being viewed by others
Notes
Recently, a naturally occurring mutation in human NaPi-IIa has been reported, involving an arginine to glutamine substitution at site 215 (see Fig 7b) [22]. Preliminary kinetic characterisation of this mutation in Xenopus oocytes revealed a significant hyperpolarizing shift in the steady-state voltage dependence. Qualitatively similar changes in IPi vs V have been reported for substitutions at sites where the native residue is uncharged (e.g. [29, 44, 110]), which indicated that the site of substitution as well as the charge can influence the voltage dependence.
The participation of Li+ ions in the NaPi-IIa cotransport cycle may explain the in vivo finding that Li+ ions are indeed reabsorbed in the rat kidney, most likely via NaPi-IIa. This could have clinical relevance for bipolar disorder treatment [102].
This is also underscored by the different voltage dependences displayed by SLC34 isoforms. For example, for the range of hyperpolarizing voltages amenable to oocyte assays, the mouse NaPi-IIb does not show obvious rate limiting behaviour unlike the flounder NaPi-IIb and NaPi-IIa isoforms so far examined (Fig 3b) and the postulated interaction of protons with the final Na+ interaction may therefore not play a significant role in affecting the overall cotransport rate.
Ideally, one should substitute novel cysteines in a so-called Cys-less background; however, removal of native cysteines often compromises functional activity as in the case of NaPi-IIa [60]. For all isoforms examined, it was confirmed experimentally that external application of the MTS reagents had insignificant effect on the wild-type (WT) activity and for NaPi-IIb internal exposure to MTS reagents by means of internal perfusion of the oocyte cytosol also resulted in unaltered transport function (unpublished experiment, K Köhler, I Gautschi, IC Forster). Thus, despite there being 12 or more native cysteines in the SLC34 family (two of which form a disulphide bond), it was most likely that these were not located in functionally important sites or were simply not accessible.
Uptake assays of bacterial members of the DASS family (e.g. [50]) indicated that transport was mediated by 2 Na+ ions but only one of these was resolved [77]. Recently, electrogenic behaviour has been demonstrated for VcINDY and its true stoichiometry found to be 3:1 [31, 81], the same as that of the eukaryotic protein NaDC1 [86, 40] and NaPi-IIa/b.
References
Abramson J, Smirnova I, Kasho V, Verner G, Iwata S, Kaback HR (2003) The lactose permease of Escherichia coli: overall structure, the sugar-binding site and the alternating access model for transport. FEBS Lett 555:96–101
Andrini O, Ghezzi C, Murer H, Forster IC (2008) The leak mode of type II Na(+)-P(i) cotransporters. Channels (Austin) 2:346–357
Andrini O, Meinild AK, Ghezzi C, Murer H, Forster IC (2012) Lithium interactions with Na+−coupled inorganic phosphate cotransporters: insights into the mechanism of sequential cation binding. Am J Phys Cell Physiol 302:C539–C554
Armstrong CM, Bezanilla F (1974) Charge movement associated with the opening and closing of the activation gates of the Na channels. J Gen Physiol 63:533–552
Bacconi A, Ravera S, Virkki LV, Murer H, Forster IC (2007) Temperature dependence of steady-state and presteady-state kinetics of a type IIb Na+/P i cotransporter. J Membr Biol 215:81–92. https://doi.org/10.1007/s00232-007-9008-1
Bacconi A, Virkki LV, Biber J, Murer H, Forster IC (2005) Renouncing electrogenicity is not free of charge: switching on electrogenicity in a Na+-coupled phosphate cotransporter. Proc Natl Acad Sci U S A 102:12606–12611
Bazzone A, Barthmes M, Fendler K (2017) SSM-based electrophysiology for transporter research. Methods Enzymol 594:31–83. https://doi.org/10.1016/bs.mie.2017.05.008
Bezanilla F (2008) How membrane proteins sense voltage. Nat Rev Mol Cell Biol 9:323–332
Bezanilla F (2018) Gating currents. J Gen Physiol 150:911–932. https://doi.org/10.1085/jgp.201812090
Biber J, Custer M, Magagnin S, Hayes G, Werner A, Lotscher M, Kaissling B, Murer H (1996) Renal Na/Pi-cotransporters. Kidney Int 49:981–985
Biber J, Hernando N, Forster I (2013) Phosphate transporters and their function. Annu Rev Physiol 75:535–550. https://doi.org/10.1146/annurev-physiol-030212-183748
Boyer CJ, Xiao Y, Dugre A, Vincent E, Delisle MC, Beliveau R (1996) Phosphate deprivation induces overexpression of two proteins related to the rat renal phosphate cotransporter NaPi-2. Biochim Biophys Acta 1281:117–123
Burckhardt G, Stern H, Murer H (1981) The influence of pH on phosphate transport into rat renal brush border membrane vesicles. Pflugers Arch 390:191–197
Busch A, Waldegger S, Herzer T, Biber J, Markovich D, Hayes G, Murer H, Lang F (1994) Electrophysiological analysis of Na+/Pi cotransport mediated by a transporter cloned from rat kidney and expressed in Xenopus oocytes. Proc Natl Acad Sci U S A 91:8205–8208
Busch AE, Wagner CA, Schuster A, Waldegger S, Biber J, Murer H, Lang F (1995) Properties of electrogenic Pi transport by a human renal brush border Na+/Pi transporter. J Am Soc Nephrol 6:1547–1551
Cha A, Bezanilla F (1998) Structural implications of fluorescence quenching in the Shaker K+ channel. J Gen Physiol 112:391–408
Cha A, Zerangue N, Kavanaugh M, Bezanilla F (1998) Fluorescence techniques for studying cloned channels and transporters expressed in Xenopus oocytes. Methods Enzymol 296:566–578
Chen XZ, Coady MJ, Jackson F, Berteloot A, Lapointe JY (1995) Thermodynamic determination of the Na+: glucose coupling ratio for the human SGLT1 cotransporter. Biophys J 69:2405–2414. https://doi.org/10.1016/S0006-3495(95)80110-4
Cheng L, Sacktor B (1981) Sodium gradient-dependent phosphate transport in renal brush border membrane vesicles. J Biol Chem 256:1556–1564
de la Horra C, Hernando N, Forster I, Biber J, Murer H (2001) Amino acids involved in sodium interaction of murine type II Na+-Pi cotransporters expressed in Xenopus oocytes. J Physiol 531:383–391
de la Horra C, Hernando N, Lambert G, Forster I, Biber J, Murer H (2000) Molecular determinants of pH sensitivity of the type IIa Na/Pi cotransporter. J Biol Chem 275:6284–6287
Dinour D, Davidovits M, Ganon L, Ruminska J, Forster IC, Hernando N, Eyal E, Holtzman EJ, Wagner CA (2016) Loss of function of NaPiIIa causes nephrocalcinosis and possibly kidney insufficiency. Pediatr Nephrol 31:2289–2297. https://doi.org/10.1007/s00467-016-3443-0
Drew D, Boudker O (2016) Shared molecular mechanisms of membrane transporters. Annu Rev Biochem 85:543–572. https://doi.org/10.1146/annurev-biochem-060815-014520
Ehnes C, Forster IC, Bacconi A, Kohler K, Biber J, Murer H (2004) Structure-function relations of the first and fourth extracellular linkers of the type IIa Na+/Pi cotransporter: II. Substrate interaction and voltage dependency of two functionally important sites. J Gen Physiol 124:489–503
Ehnes C, Forster IC, Kohler K, Bacconi A, Stange G, Biber J, Murer H (2004) Structure-function relations of the first and fourth predicted extracellular linkers of the type IIa Na+/Pi cotransporter: I. Cysteine scanning mutagenesis. J Gen Physiol 124:475–488
Ehnes C, Forster IC, Kohler K, Biber J, Murer H (2002) Functional studies on a split type II Na/Pi-cotransporter. J Membr Biol 188:227–236
Eskandari S (2009) Remarkable commonalities of electrogenic and electroneutral Na+-phosphate cotransporters. J Physiol 587:4131–4132. https://doi.org/10.1113/jphysiol.2009.179119
Faham S, Watanabe A, Besserer GM, Cascio D, Specht A, Hirayama BA, Wright EM, Abramson J (2008) The crystal structure of a sodium galactose transporter reveals mechanistic insights into Na+/sugar symport. Science 321:810–814
Fenollar-Ferrer C, Forster IC, Patti M, Knoepfel T, Werner A, Forrest LR (2015) Identification of the first sodium binding site of the phosphate cotransporter NaPi-IIa (SLC34A1). Biophys J 108:2465–2480. https://doi.org/10.1016/j.bpj.2015.03.054
Fenollar-Ferrer C, Patti M, Knopfel T, Werner A, Forster IC, Forrest LR (2014) Structural fold and binding sites of the human Na(+)-phosphate cotransporter NaPi-II. Biophys J 106:1268–1279. https://doi.org/10.1016/j.bpj.2014.01.043
Fitzgerald GA, Mulligan C, Mindell JA (2017) A general method for determining secondary active transporter substrate stoichiometry. Elife 6. https://doi.org/10.7554/eLife.21016
Forster I, Biber J, Murer H (1999) Electrophysiological analysis of renal Na+-coupled divalent anion transporters. Pharm Biotechnol 12:251–267
Forster I, Hernando N, Biber J, Murer H (1998) The voltage dependence of a cloned mammalian renal type II Na+/Pi cotransporter (NaPi-2). J Gen Physiol 112:1–18
Forster IC, Biber J, Murer H (2000) Proton-sensitive transitions of renal type II Na(+)-coupled phosphate cotransporter kinetics. Biophys J 79:215–230. https://doi.org/10.1016/S0006-3495(00)76285-0
Forster IC, Hernando N, Biber J, Murer H (2006) Proximal tubular handling of phosphate: a molecular perspective. Kidney Int 70:1548–1559
Forster IC, Hernando N, Biber J, Murer H (2012) Phosphate transport kinetics and structure-function relationships of SLC34 and SLC20 proteins. Curr Top Membr 70:313–356
Forster IC, Hernando N, Biber J, Murer H (2013) Phosphate transporters of the SLC20 and SLC34 families. Mol Asp Med 34:386–395
Forster IC, Kohler K, Biber J, Murer H (2002) Forging the link between structure and function of electrogenic cotransporters: the renal type IIa Na+/Pi cotransporter as a case study. Prog Biophys Mol Biol 80:69–108
Forster IC, Kohler K, Stange G, Biber J, Murer H (2002) Modulation of renal type IIa Na+/Pi cotransporter kinetics by the arginine modifier phenylglyoxal. J Membr Biol 187:85–96
Forster IC, Loo DD, Eskandari S (1999) Stoichiometry and Na+ binding cooperativity of rat and flounder renal type II Na+-Pi cotransporters. Am J Phys 276:F644–F649
Forster IC, Virkki LV, Bossi E, Murer H, Biber J (2006) Electrogenic kinetics of a mammalian intestinal Na+/Pi-cotransporter. J Membr Biol 212:177–190
Forster IC, Wagner CA, Busch AE, Lang F, Biber J, Hernando N, Murer H, Werner A (1997) Electrophysiological characterization of the flounder type II Na+/Pi cotransporter (NaPi-5) expressed in Xenopus laevis oocytes. J Membr Biol 160:9–25
Fromter E (1979) The Feldberg lecture 1976. Solute transport across epithelia: what can we learn from micropuncture studies in kidney tubules? J Physiol 288:1–31
Ghezzi C, Meinild AK, Murer H, Forster IC (2011) Voltage- and substrate-dependent interactions between sites in putative re-entrant domains of a Na(+)-coupled phosphate cotransporter. Pflugers Arch - Eur J Physiol 461:645–663. https://doi.org/10.1007/s00424-011-0948-z
Ghezzi C, Murer H, Forster IC (2009) Substrate interactions of the electroneutral Na+−coupled inorganic phosphate cotransporter (NaPi-IIc). J Physiol 587:4293–4307. https://doi.org/10.1113/jphysiol.2009.175596
Gisler SM, Kittanakom S, Fuster D, Wong V, Bertic M, Radanovic T, Hall RA, Murer H, Biber J, Markovich D, Moe OW, Stagljar I (2008) Monitoring protein-protein interactions between the mammalian integral membrane transporters and PDZ-interacting partners using a modified split-ubiquitin membrane yeast two-hybrid system. Mol Cell Proteomics 7:1362–1377. https://doi.org/10.1074/mcp.M800079-MCP200
Gonzales AL, Lee W, Spencer SR, Oropeza RA, Chapman JV, Ku JY, Eskandari S (2007) Turnover rate of the gamma-aminobutyric acid transporter GAT1. J Membr Biol 220:33–51. https://doi.org/10.1007/s00232-007-9073-5
Graham C, Nalbant P, Scholermann B, Hentschel H, Kinne RK, Werner A (2003) Characterization of a type IIb sodium-phosphate cotransporter from zebrafish (Danio rerio) kidney. Am J Physiol Ren Physiol 284:F727–F736
Grewer C (2014) Shedding light on conformational dynamics of na(+)-coupled transporters. Biophys J 106:1549–1550. https://doi.org/10.1016/j.bpj.2014.02.029
Hall JA, Pajor AM (2005) Functional characterization of a Na(+)-coupled dicarboxylate carrier protein from Staphylococcus aureus. J Bacteriol 187:5189–5194. https://doi.org/10.1128/JB.187.15.5189-5194.2005
Hartmann CM, Wagner CA, Busch AE, Markovich D, Biber J, Lang F, Murer H (1995) Transport characteristics of a murine renal Na/Pi-cotransporter. Pflugers Arch - Eur J Physiol 430:830–836
Hilfiker H, Hattenhauer O, Traebert M, Forster I, Murer H, Biber J (1998) Characterization of a murine type II sodium-phosphate cotransporter expressed in mammalian small intestine. Proc Natl Acad Sci U S A 95:14564–14569
Hoffmann N, Thees M, Kinne R (1976) Phosphate transport by isolated renal brush border vesicles. Pflugers Arch 362:147–156
Kaback HR, Sahin-Toth M, Weinglass AB (2001) The kamikaze approach to membrane transport. Nat Rev Mol Cell Biol 2:610–620. https://doi.org/10.1038/35085077
Karlin A, Akabas MH (1998) Substituted-cysteine accessibility method. Methods Enzymol 293:123–145
Kohl B, Wagner CA, Huelseweh B, Busch AE, Werner A (1998) The Na+−phosphate cotransport system (NaPi-II) with a cleaved protein backbone: implications on function and membrane insertion. J Physiol 508(Pt 2):341–350
Kohler K, Forster IC, Lambert G, Biber J, Murer H (2000) The functional unit of the renal type IIa Na+/Pi cotransporter is a monomer. J Biol Chem 275:26113–26120
Kohler K, Forster IC, Stange G, Biber J, Murer H (2002) Identification of functionally important sites in the first intracellular loop of the NaPi-IIa cotransporter. Am J Phys 282:F687–F696
Kohler K, Forster IC, Stange G, Biber J, Murer H (2002) Transport function of the renal type IIa Na+/Pi cotransporter is codetermined by residues in two opposing linker regions. J Gen Physiol 120:693–703
Kohler K, Forster IC, Stange G, Biber J, Murer H (2003) Essential cysteine residues of the type IIa Na+/Pi cotransporter. Pflugers Arch - Eur J Physiol 446:203–210
Krishnamurthy H, Piscitelli CL, Gouaux E (2009) Unlocking the molecular secrets of sodium-coupled transporters. Nature 459:347–355. https://doi.org/10.1038/nature08143
Krofchick D, Huntley SA, Silverman M (2004) Transition states of the high-affinity rabbit Na(+)/glucose cotransporter SGLT1 as determined from measurement and analysis of voltage-dependent charge movements. Am J Phys Cell Physiol 287:C46–C54. https://doi.org/10.1152/ajpcell.00008.2004
Lambert G, Forster IC, Biber J, Murer H (2000) Cysteine residues and the structure of the rat renal proximal tubular type II sodium phosphate cotransporter (rat NaPi IIa). J Membr Biol 176:133–141
Lambert G, Forster IC, Stange G, Biber J, Murer H (1999) Properties of the mutant Ser-460-Cys implicate this site in a functionally important region of the type IIa Na+/Pi cotransporter protein. J Gen Physiol 114:637–652
Lambert G, Forster IC, Stange G, Kohler K, Biber J, Murer H (2001) Cysteine mutagenesis reveals novel structure-function features within the predicted third extracellular loop of the type IIa Na+/Pi cotransporter. J Gen Physiol 117:533–546
Lambert G, Traebert M, Hernando N, Biber J, Murer H (1999) Studies on the topology of the renal type II NaPi-cotransporter. Pflugers Arch - Eur J Physiol 437:972–978
Longpre JP, Sasseville LJ, Lapointe JY (2012) Simulated annealing reveals the kinetic activity of SGLT1, a member of the LeuT structural family. J Gen Physiol 140:361–374. https://doi.org/10.1085/jgp.201210822
Loo DD, Hazama A, Supplisson S, Turk E, Wright EM (1993) Relaxation kinetics of the Na+/glucose cotransporter. Proc Natl Acad Sci U S A 90:5767–5771
Loo DD, Hirayama BA, Gallardo EM, Lam JT, Turk E, Wright EM (1998) Conformational changes couple Na+ and glucose transport. Proc Natl Acad Sci U S A 95:7789–7794
Loo DD, Hirayama BA, Karakossian MH, Meinild AK, Wright EM (2006) Conformational dynamics of hSGLT1 during Na+/glucose cotransport. J Gen Physiol 128:701–720. https://doi.org/10.1085/jgp.200609643
Loo DD, Jiang X, Gorraitz E, Hirayama BA, Wright EM (2013) Functional identification and characterization of sodium binding sites in Na symporters. Proc Natl Acad Sci U S A 110:E4557–E4566. https://doi.org/10.1073/pnas.1319218110
Lu CC, Hilgemann DW (1999) GAT1 (GABA:Na+:Cl-) cotransport function. Steady state studies in giant Xenopus oocyte membrane patches. J Gen Physiol 114:429–444
Mackenzie B, Loo DD, Wright EM (1998) Relationships between Na+/glucose cotransporter (SGLT1) currents and fluxes. J Membr Biol 162:101–106
Magagnin S, Werner A, Markovich D, Sorribas V, Stange G, Biber J, Murer H (1993) Expression cloning of human and rat renal cortex Na/Pi cotransport. Proc Natl Acad Sci U S A 90:5979–5983
Mager S, Cao Y, Lester HA (1998) Measurement of transient currents from neurotransmitter transporters expressed in Xenopus oocytes. Methods Enzymol 296:551–566
Mager S, Naeve J, Quick M, Labarca C, Davidson N, Lester HA (1993) Steady states, charge movements, and rates for a cloned GABA transporter expressed in Xenopus oocytes. Neuron 10:177–188
Mancusso R, Gregorio GG, Liu Q, Wang DN (2012) Structure and mechanism of a bacterial sodium-dependent dicarboxylate transporter. Nature 491:622–626. https://doi.org/10.1038/nature11542
Meinild AK, Forster IC (2012) Using lithium to probe sequential cation interactions with GAT1. Am J Phys Cell Physiol 302:C1661–C1675. https://doi.org/10.1152/ajpcell.00446.2011
Mitchell P (1957) A general theory of membrane transport from studies of bacteria. Nature 180:134–136
Mulligan C, Fenollar-Ferrer C, Fitzgerald GA, Vergara-Jaque A, Kaufmann D, Li Y, Forrest LR, Mindell JA (2016) The bacterial dicarboxylate transporter VcINDY uses a two-domain elevator-type mechanism. Nat Struct Mol Biol 23:256–263. https://doi.org/10.1038/nsmb.3166
Mulligan C, Fitzgerald GA, Wang DN, Mindell JA (2014) Functional characterization of a Na+−dependent dicarboxylate transporter from Vibrio cholerae. J Gen Physiol 143:745–759. https://doi.org/10.1085/jgp.201311141
Murer H, Ahearn G, Amstutz M, Biber J, Brown C, Gmaj P, Hagenbuch B, Malmstrom K, Mohrmann I, Mohrmann M et al (1985) Cotransport systems for inorganic sulfate and phosphate in small intestine and renal proximal tubule. Ann N Y Acad Sci 456:139–152
Murer H, Hernando N, Forster I, Biber J (2000) Proximal tubular phosphate reabsorption: molecular mechanisms. Physiol Rev 80:1373–1409
Murer H, Stern H, Burckhardt G, Storelli C, Kinne R (1980) Sodium-dependent transport of inorganic phosphate across the renal brush border membrane. Adv Exp Med Biol 128:11–23
Nalbant P, Boehmer C, Dehmelt L, Wehner F, Werner A (1999) Functional characterization of a Na+-phosphate cotransporter (NaPi-II) from zebrafish and identification of related transcripts. J Physiol 520(Pt 1):79–89
Pajor AM, Hirayama BA, Loo DD (1998) Sodium and lithium interactions with the Na+/Dicarboxylate cotransporter. J Biol Chem 273:18923–18929
Parent L, Supplisson S, Loo DD, Wright EM (1992) Electrogenic properties of the cloned Na+/glucose cotransporter: II. A transport model under nonrapid equilibrium conditions. J Membr Biol 125:63–79
Patti M, Fenollar-Ferrer C, Werner A, Forrest LR, Forster IC (2016) Cation interactions and membrane potential induce conformational changes in NaPi-IIb. Biophys J 111:973–988. https://doi.org/10.1016/j.bpj.2016.07.025
Patti M, Forster IC (2014) Correlating charge movements with local conformational changes of a na(+)-coupled cotransporter. Biophys J 106:1618–1629. https://doi.org/10.1016/j.bpj.2014.02.028S0006-3495(14)00267-7
Patti M, Ghezzi C, Forster IC (2013) Conferring electrogenicity to the electroneutral phosphate cotransporter NaPi-IIc (SLC34A3) reveals an internal cation release step. Pflugers Arch - Eur J Physiol 465:1261–1279. https://doi.org/10.1007/s00424-013-1261-9
Priest M, Bezanilla F (2015) Functional site-directed fluorometry. Adv Exp Med Biol 869:55–76. https://doi.org/10.1007/978-1-4939-2845-3_4
Radanovic T, Gisler SM, Biber J, Murer H (2006) Topology of the type IIa Na+/P(i) cotransporter. J Membr Biol 212:41–49. https://doi.org/10.1007/s00232-006-0033-2
Ravera S, Virkki LV, Murer H, Forster IC (2007) Deciphering PiT transport kinetics and substrate specificity using electrophysiology and flux measurements. Am J Phys Cell Physiol 293:C606–C620. https://doi.org/10.1152/ajpcell.00064.2007
Sacktor B, Cheng L (1981) Sodium gradient-dependent phosphate transport in renal brush border membrane vesicles. Effect of an intravesicular greater than extravesicular proton gradient. J Biol Chem 256:8080–8084
Sahin-Toth M, Lawrence MC, Kaback HR (1994) Properties of permease dimer, a fusion protein containing two lactose permease molecules from Escherichia coli. Proc Natl Acad Sci U S A 91:5421–5425
Samarzija I, Molnar V, Fromter E (1983) pH--dependence of phosphate absorption in rat renal proximal tubule. Proc Eur Dial Transplant Assoc 19:779–783
Schaffhauser DF, Patti M, Goda T, Miyahara Y, Forster IC, Dittrich PS (2012) An integrated field-effect microdevice for monitoring membrane transport in Xenopus laevis oocytes via lateral proton diffusion. PLoS One 7:e39238. https://doi.org/10.1371/journal.pone.0039238PONE-D-12-05937
Segawa H, Kaneko I, Takahashi A, Kuwahata M, Ito M, Ohkido I, Tatsumi S, Miyamoto K (2002) Growth-related renal type II Na/Pi cotransporter. J Biol Chem 277:19665–19672
Sonders MS, Amara SG (1996) Channels in transporters. Curr Opin Neurobiol 6:294–302
Strevey J, Brunette MG, Beliveau R (1984) Effect of arginine modification on kidney brush-border-membrane transport activity. Biochem J 223:793–802
Szczepanska-Konkel M, Yusufi AN, Lin JT, Dousa TP (1989) Structural requirement of monophosphates for inhibition of Na+-Pi cotransport in renal brush border membrane. Biochem Pharmacol 38:4191–4197
Uwai Y, Arima R, Takatsu C, Furuta R, Kawasaki T, Nabekura T (2014) Sodium-phosphate cotransporter mediates reabsorption of lithium in rat kidney. Pharmacol Res 87:94–98. https://doi.org/10.1016/j.phrs.2014.06.012
Vergara-Jaque A, Fenollar-Ferrer C, Kaufmann D, Forrest LR (2015) Repeat-swap homology modeling of secondary active transporters:updated protocol and prediction of elevator-type mechanisms. Front Pharmacol 6:1–12. https://doi.org/10.3389/fphar.2015.00183
Vergara-Jaque A, Fenollar-Ferrer C, Mulligan C, Mindell JA, Forrest LR (2015) Family resemblances: a common fold for some dimeric ion-coupled secondary transporters. J Gen Physiol 146:423–434. https://doi.org/10.1085/jgp.201511481
Virkki LV, Forster IC, Bacconi A, Biber J, Murer H (2005) Functionally important residues in the predicted 3rd transmembrane domain of the type IIa sodium-phosphate co-transporter (NaPi-IIa). J Membr Biol 206:227–238
Virkki LV, Forster IC, Biber J, Murer H (2005) Substrate interactions in the human type IIa sodium-phosphate cotransporter (NaPi-IIa). Am J Phys 288:F969–F981
Virkki LV, Murer H, Forster IC (2006) Mapping conformational changes of the type IIb Na+/Pi cotransporter by voltage clamp fluorometry. J Biol Chem 281:28837–28849
Virkki LV, Murer H, Forster IC (2006) Voltage clamp fluorometric measurements on a type II Na+-coupled Pi cotransporter: shedding light on substrate binding order. J Gen Physiol 127:539–555
Weiss JN (1997) The hill equation revisited: uses and misuses. FASEB J 11:835–841
Werner A, Patti M, Zinad HS, Fearn A, Laude A, Forster I (2016) Molecular determinants of transport function in zebrafish Slc34a Na-phosphate transporters. Am J Phys Regul Integr Comp Phys 311:R1213–R1222. https://doi.org/10.1152/ajpregu.00020.2016
Xiao Y, Boyer CJ, Vincent E, Dugre A, Vachon V, Potier M, Beliveau R (1997) Involvement of disulphide bonds in the renal sodium/phosphate co-transporter NaPi-2. Biochem J 323(Pt 2):401–408
Yamashita A, Singh SK, Kawate T, Jin Y, Gouaux E (2005) Crystal structure of a bacterial homologue of Na+/Cl-dependent neurotransmitter transporters. Nature 437:215–223. https://doi.org/10.1038/nature03978
Yernool D, Boudker O, Jin Y, Gouaux E (2004) Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431:811–818. https://doi.org/10.1038/nature03018
Zampighi GA, Kreman M, Boorer KJ, Loo DD, Bezanilla F, Chandy G, Hall JE, Wright EM (1995) A method for determining the unitary functional capacity of cloned channels and transporters expressed in Xenopus laevis oocytes. J Membr Biol 148:65–78
Acknowledgements
The author wishes to acknowledge the numerous contributions made by those working in the phosphate transport field past and present. The support and encouragement given by Heini Murer and Jürg Biber (University of Zurich), Ernest Wright and his past and present colleagues Don Loo, Bruce Hirayama and Sepehr Eskandari (UCLA) and more recently the collaborations with Cristina Fenollar-Ferrer and Lucy Forrest (NIH) are particularly appreciated. Special thanks are due to colleagues, postdocs and doctoral candidates at the Murer laboratory, without whom the insights gained over the years would have been impossible, and whose names and contributions appear in the original references. Most of the studies reported here were supported by grants from the Swiss National Science Foundation and Hartmann Müller-Stiftung (University of Zurich) to the author and Heini Murer, as well as other funding sources cited in the original publications. Finally, the author acknowledges the outstanding support from Steven Petrou and colleagues at the Ion Channels in Human Diseases Laboratory (Florey Institute).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the special issue on Phosphate transport in Pflügers Archiv—European Journal of Physiology
Appendix: Tools and protocols used to characterise electrogenic SLC34 proteins expressed in Xenopus oocytes
Appendix: Tools and protocols used to characterise electrogenic SLC34 proteins expressed in Xenopus oocytes
-
1.
Steady-state and presteady-state assays-characterising electrogenic behaviour at the macroscopic level
Voltage steps applied to a voltage clamped whole Xenopus oocyte expressing electrogenic cotransporters like NaPi-IIa/b reveal two components of membrane current: a presteady state, transient relaxing component and a steady-state component (Fig. 9, left data set). Analysis of both components yields important kinetic information about the transport mechanism. The currents recorded from a whole oocyte are macroscopic and represent the mean electrogenic behaviour of a large (typically ≥ 1010) population of transporters. Unlike ion channels, whose activity can be resolved at the single molecule level, the slow rate of charge translocation (~ fA or less) of membrane transporters is several orders of magnitude below that which can be presently resolved. However, like ion channels, we assume individual transporters behave independently and the population behaviour can be described in terms of the probability of occupying a particular conformational state. This depends on the membrane potential, substrate availability and the partial reaction rates associated with entering and leaving that state, according to the kinetic scheme (Fig. 1b).
Two electrode voltage clamp of whole Xenopus oocytes expressing electrogenic NaPi-IIa/b. The voltage step protocol (shown here for 20 mV steps in range − 160 to + 80 mV) is used to characterise the steady-state and presteady-state electrogenic properties of oocytes. Voltage steps evoke a capacitative component (presteady-state) comprising the linear charging/discharging of the endogenous membrane phospholipid and the non-linear transporter-related charge relaxations. In the presence of Pi (right), the steady-state holding current increases and the exogenous charge relaxations are suppressed
The steady-state current component comprises endogenous oocyte currents and, in the absence of external Pi, the constitutive leak of the transporter. In the presence of Pi (Fig. 9, right data set) the downward deflection (arrowed) of the steady-state current indicates inward movement of charge accompanying active cotransport. Subtraction of these data sets from one another eliminates the endogenous component and yields the Pi-dependent current (IPi) with a small error due to the constitutive leak that we assume is suppressed by Pi. This component can be independently assayed using the inhibitor PFA (e.g. Fig. 6a). Ignoring the leak can potentially lead to an underestimate of the cotransport activity as well as charge translocation in stoichiometry assays depending on its magnitude (e.g. [2] and see Fig. 6 in [25]). By repeating these measurements with different substrate concentrations, standard phenomenological parameters such as apparent substrate affinity and maximum transport rate are derived as a function of membrane voltage. An important caveat for all oocyte-derived kinetic data is that these parameters are generally obtained at temperatures in the range 18-20 °C and extrapolation to mammalian physiology conditions necessitates taking account of temperature.
The presteady-state component comprises the endogenous charging transient of the oocyte lipid bilayer capacitance upon which the transporter-associated presteady-state current relaxations are superimposed (Fig. 9). The former is a linear function of the test voltage and can be eliminated by procedures such as curve fitting, subtracting matching records in the presence of a blocker or P/n protocols used to resolve ion channel gating currents (e.g. [4]). Analysis of the transporter-associated relaxations focuses on the charge displaced (Q) obtained by numerical integration, and the relaxation time constant (τ) obtained by exponential fitting. Both are important for characterising transporter kinetics. The fidelity and validity of presteady-state data can be tested by confirming charge balance for voltage steps to and from the holding potential, verifying independence of total charge movement from the holding potential and voltage independence of τ for the relaxations in response to the return step to the holding potential and establishing a direct correlation between steady-state transport and the total charge displaced.
The Q-V data show a characteristic sigmoidal relationship with saturation at either or both potential extremes due to the fixed number of charges involved. This is usually fit with a single Boltzmann-type function (Eq. 1) to obtain three parameters: the effective charge (z) per transporter (from the slope), the total charge displaced (Qmax) and the midpoint potential (V0.5), an equilibrium potential at which half the charge has been displaced. This procedure is valid for a two state model, and with more than one partial reaction contributing to the charge movement, the single Boltzmann function fit is clearly an approximation (e.g. [62]). Despite this limitation, these parameters can be used to characterise and interpret changes in substrate interactions and voltage dependence. The Q-V data are sensitive to external [Na+] (Fig. 10) and this relationship is a key biophysical signature for understanding cation interactions. As external [Na+] increases, V0.5 shifts towards depolarizing potentials (arrow), consistent with Na+ ions binding more easily according to their availability. The apparent charge associated with the empty carrier and the Na+ ion interactions can be estimated from z for each data set. The invariance of Qmax with changing [Na+] is expected for a given number of transporters, each of which binds a fixed number of Na+ ions. At low [Na+] this is less obvious because of limitations of the fitting procedure and at 0 mM Na+, charge movement is due to the empty carrier alone. The growth in z with [Na+] reflects the increased contribution of Na+ interactions to the effective charge movement contributed by each transporter. The limiting slope of V0.5 vs log10 [Na+] (~ 120 mV/decade) is also consistent with a sequential interaction of 2 Na+ ions per transporter. These findings are also predicted by deriving an analytical expression for Q-V, and numerically solving for V0.5 as a function of [Na+] (see Eq. 2–4) (e.g. [3, 71, 78]. Estimates of the dissociation constants for each cation interaction can be made and compared with the effect of mutations at the predicted cation binding sites (e.g. [29, 71].
Analysis of presteady-state relaxations. Transporter-related presteady-state relaxations (a) can be integrated to give the charge movement (Q-V) (b) and fit with a single exponential to give the main relaxation time constant (τ-V) (c). As external [Na+] is increased, the midpoint of Q-V and peak of τ-V shift as shown. Fitting the Q-V data with a Boltzmann function (Eq. 1) yields the parameters Qmax, z and V0.5 which are plotted as functions of [Na+] (d). The limiting slope of the relationship between V0.5 and log10[Na] (d, inset) indicates that 2 Na+ ions interact. Examples from representative oocytes expressing flounder NaPi-IIb
In addition, the ratio Qmax/ze (where e is the electronic charge) can also be used to predict (i) the number of transporters (Nt) contributing to the charge movement (and by implication the cotransport activity) (Eq. 5) and, (ii) the turnover rate (Rt) when combined with the steady state cotransport current (e.g. [41] (Eq. 6). However, these should be taken as estimates only, given the inherent assumptions in the method (see [47, 114]).
The dependence of the relaxation time constant on voltage (τ-V) shows a “bell-shaped” form typical for non-linear charge movements associated with membrane proteins (Fig. 10). For a two state system (e.g. the empty carrier transition 0↔1, Fig. 1b), the forward and backward rate constants can be predicted from fits to the τ-V data. For a 3 state model (empty carrier and a Na+ binding step) theory predicts two time constants and analytical expressions become more complex. In practice, it may be difficult to resolve more than one relaxations component due to limitations of the curve fitting procedure and bandwidth of the voltage clamp. A single exponential fit to experimental data shows that like the shift of V0.5, the peak time constant shifts to more depolarizing potentials as [Na+] increases (arrow), consistent with having the empty carrier and at least one Na-dependent partial reaction contributing to the charge movement.
-
2.
Voltage clamp fluorometry
This technique is readily applied to whole oocytes by combining the two electrode voltage clamp with a basic fluorescence microscope (e.g. [17, 91]) (Fig. 11). The instrumentation used in Zurich to study SLC34 proteins was modified from a design by the Wright group (UCLA) [69]. It has allowed real-time measurement of electrogenic activity (steady-state and presteady-state) and simultaneous fluorescence emissions from oocytes previously labelled with a fluorophore and covalently linked to an engineered cysteine in the transporter. The same voltage step protocols used for steady-state and presteady-state analysis can be used for VCF studies although more signal averaging may be required to obtain an acceptable signal to noise ratio. As all cysteines exposed to the labelling medium will be potentially labelled, an endogenous component will contribute to the background fluorescence. Generally, this is found not vary with membrane potential, but can still potentially compromise the signal resolution if ΔF from the transporter is small, by limiting the useful dynamic range. To overcome this, electronic offset can be applied before data acquisition. Pre-labelling endogenous cysteines and other procedures can also help reduce the background signal [91]. Precautions must also be taken to take account of loss of fluorescence signal during the course of an experiment due to bleaching, washout of fluorophore or internalisation of proteins (e.g. [45, 108]). Like the presteady-state assays, the fluorescence signal is a mean of emissions arising from a population of labelled transporters that at any instant can reside in different conformational states, depending on the probability of state occupancy. For example, if the fluorophore experiences a more polar or charged environment, the fluorescence is quenched and this can then be related to state occupancy. Collisional quenching is commonly assumed to be the underlying mechanism leading to ΔF and has been observed experimentally (e.g. [88]) but other mechanisms should be considered (e.g. [16]). The ΔF-V data are usually fit with a Boltzmann function although a direct correspondence with the Q-V parameters is not always obtained because there may not necessarily be a correlation between charge movement and the change in microenvironment of the fluorophore, depending on the labelling site. Unlike the electrophysiological assays, changes in fluorescence can also be reported by electroneutral processes in response to changing substrate [45].
Whole oocyte voltage clamp fluorometry. The basic hardware arrangement for whole Xenopus oocyte recording combines a two-electrode voltage clamp and basic fluorescent microscope. Optimal fluorescence signal is obtained with the cell inverted from the normal arrangement in Fig. 9. Filter cube components can be changed for optimal response depending on the fluorophore. Typical simultaneous recordings of Im and ?F are shown (right) for an oocyte labelled with MTS-TAMRA. The bandwidth for each signal was 500 Hz
-
3.
Equations used in presteady-state analysis:
-
a)
Boltzmann Q-V relationship
$$ Q={Q}_{hyp}+\frac{Q_{max}}{1+{\mathit{\exp}}^{\frac{ze\left({V}_{0.5}-V\right)}{kT}}} $$(1)where V0.5 is the midpoint voltage, z is the apparent valency/protein, Qmax is the total charge available to move, Qhyp is the charge at the hyperpolarizing limit and is a function of the holding potential and e, k and T have their usual meanings.
-
b)
Derivation of Q-V relationship and V0.5 vs [Na] derived from the four state kinetic model (states 0,1–3, Fig. 1b).
The total charge displaced for a voltage step from ∞ (i.e. all transporters in state 0) to V and neglecting charge movement due to transition 0↔7, is given by [3]:
$$ {Q}_{\infty}^V=-{N}_te\left[\frac{\Big(\left({z}_{01}+{z}_{12}+{z}_{23}\right)+\left({z}_{12}+{z}_{23}\right)\alpha +{Z}_{23}\alpha \beta}{1+\alpha +\alpha \beta +\alpha \beta \gamma}\right] $$(2)where Nt is the number of transporters, α = k01/k10, β = k12/k21 and γ = k23/k32, (i.e. the ratio of forward to backward rate constants for each partial reaction), and zij is the apparent valence for the partial reaction that couples states i and j. Using rate theory, the forward and backward rates (kij, kji), assuming symmetrical barriers, are expressed as \( {k}_{ij}={k}_{ij}^0{\mathit{\exp}}^{-e{z}_{ij}V/2 kT} \); \( {k}_{ji}={k}_{ji}^0{\mathit{\exp}}^{e{z}_{ij}V/2 kT} \). For partial reactions involving cation interactions, the forward rates are scaled by the cation concentration.
To link Eq. 2 to the single Boltzmann function formulation of the Q-V data, the voltage at which 50% of the total charge has been displaced (V0.5) is found by setting \( {Q}_{\infty}^V=0.5{N}_te\left({z}_{12}+{z}_{23}+{z}_{23}\right) \), to give:
$$ \left({z}_{01}+{z}_{12}+{z}_{23}\right)\left(1-0.5\left(1+\alpha +\alpha \beta +\alpha \beta \gamma \right)\right)+\alpha \left({z}_{12}+{z}_{23}\right)+\alpha \beta {z}_{23}=0 $$(3)Substituting for α, β, γ, for [Na] large this reduces to:
$$ {\alpha}^0{\beta}^0{\upgamma}^0\ {\left[ Na\right]}^2\ \left({\exp}^{-\mathrm{e}{V}_{0.5}\left({z}_{01}+{z}_{12}+{z}_{23}\right)/ kT}\right)=1 $$(4)where α0 = k001/k010, β0 = k012/k021 and γ0 = k023/k032, and the superscript 0 refers to values at V = 0. A plot of V0.5 vs loge[Na] yields a limiting slope of 2kT/[e(z01 + z12 + z23)] mV/e-fold change in [Na] (or ≈ 116/(z01 + z12 + z23) mV/10-fold change in [Na]) at 20 °C; i.e. for one net charge translocated across the transmembrane field, the predicted slope is then 116 mV/10-fold change in [Na] [3] as observed experimentally (see Fig. 5e).
-
c)
Estimation of number of transporters (Nt) and turnover rate (Rt)
$$ {N}_{\mathrm{t}}={Q}_{\mathrm{max}}/\mathrm{ze} $$(5)$$ {R}_{\mathrm{t}}={I_{\mathrm{Pi}}}^{\mathrm{max}}/\left({N}_{\mathrm{t}}{\mathrm{z}}_{\mathrm{t}}\mathrm{e}\right) $$(6)where IPimax is the maximum transport current and zt is the charge translocated/cycle. The estimates of Rt require specification of the experimental conditions ([Na+], Vm etc.) and are dependent on the assumptions related to estimating Nt from a single Boltzmann fit [114].
-
a)
Rights and permissions
About this article
Cite this article
Forster, I.C. The molecular mechanism of SLC34 proteins: insights from two decades of transport assays and structure-function studies. Pflugers Arch - Eur J Physiol 471, 15–42 (2019). https://doi.org/10.1007/s00424-018-2207-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-018-2207-z