Abstract
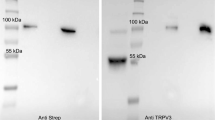
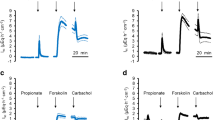
Large quantities of protein are degraded in the fermentative parts of the gut to ammonia, which is absorbed, detoxified to urea, and excreted, leading to formation of nitrogenous compounds such as N2O that are associated with global warming. In ruminants, channel-mediated uptake of NH4 + from the rumen predominates. The molecular identity of these channels remains to be clarified. Ruminal cells and epithelia from cows and sheep were investigated using patch clamp, Ussing chamber, microelectrode techniques, and qPCR. In patch clamp experiments, bovine ruminal epithelial cells expressed a conductance for NH4 + that could be blocked in a voltage-dependent manner by divalent cations. In the native epithelium, NH4 + depolarized the apical potential, acidified the cytosol and induced a rise in short-circuit current (I sc) that persisted after the removal of Na+, was blocked by verapamil, enhanced by the removal of divalent cations, and was sensitive to certain transient receptor potential (TRP) channel modulators. Menthol or thymol stimulated the I sc in Na+ or NH4 + containing solutions in a dose-dependent manner and modulated transepithelial Ca2+ fluxes. On the level of messenger RNA (mRNA), ovine and bovine ruminal epithelium expressed TRPA1, TRPV3, TRPV4, TRPM6, and TRPM7, with any expression of TRPV6 marginal. No bands were detected for TRPV1, TRPV5, or TRPM8. Functional and molecular biological data suggest that the transport of NH4 +, Na+, and Ca2+ across the rumen involves TRP channels, with TRPV3 and TRPA1 emerging as prime candidate genes. TRP channels may also contribute to the transport of NH4 + across other epithelia.








Similar content being viewed by others
References
Abdoun K, Stumpff F, Wolf K, Martens H (2005) Modulation of electroneutral Na transport in sheep rumen epithelium by luminal ammonia. Am J Physiol Gastrointest Liver Physiol 289:G508–G520. doi:10.1152/ajpgi.00436.2004
Alpizar YA, Gees M, Sanchez A, Apetrei A, Voets T, Nilius B, Talavera K (2013) Bimodal effects of cinnamaldehyde and camphor on mouse TRPA1. Pflugers Arch 465:853–864. doi:10.1007/s00424-012-1204-x
Aschenbach JR, Bilk S, Tadesse G, Stumpff F, Gäbel G (2009) Bicarbonate-dependent and bicarbonate-independent mechanisms contribute to nondiffusive uptake of acetate in the ruminal epithelium of sheep. Am J Physiol Gastrointest Liver Physiol 296:G1098–G1107. doi:10.1152/ajpgi.90442.2008
Aschenbach JR, Penner GB, Stumpff F, Gabel G (2011) Ruminant nutrition symposium: role of fermentation acid absorption in the regulation of ruminal pH. J Anim Sci 89:1092–1107. doi:10.2527/jas.2010-3301
Bandell M, Dubin AE, Petrus MJ, Orth A, Mathur J, Hwang SW, Patapoutian A (2006) High-throughput random mutagenesis screen reveals TRPM8 residues specifically required for activation by menthol. Nat Neurosci 9:493–500. doi:10.1038/nn1665
Barry PH (2012) Ionic mobility tables. http://web.med.unsw.edu.au/phbsoft/mobility_listings.htm
Barry PH, Lynch JW (1991) Liquid junction potentials and small cell effects in patch-clamp analysis. J Membr Biol 121:101–117. doi:10.1007/BF01870526
Behera SN, Sharma M, Aneja VP, Balasubramanian R (2013) Ammonia in the atmosphere: a review on emission sources, atmospheric chemistry and deposition on terrestrial bodies. Environ Sci Pollut Res Int 20:8092–8131. doi:10.1007/s11356-013-2051-9
Blaydon DC, Kelsell DP (2014) Defective channels lead to an impaired skin barrier. J Cell Sci 127:4343–4350. doi:10.1242/jcs.154633
Bödeker D, Kemkowski J (1996) Participation of NH4+ in total ammonia absorption across the rumen epithelium of sheep (Ovis aries). Comp Biochem Physiol A Physiol 114:305–310. doi:10.1016/0300-9629(96)00012-6
Boron WF (2010) Sharpey-Schafer lecture: gas channels. Exp Physiol 95:1107–1130. doi:10.1113/expphysiol.2010.055244
Bouwman AF, Lee DS, Asman WAH, Dentener FJ, Van Der Hoek KW, Olivier JGJ (1997) A global high-resolution emission inventory for ammonia. Glob Biogeochem Cycles 11:561–587. doi:10.1029/97gb02266
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824. doi:10.1038/39807
Damann N, Voets T, Nilius B (2008) TRPs in our senses. Curr Biol 18:R880–R889. doi:10.1016/j.cub.2008.07.063
Delgado-Elorduy A, Theurer CB, Huber JT, Alio A, Lozano O, Sadik M, Cuneo P, De Young HD, Simas IJ, Santos JE, Nussio L, Nussio C, Webb KE Jr, Tagari H (2002) Splanchnic and mammary nitrogen metabolism by dairy cows fed dry-rolled or steam-flaked sorghum grain. J Dairy Sci 85:148–159. doi:10.3168/jds.S0022-0302(02)74063-0
Gäbel G, Butter H, Martens H (1999) Regulatory role of cAMP in transport of Na+, Cl- and short-chain fatty acids across sheep ruminal epithelium. Exp Physiol 84:333–345. doi:10.1111/j.1469-445X.1999.01758.x
Galfi P, Neogrady S, Kutas F (1981) Culture of epithelial cells from bovine ruminal mucosa. Vet Res Commun 4:295–300. doi:10.1007/BF02278507
Gärtner K, von Engelhardt W (1964) Experiments concerning the resorption mechanism of ammonia through the ruminal mucosa of ruminants. Dtsch Tierarztl Wochenschr 71:57–60
Gees M, Colsoul B, Nilius B (2010) The role of transient receptor potential cation channels in Ca2+ signaling. Cold Spring Harb Perspect Biol 2:a003962. doi:10.1101/cshperspect.a003962
Georgi MI, Rosendahl J, Ernst F, Gunzel D, Aschenbach JR, Martens H, Stumpff F (2014) Epithelia of the ovine and bovine forestomach express basolateral maxi-anion channels permeable to the anions of short-chain fatty acids. Pflugers Arch 466:1689–1712. doi:10.1007/s00424-013-1386-x
Grubisha O, Mogg AJ, Sorge JL, Ball LJ, Sanger H, Ruble CL, Folly EA, Ursu D, Broad LM (2014) Pharmacological profiling of the TRPV3 channel in recombinant and native assays. Br J Pharmacol 171:2631–2644. doi:10.1111/bph.12303
Handlogten ME, Hong SP, Zhang L, Vander AW, Steinbaum ML, Campbell-Thompson M, Weiner ID (2005) Expression of the ammonia transporter proteins Rh B glycoprotein and Rh C glycoprotein in the intestinal tract. Am J Physiol Gastrointest Liver Physiol 288:G1036–G1047. doi:10.1152/ajpgi.00418.2004
Harmeyer J, Martens H (1980) Aspects of urea metabolism in ruminants with reference to the goat. J Dairy Sci 63:1707–1728. doi:10.3168/jds.S0022-0302(80)83132-8
Hille B (2001) Ion Channels of Excitable Membranes. 3rd edn. Sinauer Associates, Sunderland, Mass. doi:ISBN 978–0878933211
Hodgkin AL (1951) The ionic basis of electrical activity in nerve and muscle. Biol Rev 26:339–409. doi:10.1111/j.1469-185X.1951.tb01204.x
Höller H, Breves G, Kocabatmaz M, Gerdes H (1988) Flux of calcium across the sheep rumen wall in vivo and in vitro. Q J Exp Physiol 73:609–618
Hsu YJ, Hoenderop JG, Bindels RJ (2007) TRP channels in kidney disease. Biochim Biophys Acta 1772:928–936. doi:10.1016/j.bbadis.2007.02.001
Karashima Y, Damann N, Prenen J, Talavera K, Segal A, Voets T, Nilius B (2007) Bimodal action of menthol on the transient receptor potential channel TRPA1. J Neurosci 27:9874–9884. doi:10.1523/JNEUROSCI.2221-07.2007
Kleen JL, Upgang L, Rehage J (2013) Prevalence and consequences of subacute ruminal acidosis in German dairy herds. Acta Vet Scand 55:48. doi:10.1186/1751-0147-55-48
Leonhard-Marek S (2002) Divalent cations reduce the electrogenic transport of monovalent cations across rumen epithelium. J Comp Physiol B 172:635–641. doi:10.1007/s00360-002-0292-x
Leonhard-Marek S, Stumpff F, Brinkmann I, Breves G, Martens H (2005) Basolateral Mg2+/Na + exchange regulates apical nonselective cation channel in sheep rumen epithelium via cytosolic Mg2+. Am J Physiol Gastrointest Liver Physiol 288:G630–G645. doi:10.1152/ajpgi.00275.2004
Li H, Sheppard DN, Hug MJ (2004) Transepithelial electrical measurements with the Ussing chamber. J Cyst Fibros 3(Suppl 2):123–126. doi:10.1016/j.jcf.2004.05.026
Lu Z, Stumpff F, Deiner C, Rosendahl J, Braun H, Abdoun K, Aschenbach JR, Martens H (2014) Modulation of sheep ruminal urea transport by ammonia and pH. Am J Physiol Regul Integr Comp Physiol 307:R558–R570. doi:10.1152/ajpregu.00107.2014
Marini AM, Matassi G, Raynal V, Andre B, Cartron JP, Cherif-Zahar B (2000) The human Rhesus-associated RhAG protein and a kidney homologue promote ammonium transport in yeast. Nat Genet 26:341–344. doi:10.1038/81656
Martens H, Gäbel G, Strozyk B (1991) Mechanism of electrically silent Na and Cl transport across the rumen epithelium of sheep. Exp Physiol 76:103–114. doi:10.1113/expphysiol.1991.sp00347
Montell C, Rubin GM (1989) Molecular characterization of the Drosophila trp locus: a putative integral membrane protein required for phototransduction. Neuron 2:1313–1323. doi:10.1016/0896-6273(89)90069-X
Musa-Aziz R, Chen LM, Pelletier MF, Boron WF (2009) Relative CO2/NH3 selectivities of AQP1, AQP4, AQP5, AmtB, and RhAG. Proc Natl Acad Sci U S A 106:5406–5411. doi:10.1073/pnas.0813231106
Nilius B, Prenen J, Owsianik G (2011) Irritating channels: the case of TRPA1. J Physiol 589:1543–1549. doi:10.1113/jphysiol.2010.200717
Nilius B, Szallasi A (2014) Transient receptor potential channels as drug targets: from the science of basic research to the art of medicine. Pharmacol Rev 66:676–814. doi:10.1124/pr.113.008268
Owsianik G, Talavera K, Voets T, Nilius B (2006) Permeation and selectivity of TRP channels. Annu Rev Physiol 68:685–717. doi:10.1146/annurev.physiol.68.040204.101406
Palazzo E, Rossi F, de Novellis V, Maione S (2013) Endogenous modulators of TRP channels. Curr Top Med Chem 13:398–407. doi:10.2174/1568026611313030014
Peier AM, Moqrich A, Hergarden AC, Reeve AJ, Andersson DA, Story GM, Earley TJ, Dragoni I, McIntyre P, Bevan S, Patapoutian A (2002) A TRP channel that senses cold stimuli and menthol. Cell 108:705–715. doi:10.1016/S0092-8674(02)00652-9
Ramsey IS, Delling M, Clapham DE (2006) An introduction to TRP channels. Annu Rev Physiol 68:619–647. doi:10.1146/annurev.physiol.68.040204.100431
Reiter B, Kraft R, Günzel D, Zeissig S, Schulzke JD, Fromm M, Harteneck C (2006) TRPV4-mediated regulation of epithelial permeability. FASEB J 20:1802–1812. doi:10.1096/fj.06-5772com
Sabirov RZ, Merzlyak PG, Islam MR, Okada T, Okada Y (2016) The properties, functions, and pathophysiology of maxi-anion channels. Pflugers Arch. doi:10.1007/s00424-015-1774-5
Saparov SM, Liu K, Agre P, Pohl P (2007) Fast and selective ammonia transport by aquaporin-8. J Biol Chem 282:5296–5301. doi:10.1074/jbc.M609343200
Schröder B, Rittmann I, Pfeffer E, Breves G (1997) In vitro studies on calcium absorption from the gastrointestinal tract in small ruminants. J Comp Physiol B 167:43–51
Schroder B, Wilkens MR, Ricken GE, Leonhard-Marek S, Fraser DR, Breves G (2015) Calcium transport in bovine rumen epithelium as affected by luminal Ca concentrations and Ca sources. Physiol Rep 3. doi:10.14814/phy2.12615
Schultheiss G, Martens H (1999) Ca-sensitive Na transport in sheep omasum. Am J Physiol 276:G1331–G1344. doi:10.1152/ajpgi.90532.2008
Schweigel M, Kolisek M, Nikolic Z, Kuzinski J (2008) Expression and functional activity of the Na/Mg exchanger, TRPM7 and MagT1 are changed to regulate Mg homeostasis and transport in rumen epithelial cells. Magnes Res 21:118–123
Sherkheli MA, Benecke H, Doerner JF, Kletke O, Vogt-Eisele AK, Gisselmann G, Hatt H (2009) Monoterpenoids induce agonist-specific desensitization of transient receptor potential vanilloid-3 (TRPV3) ion channels. J Pharm Pharm Sci 12:116–128
Stevens CE, Hume ID (1998) Contributions of microbes in vertebrate gastrointestinal tract to production and conservation of nutrients. Physiol Rev 78:393–427
Stokstad E (2014) Air pollution. Ammonia pollution from farming may exact hefty health costs. Science 343:238. doi:10.1126/science.343.6168.238
Stumpff F, Lodemann U, Van Kessel AG, Pieper R, Klingspor S, Wolf K, Martens H, Zentek J, Aschenbach JR (2013) Effects of dietary fibre and protein on urea transport across the cecal mucosa of piglets. J Comp Physiol B 183:1053–1063. doi:10.1007/s00360-013-0771-2
Stumpff F, Martens H, Bilk S, Aschenbach JR, Gäbel G (2009) Cultured ruminal epithelial cells express a large-conductance channel permeable to chloride, bicarbonate, and acetate. Pflugers Arch 457:1003–1022
Uppal SK, Wolf K, Martens H (2003) The effect of short chain fatty acids on calcium flux rates across isolated rumen epithelium of hay-fed and concentrate-fed sheep. J Anim Physiol Anim Nutr (Berl) 87:12–20
Villalta PC, Townsley MI (2013) Transient receptor potential channels and regulation of lung endothelial permeability. Pulm Circ 3:802–815. doi:10.1086/674765
Voets T, Janssens A, Prenen J, Droogmans G, Nilius B (2003) Mg2 + −dependent gating and strong inward rectification of the cation channel TRPV6. J Gen Physiol 121:245–260
Vogt-Eisele AK, Weber K, Sherkheli MA, Vielhaber G, Panten J, Gisselmann G, Hatt H (2007) Monoterpenoid agonists of TRPV3. Br J Pharmacol 151:530–540. doi:10.1038/sj.bjp.0707245
Vriens J, Nilius B, Vennekens R (2008) Herbal compounds and toxins modulating TRP channels. Curr Neuropharmacol 6:79–96. doi:10.2174/157015908783769644
Weiner ID (2004) The Rh gene family and renal ammonium transport. Curr Opin Nephrol Hypertens 13:533–540
Weiner ID, Verlander JW (2014) Ammonia transport in the kidney by Rhesus glycoproteins. Am J Physiol Renal Physiol 306:F1107–F1120. doi:10.1152/ajprenal.00013.2014
Wilkens MR, Kunert-Keil C, Brinkmeier H, Schröder B (2009) Expression of calcium channel TRPV6 in ovine epithelial tissue. Vet J 182:294–300. doi:10.1016/j.tvjl.2008.06.020
Wilkens MR, Mrochen N, Breves G, Schröder B (2011) Gastrointestinal calcium absorption in sheep is mostly insensitive to an alimentary induced challenge of calcium homeostasis. Comp Biochem Physiol B Biochem Mol Biol 158:199–207. doi:10.1016/j.cbpb.2010.11.008
Wilkens MR, Praechter C, Breves G, Schröder B (2015) Stimulating effects of a diet negative in dietary cation-anion difference on calcium absorption from the rumen in sheep. J Anim Physiol Anim Nutr (Berl). doi:10.1111/jpn.12296
Winkler FK (2006) Amt/MEP/Rh proteins conduct ammonia. Pflugers Arch 451:701–707
Wolffram S, Frischknecht R, Scharrer E (1989) Influence of theophylline on the electrical potential difference and ion fluxes (Na, Cl, K) across the isolated rumen epithelium of sheep. Zentralbl Veterinarmed A 36:755–762
Xu H, Delling M, Jun JC, Clapham DE (2006) Oregano, thyme and clove-derived flavors and skin sensitizers activate specific TRP channels. Nat Neurosci 9:628–635. doi:10.1038/nn1692
Xu H, Ramsey IS, Kotecha SA, Moran MM, Chong JA, Lawson D, Ge P, Lilly J, Silos-Santiago I, Xie Y, DiStefano PS, Curtis R, Clapham DE (2002) TRPV3 is a calcium-permeable temperature-sensitive cation channel. Nature 418:181–186. doi:10.1038/nature00882
Acknowledgments
Financial support for this study came from the Forschungskommission of the Freie Universität Berlin and from the European Social Fund (ESF) and the German Ministry of Economics and Technology based on a decision of the German Parliament (Projektträger Jülich 03EFABE057). In the latter part of the study, Katharina Schrapers (née Hille) was supported by the “Akademie für Tiergesundheit.” The expert technical help of Gisela Manz, Susanne Trappe, Martin Grunau, Uwe Tietjen, and last but certainly not least, Katharina Wolf is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
This study was performed for purely scientific reasons within a conventional academic framework but has recently led to a patent that is pending (involving the authors Friederike Stumpff and Julia Rosendahl, both as employees of the Freie Universität) and a start-up company supported by “Exist” and the Freie Universität Berlin. The company is owned by Julia Rosendahl, Hannah Braun, Katharina Schrapers, and Friederike Stumpff. This publication will have no impact on the commercial success or failure of that enterprise, and there is no conflict of interest.
Some preliminary results were published in the form of abstracts and a thesis (Julia Rosendahl).
Rights and permissions
About this article
Cite this article
Rosendahl, J., Braun, H.S., Schrapers, K.T. et al. Evidence for the functional involvement of members of the TRP channel family in the uptake of Na+ and NH4 + by the ruminal epithelium. Pflugers Arch - Eur J Physiol 468, 1333–1352 (2016). https://doi.org/10.1007/s00424-016-1835-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-016-1835-4