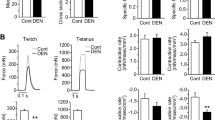
Abstract
Causes of disuse atrophy include loss of upper motor neurons, which occurs in spinal cord injury (SCI) or lower motor neurons (denervation). Whereas denervation quickly results in muscle fibrillations, SCI causes delayed onset of muscle spasticity. To compare the influence of denervation or SCI on muscle atrophy and atrophy-related gene expression, male rats had transection of either the spinal cord or sciatic nerve and were sacrificed 3, 7, or 14 days later. Rates of atrophy increased gradually over the first week after denervation and then were constant. In contrast, atrophy after SCI peaked at 1 week, then declined sharply. The greater atrophy after SCI compared to denervation was preceded by high levels of ubiquitin ligase genes, MAFbx and MuRF1, which then also markedly declined. After denervation, however, expression of these genes remained elevated at lower levels throughout the 2-week time course. Interestingly, expression of the muscle growth factor, IGF-1 was increased at 3 days after denervation when fibrillation also peaks compared to SCI. Expression of IGF-1R, GADD45, myogenin, and Runx1 were also initially increased after denervation or SCI, with later declines in expression levels which correlated less well with rates of atrophy. Thus, there were significant time-dependent differences in muscle atrophy and MAFbx, MuRF1, and IGF-1 expression following SCI or denervation which may result from distinct temporal patterns of spontaneous muscle contractile activity due to injury to upper versus lower motor neurons.






Similar content being viewed by others
References
al-Amood WS, Lewis DM (1989) A comparison of the effects of denervation on the mechanical properties of rat and guinea-pig skeletal muscle. J Physiol 414:1–16
Batt J, Bain J, Goncalves J, Michalski B, Plant P, Fahnestock M, Woodgett J (2006) Differential gene expression profiling of short and long term denervated muscle. FASEB J 20:115–117
Bhasin S, Woodhouse L, Casaburi R, Singh AB, Bhasin D, Berman N, Chen X, Yarasheski KE, Magliano L, Dzekov C, Dzekov J, Bross R, Phillips J, Sinha-Hikim I, Shen R, Storer TW (2001) Testosterone dose–response relationships in healthy young men. Am J Physiol Endocrinol Metab 281:E1172–E1181
Bodine SC, Latres E, Baumhueter S, Lai VK, Nunez L, Clarke BA, Poueymirou WT, Panaro FJ, Na E, Dharmarajan K, Pan ZQ, Valenzuela DM, DeChiara TM, Stitt TN, Yancopoulos GD, Glass DJ (2001) Identification of ubiquitin ligases required for skeletal muscle atrophy. Science 294:1704–1708
Cai D, Frantz JD, Tawa NE Jr, Melendez PA, Oh BC, Lidov HG, Hasselgren PO, Frontera WR, Lee J, Glass DJ, Shoelson SE (2004) IKKbeta/NF-kappaB activation causes severe muscle wasting in mice. Cell 119:285–298
Caiozzo VJ, Wu YZ, Baker MJ, Crumley R (2004) Effects of denervation on cell cycle control in laryngeal muscle. Arch Otolaryngol Head Neck Surg 130:1056–1068
Carlson BM, Borisov AB, Dedkov EI, Khalyfa A, Kostrominova TY, Macpherson PC, Wang E, Faulkner JA (2002) Effects of long-term denervation on skeletal muscle in old rats. J Gerontol A Biol Sci Med Sci 57:B366–B374
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159
Clarke BA, Drujan D, Willis MS, Murphy LO, Corpina RA, Burova E, Rakhilin SV, Stitt TN, Patterson C, Latres E, Glass DJ (2007) The E3 ligase MuRF1 degrades myosin heavy chain protein in dexamethasone-treated skeletal muscle. Cell Metab 6:376–385
Czerwinski SM, Novakofski J, Bechtel PJ (1993) Modulation of IGF mRNA abundance during muscle denervation atrophy. Med Sci Sports Exerc 25:1005–1008
Ditunno JF, Little JW, Tessler A, Burns AS (2004) Spinal shock revisited: a four-phase model. Spinal Cord 42:383–395
Du J, Wang X, Miereles C, Bailey JL, Debigare R, Zheng B, Price SR, Mitch WE (2004) Activation of caspase-3 is an initial step triggering accelerated muscle proteolysis in catabolic conditions. J Clin Invest 113:115–123
Fareed MU, Evenson AR, Wei W, Menconi M, Poylin V, Petkova V, Pignol B, Hasselgren PO (2006) Treatment of rats with calpain inhibitors prevents sepsis-induced muscle proteolysis independent of atrogin-1/MAFbx and MuRF1 expression. Am J Physiol Regul Integr Comp Physiol 290:R1589–R1597
Ferrando AA, Sheffield-Moore M, Yeckel CW, Gilkison C, Jiang J, Achacosa A, Lieberman SA, Tipton K, Wolfe RR, Urban RJ (2002) Testosterone administration to older men improves muscle function: molecular and physiological mechanisms. Am J Physiol Endocrinol Metab 282:E601–E607
Flück M, Hoppeler H (2003) Molecular basis of skeletal muscle plasticity—from gene to form and function. Rev Physiol Biochem Pharmacol 146:159–216
Furuno K, Goodman MN, Goldberg AL (1990) Role of different proteolytic systems in the degradation of muscle proteins during denervation atrophy. J Biol Chem 265:8550–8557
Glazner GW, Ishii DN (1995) Insulinlike growth factor gene expression in rat muscle during reinnervation. Muscle Nerve 18:1433–1442
Glotzer M, Murray AW, Kirschner MW (1991) Cyclin is degraded by the ubiquitin pathway. Nature 349:132–138
Gregory CM, Vandenborne K, Huang HF, Ottenweller JE, Dudley GA (2003) Effects of testosterone replacement therapy on skeletal muscle after spinal cord injury. Spinal Cord 41:23–28
Greig CA, Hameed M, Young A, Goldspink G, Noble B (2006) Skeletal muscle IGF-I isoform expression in healthy women after isometric exercise. Growth Horm IGF Res 16:373–376
Haddad F, Roy RR, Zhong H, Edgerton VR, Baldwin KM (2003) Atrophy responses to muscle inactivity. II. Molecular markers of protein deficits. J Appl Physiol 95:791–802
Hafer-Macko CE, Yu S, Ryan AS, Ivey FM, Macko RF (2005) Elevated tumor necrosis factor-alpha in skeletal muscle after stroke. Stroke 36:2021–2023
Harris RL, Bobet J, Sanelli L, Bennett DJ (2006) Tail muscles become slow but fatigable in chronic sacral spinal rats with spasticity. J Neurophysiol 95:1124–1133
Heinemeyer W, Kleinschmidt JA, Saidowsky J, Escher C, Wolf DH (1991) Proteinase yscE, the yeast proteasome/multicatalytic multifunctional proteinase: mutants unravel its function in stress induced proteolysis and uncover its necessity for cell survival. EMBO J 10:555–562
Huang HF, Linsenmeyer TA, Li MT, Giglio W, Anesetti R, von Hagen J, Ottenweller JE, Serenas C, Pogach L (1995) Acute effects of spinal cord injury on the pituitary-testicular hormone axis and Sertoli cell functions: a time course study. J Androl 16:148–157
Hunter RB, Kandarian SC (2004) Disruption of either the Nfkb1 or the Bcl3 gene inhibits skeletal muscle atrophy. J Clin Invest 114:1504–1511
Hyatt JP, Roy RR, Baldwin KM, Wernig A, Edgerton VR (2006) Activity-unrelated neural control of myogenic factors in a slow muscle. Muscle Nerve 33:49–60
Hyatt JP, Roy RR, Baldwin KM, Edgerton VR (2003) Nerve activity-independent regulation of skeletal muscle atrophy: role of MyoD and myogenin in satellite cells and myonuclei. Am J Physiol Cell Physiol 285:C1161–C1173
Jiang GL, Zhang LY, Shen LY, Xu JG, Gu YD (2000) Fibrillation potential amplitude to quantitatively assess denervation muscle atrophy. Neuromuscul Disord 10:85–91
Jones SW, Hill RJ, Krasney PA, O'Conner B, Peirce N, Greenhaff PL (2004) Disuse atrophy and exercise rehabilitation in humans profoundly affects the expression of genes associated with the regulation of skeletal muscle mass. FASEB J 18:1025–1027
Kedar V, McDonough H, Arya R, Li HH, Rockman HA, Patterson C (2004) Muscle-specific RING finger 1 is a bona fide ubiquitin ligase that degrades cardiac troponin I. Proc Natl Acad Sci USA 101:18135–18140
Kitzman P (2005) Alteration in axial motoneuronal morphology in the spinal cord injured spastic rat. Exp Neurol 192:100–1008
Kostrominova TY, Dow DE, Dennis RG, Miller RA, Faulkner JA (2005) Comparison of gene expression of 2-mo denervated, 2-mo stimulated-denervated, and control rat skeletal muscles. Physiol Genomics 22:227–243
Krempler A, Brenig B (1999) Zinc finger proteins: watchdogs in muscle development. Mol Gen Genet 261:209–215
Lagirand-Cantaloube J, Offner N, Csibi A, Leibovitch MP, Batonnet-Pichon S, Tintignac LA, Segura CT, Leibovitch SA (2008) The initiation factor eIF3-f is a major target for atrogin1/MAFbx function in skeletal muscle atrophy. EMBO J 27:1266–1276
Lang CH, Huber D, Frost RA (2007) Burn-induced increase in atrogin-1 and MuRF-1 in skeletal muscle is glucocorticoid-independent but down regulated by IGF-I. Am J Physiol Regul Integr Comp Physiol 292:R328–R336
Latres E, Amini AR, Amini AA, Griffiths J, Martin FJ, Wei Y, Lin HC, Yancopoulos GD, Glass DJ (2005) Insulin-like growth factor-1 (IGF-1) inversely regulates atrophy-induced genes via the phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin (PI3K/AKT/mTOR) pathway. J Biol Chem 280:2737–2744
Lecker SH, Jagoe RT, Gilbert A, Gomes M, Baracos V, Bailey J, Price SR, Mitch WE, Goldberg AL (2004) Multiple types of skeletal muscle atrophy involve a common program of changes in gene expression. FASEB J 18:39–51
Li HH, Kedar V, Zhang C, McDonough H, Arya R, Wang DZ, Patterson C (2004) Atrogin-1/muscle atrophy F-box inhibits calcineurin-dependent cardiac hypertrophy by participating in an SCF ubiquitin ligase complex. J Clin Invest 114:1058–1071
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Mauras N, Hayes V, Welch S, Rini A, Helgeson K, Dokler M, Veldhuis JD, Urban RJ (1998) Testosterone deficiency in young men: marked alterations in whole body protein kinetics, strength, and adiposity. J Clin Endocrinol Metab 83:1886–1892
Midrio M (2006) The denervated muscle: facts and hypotheses. A historical review. Eur J Appl Physiol 98:1–21
Midrio M, Danieli-Betto D, Megighian A, Velussi C, Catani C, Carraro U (1992) Slow-to-fast transformation of denervated soleus muscle of the rat, in the presence of an antifibrillatory drug. Pflugers Arch 420:446–450
National Spinal Cord Injury Statistical Center (2006) Spinal cord injury: facts and figures at a glance. J Spinal Cord Medicine 29:89–90
Pickart CM (2001) Mechanisms underlying ubiquitination. Annu Rev Biochem 70:503–533
Sacheck JM, Hyatt JP, Raffaello A, Jagoe RT, Roy RR, Edgerton VR, Lecker SH, Goldberg AL (2007) Rapid disuse and denervation atrophy involve transcriptional changes similar to those of muscle wasting during systemic diseases. FASEB J 21:140–155
Sacheck JM, Ohtsuka A, McLary SC, Goldberg AL (2004) IGF-I stimulates muscle growth by suppressing protein breakdown and expression of atrophy-related ubiquitin ligases, atrogin-1 and MuRF1. Am J Physiol Endocrinol Metab 287:E591–E601
Salafsky B, Bell J, Prewitt MA (1968) Development of fibrillation potentials in denervated fast and slow skeletal muscle. Am J Physiol 215:637–643
Sakuma K, Watanabe K, Sano M, Uramoto I, Totsuka T (2000) Differential adaptation of growth and differentiation factor 8/myostatin, fibroblast growth factor 6 and leukemia inhibitory factor in overloaded, regenerating and denervated rat muscles. Biochim Biophys Acta 1497:77–88
Sandri M, Sandri C, Gilbert A, Skurk C, Calabria E, Picard A, Walsh K, Schiaffino S, Lecker SH, Goldberg AL (2004) Foxo transcription factors induce the atrophy-related ubiquitin ligase atrogin-1 and cause skeletal muscle atrophy. Cell 117:399–412
Scelsi R, Poggi P, Padovani R, Lotta S, Cairoli S, Saitta A (1986) Skeletal muscle changes following myelotomy in paraplegic patients. Paraplegia 24:250–259
Smith ML, Seo YR (2002) p53 regulation of DNA excision repair pathways. Mutagenesis 17:149–156
Stitt TN, Drujan D, Clarke BA, Panaro F, Timofeyva Y, Kline WO, Gonzalez M, Yancopoulos GD, Glass DJ (2004) The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol Cell Biol 14:395–403
Tews DS (2002) Apoptosis and muscle fibre loss in neuromuscular disorders. Neuromuscul Disord 12:613–622
Tintignac LA, Lagirand J, Batonnet S, Sirri V, Leibovitch MP, Leibovitch SA (2005) Degradation of MyoD mediated by the SCF (MAFbx) ubiquitin ligase. J Biol Chem 280:2847–2856
Traenckner EB, Wilk S, Baeuerle PA (1994) A proteasome inhibitor prevents activation of NF-kB and stabilizes a newly phosphorylated form of IkB-a that is still bound to NF-kB. EMBO J 13:5433–5441
Tsitouras PD, Zhong YG, Spungen AM, Bauman WA (1995) Serum testosterone and growth hormone/insulin-like growth factor-I in adults with spinal cord injury. Horm Metab Res 27:287–292
Wang X, Blagden C, Fan J, Nowak SJ, Taniuchi I, Littman DR, Burden SJ (2005) Runx1 prevents wasting, myofibrillar disorganization, and autophagy of skeletal muscle. Genes Dev 19:1715–1722
Wimalawansa SM, Chapa MT, Wei JN, Westlund KN, Quast MJ, Wimalawansa SJ (1999) Reversal of weightlessness-induced musculoskeletal losses with androgens: quantification by MRI. J Appl Physiol 86:1841–1846
Witt SH, Granzier H, Witt CC, Labeit S (2005) MURF-1 and MURF-2 target a specific subset of myofibrillar proteins redundantly: towards understanding MURF-dependent muscle ubiquitination. J Mol Biol 350:713–722
Wray CJ, Mammen JM, Hershko DD, Hasselgren PO (2003) Sepsis upregulates the gene expression of multiple ubiquitin ligases in skeletal muscle. Int J Biochem Cell Biol 35:698–705
Wu Y, Zhao W, Zhao J, Pan J, Wu Q, Zhang Y, Bauman WA, Cardozo CP (2007) Identification of androgen response elements in the insulin-like growth factor I upstream promoter. Endocrinology 148:2984–2993
Yates C, Charlesworth A, Allen SR, Reese NB, Skinner RD, Garcia-Rill E (2008) The onset of hyperreflexia in the rat following complete spinal cord transaction. Spinal Cord 46:798–803
Zeman RJ, Hirschman A, Hirschman ML, Guo G, Etlinger JD (1991) Clenbuterol, a beta 2-receptor agonist, reduces net bone loss in denervated hindlimbs. Am J Physiol 261:E285–E289
Zerbini LF, Libermann TA (2005) Life and death in cancer. GADD45 alpha and gamma are critical regulators of NF-kappaB mediated escape from programmed cell death. Cell Cycle 4:18–20
Zhao J, Zhang Y, Zhao W, Wu Y, Pan J, Bauman WA, Cardozo C (2008) Effects of nandrolone on denervation atrophy depend upon time after nerve transection. Muscle Nerve 37:42–49
Acknowledgements
This work was supported by Veterans Health Administration, Rehabilitation Research and Development Service (B4162C to WAB, B3347K to CC), and United Spinal Association (to WAB).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zeman, R.J., Zhao, J., Zhang, Y. et al. Differential skeletal muscle gene expression after upper or lower motor neuron transection. Pflugers Arch - Eur J Physiol 458, 525–535 (2009). https://doi.org/10.1007/s00424-009-0643-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-009-0643-5