Abstract
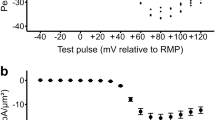
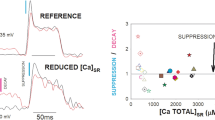
Time-dependent changes in sarcoplasmic reticulum (SR) Ca2+-handling and Na+-K+-ATPase activity, as assessed in vitro, were investigated in the superficial (GS) and deep regions (GD) of rat gastrocnemius muscles undergoing short-term (up to 30 min) electrical stimulation. There was a rapid and progressive loss of force output during the first 5 min of stimulation. For GS, significant depressions (P < 0.05) in SR Ca2+-uptake rate and Ca2+-ATPase activity were observed during only the first 1 min. No further reductions occurred with stimulation time. SR Ca2+-release rate was significantly (P < 0.05) decreased at 3 min. For GD, significant reductions (P < 0.05) in Ca2+-uptake rate, Ca2+-release rate and Ca2+-ATPase activity were manifested after 3, 5, and 5 min, respectively. A decay in Na+-K+-ATPase activity was found only in 1-min stimulated GD and 30-min stimulated GS. After 30 min, the depressed functions reverted to resting levels in GD but not in GS. The alterations in any variables examined were not parallel with changes in force output. These results suggest that, at least under the conditions used in this study, in vivo disruptions in cation regulation mediated by vigorous contractile activity would be attributable primarily to events other than structural alterations to the respective proteins.








Similar content being viewed by others
References
Allen D, Westerblad H (2004) Lactic acid—the latest performance-enhanced drug. Science 305:1112–1113
Allen DG (2004) Skeletal muscle function: role of ionic changes in fatigue, damage and disease. Clin Exp Pharmacol Physiol 31:485–493
Baker MA, Cerniglia GJ, Zaman A (1990) Microtiter plate assay for the measurement of glutathione and glutathione disulfide in large numbers of biological samples. Anal Biochem 190:360–365
de Ruiter CJ, de Haan A, Sargeant AJ (1995) Physiological characteristics of two extreme muscle compartments in gastrocnemius medialis of the anaesthetized rat. Acta Physiol Scand 153:313–324
Dutka TL, Cole L, Lamb GD (2005) Calcium-phosphate precipitation in the sarcoplasmic reticulum reduces action potential-mediated Ca2+ release mammalian skeletal muscle. Am J Physiol 289:C1502–C1512
Fraser SF, McKenna MJ (1998) Measurement of Na+, K+–ATPase activity in human skeletal muscle. Anal Biochem 258:63–67
Green HJ (2004) Membrane excitability, weakness, and fatigue. Can J Appl Physiol 29:291–307
Green HJ, Dusterhoft S, Dux L, Pette D (1992) Metabolite patterns related to exhaustion, recovery and transformation of chronically stimulated rabbit fast-twitch muscle. Pflügers Arch 420:359–366
Grynkiewicz G, Poenie M, Tsien RY (1985) A new generation of Ca2+ indicators with greatly improved fluorescent properties. J Biol Chem 260:3440–3450
Holloway GP, Green HJ, Tupling AR (2006) Differential effects of repetitive activity on sarcoplasmic reticulum responses in rat muscles of different oxidative potential. Am J Physiol 290:R393–R404
Inashima S, Matsunaga S, Yasuda T, Wada M (2003) Effect of endurance training and acute exercise on sarcoplasmic reticulum function in rat fast- and slow-twitch muscles. Eur J Appl Physiol 89:142–149
Klebl BM, Ayoub AT, Pette D (1998) Protein oxidation, tyrosine nitration, and inactivation of sarcoplasmic reticulum Ca2+–ATPase in low-frequency stimulated rabbit muscle. FEBS Lett 422:381–384
Laver DR (2006) Regulation of ryanodine receptors from skeletal and cardiac muscle during rest and excitation. Clin Exp Pharmacol Physiol 33:1107–1113
Leppik JA, Aughey RJ, Medved I, Fairweather I, Carey MF, McKenna MJ (2004) Prolonged exercise to fatigue in human impairs skeletal muscle Na+–K+–ATPase activity, sarcoplasmic reticulum Ca2+ release, and Ca2+ uptake. J Appl Physiol 97:1414–1423
Matsunaga S, Harmon S, Gohlsch B, Ohlendieck K, Pette D (2001) Inactivation of sarcoplasmic reticulum Ca2+-ATPase in low-frequency stimulated rat muscle. J Muscle Res Cell Motil 22:685–691
Matsunaga S, Inashima S, Yamada T, Watanabe H, Hazama T, Wada M (2003) Oxidation of sarcoplasmic reticulum Ca2+–ATPase induced by high-intensity exercise. Pflügers Arch 446:394–399
Matsunaga S, Yamada T, Mishima T, Sakamoto M, Sugiyama M, Wada M (2007) Effects of high-intensity training and acute exercise on in vitro function of rat sarcoplasmic reticulum. Eur J Appl Physiol 99:641–649
McKenna MJ, Medved I, Goodman CA, Brown MJ, Bjorlsten AR, Murphy KT, Petersen AC, Sostaric S, Gonh X (2006) N-acetylcysteine attenuates the decline in muscle Na+, K+-pump activity and delays fatigue during prolong exercise in humans. J Physiol (Lond) 576:279–288
Nielsen OB, Clausen T (2000) The Na+/K+-pump protects muscle excitability and contractility during exercise. Exerc Sport Sci Rev 28:159–164
Okamoto K, Wang W, Rounds J, Chambers EA, Jacobs DO (2001) ATP from glycolysis is required for normal sodium homeostasis in resting fast-twitch rodent skeletal muscle. Am J Physiol 281:E479–E488
Posterino GS, Cellini MA, Lamb GD (2003) Effects of oxidation and cytosolic redox conditions on excitation–contraction coupling in rat skeletal muscle. J Physiol (Lond) 547:807–823
Reid MB (2001) Plasticity in skeletal, cardiac, and smooth muscle: redox modulation of skeletal muscle contraction: what we know and what we don’t. J Appl Physiol 90:724–731
Sandiford SDE, Green HJ, Ouyang J (2005) Mechanisms underlying increases in rat soleus Na+–K+–ATPase activity by induced contractions. J Appl Physiol 99:2222–2232
Sen CK (1998) Glutathione: a key role in skeletal muscle metabolism. In: Reznick AZ, Packer L, Sen CK, Holloszy JO, Jackson MJ (eds) Oxidative stress in skeletal muscle. Birkhäuser, Berlin, pp 127–139
Senisterra GA, Huntley SA, Escaravage M, Sekhar KR, Freeman ML, Borrelli M, Lepock JR (1997) Destabilization of the Ca2+–ATPase of sarcoplasmic reticulum by thiol-specific heat shock inducers results in thermal denaturation at 37°C. Biochemistry 36:11002–11011
Simonides WS, van Hardeveld C (1990) An assay for sarcoplasmic reticulum Ca2+–ATPase activity in muscle homogenate. Anal Biochem 191:321–331
Sulova Z, Vyskocil F, Stankovicova T, Breier A (1998) Ca2+-induced inhibition of sodium pump: effects on energetic metabolism of mouse diaphragm tissue. Gen Physiol Biophys 17:271–283
Sun J, Xu L, Eu JP, Stamler JS, Meissner G (2001) Class of thiols that influence the activity of the skeletal muscle calcium release channel. J Biol Chem 276:15625–15630
Tavi P, Allen DG, Niomela P, Vuolteenaho O, Weckstrom M, Westerblad H (2003) Calmodulin kinase modulates Ca2+ release in mouse skeletal muscle. J Physiol (Lond) 551:5–12
Tupling AR (2004) The sarcoplasmic reticulum in muscle fatigue and disease: role of the sarco(endo)plasmic reticulum Ca2+–ATPase. Can J Appl Physiol 29:308–329
Vandenboom R (2004) The myofibrillar complex and fatigue. Can J Appl Physiol 29:330–356
Ward CW, Spangenburg EE, Diss LM, Williams JH (1998) Effects of varied fatigue protocols on sarcoplasmic reticulum uptake and release rates. Am J Physiol 275:R99–R104
Westerblad H, Allen DG (2003) Cellular mechanisms of skeletal muscle fatigue. Adv Exp Med Biol 538:563–570
Xu KY, Zweier JL, Becker LC (1997) Hydroxyl radical inhibits sarcoplasmic reticulum Ca2+–ATPase function by direct attack on the ATP binding site. Circ Res 80:76–81
Acknowledgement
Financial support for this study was received from Grants-Aid for Scientific Research of Japan (no.17300209; M. Wada).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mishima, T., Yamada, T., Sakamoto, M. et al. Time course of changes in in vitro sarcoplasmic reticulum Ca2+-handling and Na+-K+-ATPase activity during repetitive contractions. Pflugers Arch - Eur J Physiol 456, 601–609 (2008). https://doi.org/10.1007/s00424-007-0427-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-007-0427-8