Abstract
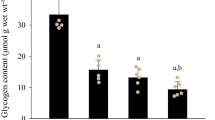
Rabbit fast-twitch tibialis anterior muscle was subjected to chronic low-frequency stimulation (10 Hz, 24 h/day). Measurements of the time course of changes in the concentration of metabolites of energy metabolism were performed in order to test the hypothesis whether or not alterations in the metabolite profile might represent possible signals for triggering muscle fibre type transformation. Most of the investigated metabolites displayed triphasic changes in response to persistently increased contractile activity. During the first 15 min of stimulation, drastic reductions were observed for adenosine triphoshate (ATP, 56%), phosphocreatine (PCr, 60%) and glycogen (76%), as well as 3- to 4-fold and 10-fold increases for glucose and lactate, respectively. This early metabolic perturbance coincided with a rapid reduction of isometric force. The next phase, extending to 4 days of stimulation, was characterized by a nearly complete recovery of ATP and PCr, and an overshoot in glycogen. The first signs of metabolic recovery were already detectable in 60-min-stimulated muscle when isometric force was still markedly depressed. These results demonstrated an impressive capability of the muscle to recover with ongoing stimulation from an initial, dramatic disturbance in energy metabolism. During the final phase, extending to 50 days, the metabolite profile approached that of a slow-twitch muscle with moderate reductions in total adenine nucleotides, ATP, total creatine, PCr and glycogen. A conspicuous result was the finding that, contrary to the recovery of most metabolites, the ratio of ATP to the product of free adenosine diphosphate and resting free inorganic phosphate was persistently depressed with ongoing stimulation. Therefore, the depressed phosphorylation potential of the adenylate system may be an important signal triggering muscle fibre type transformation.
Similar content being viewed by others
References
Chance B, Leigh Jr JS, Kent J, McCully K (1986) Metabolic control principles and 31P NMR. Fed Proc 45:2915–2920
Connett RJ (1985) In vivo glycolytic equilibria in dog gracilis muscle. J Biol Chem 260:3314–3320
Dudley GA, Terjung RL (1985) Influence of acidosis on AMP deaminase activity in contracting fast-twitch muscle. Am J Physiol 248:C43-C50
Dudley GA, Tullson PC, Terjung RL (1987) Influence of mitochondrial content on the sensitivity of respiratory control. J Biol Chem 262:9109–9114
Green HJ, Düsterhöft S, Dux L, Pette D (1990) Time dependent changes in metabolites of energy metabolism in low-frequency stimulated rabbit fast-twitch muscle. In: Pette D (ed) Dynamic state of muscle fibers. De Gruyter, Berlin, pp 617–628
Heilig A, Pette D (1988) Albumin in rabbit skeletal muscle. Origin, distribution and regulation by contractile activity. Eur J Biochem 171:503–508
Hellsten Y, Ahlborg G, Jensen-Urstad M, Sjödin B (1988) Indication of in vivo xanthine oxidase activity in human skeletal muscle during exercise. Acta Physiol Scand 134:159–160
Henriksson J, Chi MM-Y, Hintz CS, Young DA, Kaiser KK, Salmons S, Lowry OH (1986). Chronic stimulation of mammalian muscle: changes in enzymes of six metabolic pathways. Am J Physiol 251:C614-C632
Henriksson J, Salmons S, Chi MM-Y, Hintz CS, Lowry OH (1988) Chronic stimulation of mammalian muscle: changes in metabolite concentrations in individual fibers. Am J Physiol 255:C543-C551
Hintz CS, Chi MM-Y, Fell RD, Ivy JL, Kaiser KK, Lowry CV, Lowry OH (1982) Metabolite changes in individual rat muscle fibers during stimulation. Am J Physiol 242:C218-C228
Hudlická O, Brown, M, Cotter M, Smith M, Vrbová G (1977) The effect of long-term stimulation of fast muscles on their blood flow, metabolism and ability to withstand fatigue. Pflügers Arch 369:141–149
Hudlická O, Tyler KR, Aitman T (1980) The effect of long-term electrical stimulation on fuel uptake and performance in fast skeletal muscles. In: Pette D (ed) Plasticity of muscle. De Gruyter, Berlin, pp 401–408
Katz A, Broberg S, Sahlin K, Wahren J (1986) Leg glucose uptake during maximal dynamic exercise in humans. Am J Physiol 251:E65-E70
Kushmerick MJ, Meyer RA (1985) Chemical changes in rat leg muscle by phosphorus nuclear magnetic resonance. Am J Physiol 248:C542-C549
Lawson JW, Veech RL (1979) Effects of pH and free Mg2+ on the Keq of the creatine kinase reaction and other phosphate hydrolyses and phosphate transfer reactions. J Biol Chem 254:6528–6537
Le Rumeur E, Le Moyec L, Toulouse P, Le Bars R, Certaines JD de (1990) Muscle fatigue unrelated to phosphocreatine and pH: an “in vivo” 31-P NMR spectroscopy study. Muscle Nerve 13:438–444
Lindinger MI, Heigenhauser GJF, Spriet LL (1987) Effects of intense swimming and tetanic electrical stimulation on skeletal muscle ions and metabolites. J Appl Physiol 63:2331–2339
Lowry OH, Passonneau JV (1972) A flexible system of enzymatic analysis. Academic Press, New York
Maier A, Pette D (1987) The time course of glycogen depletion in single fibers of chronically stimulated rabbit fast-twitch muscle. Pflügers Arch 408:338–342
Maier A, Gambke B, Pette D (1986) Degeneration-regeneration as a mechanism contributing to the fast to slow conversion of chronically stimulated fast-twitch rabbit muscle. Cell Tissue Res 244:635–643.
Meyer RA, Terjung RL (1979) Differences in ammonia and adenylate metabolism in contracting slow and fast muscle. Am J Physiol 237:C1111-C1118
Meyer RA, Terjung RL (1980) AMP deamination and IMP reamination in working skeletal muscle. Am J Physiol 239:C32-C38
Pette D (1991) Effects of chronic electrostimulation on muscle gene expression. Semin Thorac Cardiovasc. Surg 3:101–105
Pette D, Smith ME, Staudte HW, Vrbová G (1973) Effects of long-term electrical stimulation on some contractile and metabolic characteristics of fast rabbit muscles. Pflügers Arch 338:257–272
Pette D, Ramirez BU, Müller W, Simon R, Exner GU, Hildebrand R (1975) Influence of intermittent long-term stimulation on contractile, histochemical and metabolic properties of fibre populations in fast and slow rabbit muscles. Pflügers Arch 361:1–7
Reichmann H, Hoppeler H, Mathieu-Costello O, Bergen F von, Pette D (1985) Biochemical and ultrastructural changes of skeletal muscle mitochondria after chronic electrical stimulation in rabbits. Pflügers Arch 404:1–9
Reitman J, Baldwin KM, Holloszy JO (1973) Intramuscular triglyceride utilization by red, white, and intermediate skeletal muscle and heart during exhausting exercise. Proc Soc Exp Biol Med 142:628–631
Sahlin K, Harris RC, Hultman E (1975) Creatine kinase equilibrium and lactate content compared with muscle pH in tissue samples obtained after isometric exercise. Biochem J 152:173–180
Sahlin K, Katz A, Henriksson J (1987) Redox state and lactate accumulation in human skeletal muscle during dynamic exercise. Biochem J 245:551–556
Stocchi V, Cucchiarini L, Magnani M, Chiarantini L, Palma P, Crescentini G (1985) Simultaneous extraction and reverse-phase high performance liquid chromatographic determination of adenine and pyridine nucleotides in human red blood cells. Anal Biochem 146:118–124
Takata S, Takai H, Ikata T, Miura I (1988) Observation of fatigue unrelated to gross energy reserve of skeletal muscle during tetanic contraction: an application of 31P MRS. Bio-chem Biophys Res Commun 157:225–231
Weber FE, Pette D (1990) Changes in free and bound forms and total amount of hexokinase isozyme II of rat muscle in response to contractile activity. Eur J Biochem 191:85–90
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Green, H.J., Düsterhöft, S., Dux, L. et al. Metabolite patterns related to exhaustion, recovery and transformation of chronically stimulated rabbit fast-twitch muscle. Pflügers Arch 420, 359–366 (1992). https://doi.org/10.1007/BF00374471
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00374471