Abstract
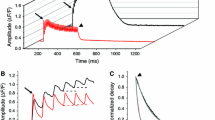
The effects of alterations in intracellular calcium homeostasis on surface membrane excitability were investigated in resting Rana temporaria sartorius muscle. This was prompted by initial results from a fatiguing stimulation protocol study that demonstrated a fibre subpopulation in which action potential generation in response to a standard 1.5 V electrical stimulus failed despite mean membrane potentials [E m, −69±2.3 mV (n=14)] compatible with spike firing in a control set of quiescent muscle fibres. Intracellular micro-electrode recordings showed a similar reversible loss of excitability, attributable to an increased threshold, despite only small (7.1±1.8 mV) positive changes in E m after approximately 60-min exposures to nominally 0 Ca2+ Ringer solutions in which Ca2+ was replaced by Mg2+. This effect was not reproduced by addition of Mg2+ to the Ringer solution and persisted under conditions of Cl− deprivation. The effects of three pharmacological agents, cyclopiazonic acid (CPA), caffeine and 4-chloro-m-cresol (4-CmC), each known to deplete store Ca2+ and increase cytosolic Ca2+ through contrasting mechanisms without influencing E m, were then investigated. All three agents produced a more rapid, but nevertheless still reversible, loss of membrane excitability than in 0 Ca2+ Ringer solution alone. This reduction in membrane excitability persisted in fibres studied in solutions containing a normal [Ca2+] following prior depletion of store Ca2+ using CPA- and 4-CmC-containing solutions. These novel findings suggest that sarcoplasmic reticulum Ca2+ content profoundly influences surface membrane excitability, thereby providing a potential mechanism by which spike firing fails in well-polarised fibres during fatigue.





Similar content being viewed by others
References
Allen DG, Kabbara AA, Westerblad H (2002) Muscle fatigue: role of intracellular calcium stores. Can J Appl Physiol 27:83–96
Allen DG, Lee JA, Westerblad H (1989) Intracellular calcium and tension during fatigue in isolated single muscle fibres from Xenopus laevis. J Physiol 415:433–458
Allen DG, Westerblad H (2001) Role of phosphate and calcium stores in muscle fatigue. J Physiol 536:657–665
Axelsson J, Thesleff S (1958) Activation of the contractile mechanism in striated muscle. Acta Physiol Scand 44:55–66
Baker PF, Hodgkin AL, Meves H (1964) The effect of diluting the internal solution on the electrical properties of a perfused giant axon. J Physiol 170:541–560
Baker PF, Hodgkin AL, Shaw TI (1962) The effects of changes in internal ionic concentrations on the electrical properties of perfused giant nerve fibres. J Physiol 164:355–374
Bianchi CP (1961) The effect of caffeine on radiocalcium movement in frog sartorius. J Gen Physiol 44:845–858
Bigland-Richie B (1981) EMG and fatigue of human voluntary and stimulated contractions. In: Porter R, Whelan J (eds) Human muscle fatigue: physiological mechanisms. Pitman, London, pp 130–156
Bigland-Richie B (1984) Muscle fatigue and the influence of changing neural drive. Clin Chest Med 5:21–34
Bigland-Richie B, Jones DA, Woods JJ (1979) Excitation frequency and muscle fatigue: electrical responses during human voluntary and stimulated contractions. Exp Neurol 64:414–427
Blinks JR, Rudel R, Taylor SR (1978) Calcium transients in isolated amphibian skeletal muscle fibres: detection with aequorin. J Physiol 277:291–323
Bruton JD, Westerblad H, Lannergren J (1996) Reversible depression of action potentials and force production in frog single muscle fibres by calmodulin-inhibitors. Acta Physiol Scand 156:441–446
Caputo C, Gimenez M (1967) Effects of external calcium deprivation on single muscle fibres. J Gen Physiol 50:2177–2195
Chawla S, Skepper JN, Huang CL-H (2002) Differential effects of sarcoplasmic reticular Ca2+-ATPase inhibition on charge movements and calcium transients in intact amphibian skeletal muscle fibres. J Physiol 539:869–882
Csernoch L, Pizarro G, Garcia J, Szucs G, Stefani E, Rios E (1992) Effects of calcium release from the sarcoplasmic reticulum on intramembrane charge movement in skeletal muscle. Adv Exp Med Biol 311:137–148
Curtis HJ, Cole KS (1942) Membrane resting and action potentials from the squid giant axon. J Cell Comp Physiol 19:135–144
De Luca CJ (1984) Myoelectrical manifestations of localized muscular fatigue in man. Crit Rev Biomed Eng 11:251–279
Delbono O, Obejero Paz CA, Muchnik S (1987) The effect of verapamil and Ca free solution on mechanical and electrical properties in fast twitch mammalian skeletal muscle. Acta Physiol Pharmacol Ther Latinoam 37:423–435
Duncan CJ, Smith JL (1980) Action of caffeine in initiating myofilament degradation and subdivision of mitochondria in mammalian skeletal muscle. Comp Biol Physiol 65C:143–145
Edman KAP, Grieve DW (1964) On the role of calcium in the excitation–contraction process of frog sartorius muscle. J Physiol 170:138–152
Feng J, Somlyo AV, Somlyo AP (2004) A system for acquiring simultaneous electron energy-loss and X-ray spectrum-images. J Microsc 215:92–99
Fitts RH (1994) Cellular mechanisms of muscle fatigue. Physiol Rev 74:49–94
Frank GB (1962) Utilization of bound calcium in the action of caffeine and certain multivalent cations on skeletal muscle. J Physiol 163:254–286
Frank GB (1982) The effects of reducing the extracellular calcium concentration on the twitch in isolated frog’s skeletal muscle fibres. Jpn J Physiol 32:589–608
Grabowski W, Lobsiger EA, Luttgau HC (1972) The effect of repetitive stimulation at low frequencies upon the electrical and mechanical activity of single muscle fibres. Pflugers Arch 334:222–239
Gyorke S (1993) Effects of repeated tetanic stimulation on excitation–contraction coupling in cut muscle fibres of the frog. J Physiol 464:699–710
Hain J, Nath S, Mayrleitner M, Fleischer S, Schlinder H (1994) Phosphorylation modulates the function of the calcium release channel of sarcoplasmic reticulum from skeletal muscle. Biophys J 67:1823–1833
Hanson J (1974) The effects of repetitive stimulation on the action potential and twitch of rat muscle. Acta Physiol Scand 90:387–400
Herrmann-Frank A, Richter M, Sarkozi S, Mohr U, Lehmann-Horn F (1996) 4-Chloro-m-cresol, a potent and specific activator of the skeletal muscle ryanodine receptor. Biochim Biophys Acta 1289:31–40
Hodgkin AL, Huxley AF (1952) A quantitative description of membrane current and its application to conduction and excitation in nerve. J Physiol 117:500–544
Hong SJ, Liang, H-C, Shen C-J (2003) Dependence of cyclopiazonic-acid-induced muscle contractures on extracellular Ca2+. Can J Physiol Pharmacol 81:1101–1109
Huang CL-H (1998) The influence of caffeine on intramembrane charge movements in intact frog striated muscle. J Physiol 512:707–721
Ichikawa M, Urayama M, Matsumoto G (1991) Anticalmodulin drugs block the sodium gating current of squid giant axons. J Membr Biol 120:211–222
Jenden DJ, Reger JF (1963) The role of resting potential changes in the contractile failure of frog sartorius muscles during calcium deprivation. J Physiol 169:889–901
Jones DA (1981) Muscle fatigue due to changes beyond the neuromuscular junction. In: Porter R, Whelan J (eds) Human muscle fatigue: physiological mechanisms. Pitman, London, pp 178–196
Jones DA, Bigland-Richie B (1986) Exercise-induced muscle fatigue. Electrical and contractile changes in muscle fatigue. In: Saltin B (ed) International series on sport sciences. Biochemistry of exercise VI. Human Kinetics, Champaign, pp 377–392
Kabbara AA, Allen DG (1999) Measurement of sarcoplasmic reticulum Ca2+ content in intact amphibian skeletal muscle fibres with 4-chloro-m-cresol. Cell Calcium 25:227–235
Kabbara AA, Allen DG (1999) The role of calcium stores in fatigue of isolated single muscle fibres from the cane toad. J Physiol 519:169–176
Kurebayashi N, Ogawa Y (1991) Discrimination of Ca2+-ATPase activity of the sarcoplasmic reticulum from actomyosin-type ATPase activity of myofibrils in skinned mammalian skeletal muscle fibres: distinct effects of cyclopiazonic acid on the two ATPase activities. J Muscle Res Cell Motil 12:355–365
Kurebayashi N, Ogawa Y (2001) Depletion of Ca2+ in the sarcoplasmic reticulum stimulates Ca2+ entry into mouse skeletal muscle fibres. J Physiol 533:185–199
Lannergren J, Westerblad H (1986) Force and membrane potential during and after fatiguing, continuous high-frequency stimulation of single Xenopus muscle fibres. Acta Physiol Scand 128:359–368
Lannergren J, Westerblad H (1987) Action potential fatigue in single skeletal muscle fibres of Xenopus. Acta Physiol Scand 129:311–318
Launikonis BS, Barnes M, Stephenson DG (2003) Identification of the coupling between skeletal muscle store-operated Ca2+ entry and the inositol triphosphate receptor. Proc Natl Acad Sci U S A 100:2941–2944
Luttgau HC (1963) The action of calcium ions on potassium contractures of single muscle fibres. J Physiol 168:679–697
Luttgau HC, Oetliker H (1968) The action of caffeine on the activation of the contractile mechanism in striated muscle fibres. J Physiol 194:51–74
Luttgau HC, Spiecker W (1979) The effects of calcium deprivation upon mechanical and electrophysiological parameters in skeletal muscle fibres of the frog. J Physiol 296:411–429
Meme W, Huchet-Cadiou C, Leoty C (1998) Cyclopiazonic acid-induced changes in the contraction and Ca2+ transient of frog fast-twitch skeletal muscle. Am J Physiol 274:C253–C261
Metzger JM, Fitts RH (1986) Fatigue from high- and low-frequency muscle stimulation: role of sarcolemma action potentials. Exp Neurol 93:320–333
Morgan DL, Claflin DR, Julian FJ (1997) The relationship between tension and slowly varying intracellular calcium concentration in intact frog skeletal muscle. J Physiol 500:177–192
Putney JW Jr (1986) A model for receptor-regulated calcium entry. Cell Calcium 7:1–12
Putney JW Jr (1997) Capacitative calcium entry. Landes, Austin
Putney JW Jr, Broad LM, Braun F-J, Lievremont J-P, Bird G (2001) Mechanisms of capacitative calcium entry. J Cell Sci 114:2223–2229
Rich MM, Pinter MJ (2001) Sodium channel inactivation in an animal model of acute quadriplegic myopathy. Ann Neurol 50:26–33
Rousseau E, Ladine J, Liu Q, Meissner G (1988) Activation of the Ca2+ release channel of skeletal muscle sarcoplasmic reticulum by caffeine and related compounds. Arch Biochem Biophys 267:75–86
Ruff RL (1996) Sodium channel slow inactivation and the distribution of sodium channels on skeletal muscle fibres enable the performance properties of different skeletal muscle fibre types. Acta Physiol Scand 156:159–168
Seidler NW, Jona I, Vegh M, Martonosi A (1989) Cyclopiazonic acid is a specific inhibitor of the Ca2+-ATPase of sarcoplasmic reticulum. J Biol Chem 264:17816–17823
Shah VN, Wingo TL, Weiss KL, Williams CK, Balser JR, Chazin WJ (2006) Calcium-dependent regulation of the voltage-gated sodium channel hH1: intrinsic and extrinsic sensors use a common molecular switch. Proc Natl Acad Sci U S A 103:3592–3597
Smith JS, Rousseau E, Meissner G (1989) Calmodulin modulation of single sarcoplasmic reticulum Ca2+-release channels from cardiac and skeletal muscle. Circ Res 64:352–359
Stefani E, Chiarandini DJ (1973) Skeletal muscle: dependence of potassium contractures on extracellular calcium. Pflugers Arch 343:143–150
Usher-Smith JA, Fraser JA, Bailey PSJ, Griffin JL, Huang CL-H (2006) The influence of intracellular lactate and H+ on cell volume in amphibian skeletal muscle. J Physiol 573:799–818
Usher-Smith JA, Skepper JN, Fraser JA, Huang CL-H (2006) Effect of repetitive stimulation on cell volume and its relationship to membrane potential in amphibian skeletal muscle. Pflugers Arch 452:231–239
Westerblad H, Allen DG (1991) Changes of myoplasmic calcium concentration during fatigue in single mouse muscle fibres. J Gen Physiol 98:615–635
Westerblad H, Andrade FH, Islam MS (1998) Effects of ryanodine receptor agonist 4-chloro-m-cresol on myoplasmic free Ca2+ concentration and force of contraction in mouse skeletal muscle. Cell Calcium 24:105–115
Westerblad H, Lannergren J (1986) Force and membrane potential during and after fatiguing, intermittent tetanic stimulation of single Xenopus muscle fibres. Acta Physiol Scand 128:369–378
Westerblad H, Lannergren J (1988) The relation between force and intracellular pH in fatigued, single Xenopus muscle fibres. Acta Physiol Scand 133:83–89
Westerblad H, Lee JA, Lamb AG, Bolsover SR, Allen DG (1990) Spatial gradients of intracellular calcium in skeletal muscle during fatigue. Pflugers Arch 415:734–740
Westerblad H, Lee JA, Lannergren J, Allen DG (1991) Cellular mechanisms of fatigue in skeletal muscle. Am J Physiol 261:C195–C209
Yoshioka T, Shirota T, Tazoe T, Tanaka O, Kimura M, Yamashita-Goto K (1995) A-band movement and junctional gap dissociation during caffeine-induced contracture of skeletal muscle fibres. Tokai J Exp Clin Med 20:99–108
Acknowledgments
We thank Paul Frost for skilled assistance. C.L.-H.H. thanks the Medical Research Council, the Wellcome Trust and the British Heart Foundation for generous support. J.A.U-S. thanks Astra Zeneca and acknowledges additional support from the James Baird Fund. J.A.F. thanks Gonville and Caius College for a research fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors Usher-Smith and Xu were equal contributors to this paper.
Rights and permissions
About this article
Cite this article
Usher-Smith, J.A., Xu, W., Fraser, J.A. et al. Alterations in calcium homeostasis reduce membrane excitability in amphibian skeletal muscle. Pflugers Arch - Eur J Physiol 453, 211–221 (2006). https://doi.org/10.1007/s00424-006-0132-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-006-0132-z