Abstract
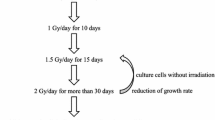
Radiotherapy (RT) is one of the major modalities for the treatment of human cancer and has been established as an excellent local treatment for malignant tumors. However, the existence of radioresistant cells remains one of the most critical obstacles in RT. To know the characteristics of radioresistant cells, clinically relevant radioresistant (CRR) cell lines were established. CRR cells can continue to proliferate in vitro and in vivo after exposure to 2 Gy/day of X-rays for more than 30 days. Daily microscopic observation of the irradiated CRR cells has indicated that the increase in cell death is not observed within 7 days of irradiation with 10 Gy of X-rays, suggesting that cell death is involved in cellular radioresistance. Radiation-induced regulated cell death (RCD) can be classified into three categories: apoptosis, autophagy-dependent cell death and necrosis (necroptosis). This review focuses on an aspect of radiation-induced RCD that has often been neglected: the manner in which the cells are destroyed. In many studies, apoptosis is considered the primary mode of RCD in irradiated cancer cells; however, it is necessary to consider necrosis or necroptosis as one of the modes of radiation-induced RCD.





Similar content being viewed by others
References
Abend M (2003) Reasons to reconsider the significance of apoptosis for cancer therapy. Int J Radiat Biol 79:927–941. https://doi.org/10.1080/09553000310001632958
Balcer-Kubiczek EK (2012) Apoptosis in radiation therapy: a double-edged sword. Exp Oncol 34:277–285
Brown JM, Attardi LD (2005) The role of apoptosis in cancer development and treatment response. Nat Rev Cancer 5:231–237. https://doi.org/10.1038/nrc1560
Castedo M, Perfettini JL, Roumier T, Andreau K, Medema R, Kroemer G (2004) Cell death by mitotic catastrophe: a molecular definition. Oncogene 23:2825–2837. https://doi.org/10.1038/sj.onc.1207528
Chang JT et al (2007) Differentially expressed genes in radioresistant nasopharyngeal cancer cells: gp96 and GDF15. Mol Cancer Ther 6:2271–2279. https://doi.org/10.1158/1535-7163.MCT-06-0801
Chang L, Graham PH, Ni J, Hao J, Bucci J, Cozzi PJ, Li Y (2015) Targeting PI3K/Akt/mTOR signaling pathway in the treatment of prostate cancer radioresistance. Crit Rev Oncol Hematol 96:507–517. https://doi.org/10.1016/j.critrevonc.2015.07.005
Chen Z, Wang B, Yu F, Chen Q, Tian Y, Ma S, Liu X (2016) The roles of mitochondria in radiation-induced autophagic cell death in cervical cancer cells. Tumour Biol 37:4083–4091. https://doi.org/10.1007/s13277-015-4190-8
Chipuk JE, Green DR (2005) Do inducers of apoptosis trigger caspase-independent cell death? Nat Rev Mol Cell Biol 6:268–275. https://doi.org/10.1038/nrm223910.1038/nrm1573
Clarke PG (1990) Developmental cell death: morphological diversity and multiple mechanisms. Anat Embryol (Berl) 181:195–213
Dayal R, Singh A, Pandey A, Mishra KP (2014) Reactive oxygen species as mediator of tumor radiosensitivity. J Cancer Res Ther 10:811–818. https://doi.org/10.4103/0973-1482.146073
Declercq W, Vanden Berghe T, Vandenabeele P (2009) RIP kinases at the crossroads of cell death and survival. Cell 138:229–232. https://doi.org/10.1016/j.cell.2009.07.006
Degterev A et al (2008) Identification of RIP1 kinase as a specific cellular target of necrostatins. Nat Chem Biol 4:313–321. https://doi.org/10.1038/nchembio.83
Dewey WC, Ling CC, Meyn RE (1995) Radiation-induced apoptosis: relevance to radiotherapy. Int J Radiat Oncol Biol Phys 33:781–796. https://doi.org/10.1016/0360-3016(95)00214-8
Duque-Parra JE (2005) Note on the origin and history of the term “apoptosis”. Anat Rec B New Anat 283:2–4. https://doi.org/10.1002/ar.b.20047
Elmore S (2007) Apoptosis: a review of programmed cell death. Toxicol Pathol 35:495–516. https://doi.org/10.1080/01926230701320337
Eriksson D, Stigbrand T (2010) Radiation-induced cell death mechanisms. Tumour Biol 31:363–372. https://doi.org/10.1007/s13277-010-0042-8
Fukumoto M (2014) Radiation pathology: from thorotrast to the future beyond radioresistance. Pathol Int 64:251–262. https://doi.org/10.1111/pin.12170
Fulda S (2013) The mechanism of necroptosis in normal and cancer cells. Cancer Biol Ther 14:999–1004. https://doi.org/10.4161/cbt.26428
Fulda S (2014) Therapeutic exploitation of necroptosis for cancer therapy. Semin Cell Dev Biol 35:51–56. https://doi.org/10.1016/j.semcdb.2014.07.002
Galluzzi L, Kroemer G (2008) Necroptosis: a specialized pathway of programmed necrosis. Cell 135:1161–1163. https://doi.org/10.1016/j.cell.2008.12.004
Galluzzi L et al (2012) Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ 19:107–120. https://doi.org/10.1038/cdd.2011.96
Galluzzi L, Bravo-San Pedro JM, Kepp O, Kroemer G (2016) Regulated cell death and adaptive stress responses. Cell Mol Life Sci 73:2405–2410. https://doi.org/10.1007/s00018-016-2209-y
Galluzzi L et al (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ 25:486–541. https://doi.org/10.1038/s41418-017-0012-4
Giampietri C, Starace D, Petrungaro S, Filippini A, Ziparo E (2014) Necroptosis: molecular signalling and translational implications. Int J Cell Biol 2014:490275. https://doi.org/10.1155/2014/490275
Gozuacik D, Kimchi A (2004) Autophagy as a cell death and tumor suppressor mechanism. Oncogene 23:2891–2906. https://doi.org/10.1038/sj.onc.1207521
Guo WF, Lin RX, Huang J, Zhou Z, Yang J, Guo GZ, Wang SQ (2005) Identification of differentially expressed genes contributing to radioresistance in lung cancer cells using microarray analysis. Radiat Res 164:27–35
Hasegawa M, Wilson G, Russell LD, Meistrich ML (1997) Radiation-induced cell death in the mouse testis: relationship to apoptosis. Radiat Res 147:457–467
Huang G, Wang H, Yang LX (2010) Enhancement of radiation-induced DNA damage and inhibition of its repair by a novel camptothecin analog. Anticancer Res 30:937–944
Ishigami T et al (2007) Genes and molecular pathways related to radioresistance of oral squamous cell carcinoma cells. Int J Cancer 120:2262–2270. https://doi.org/10.1002/ijc.22561
Janku F, McConkey DJ, Hong DS, Kurzrock R (2011) Autophagy as a target for anticancer therapy. Nat Rev Clin Oncol 8:528–539. https://doi.org/10.1038/nrclinonc.2011.71
Jiang YG, Peng Y, Koussougbo KS (2011) Necroptosis: a novel therapeutic target for glioblastoma. Med Hypotheses 76:350–352. https://doi.org/10.1016/j.mehy.2010.10.037
Jo GH et al (2015) Radiation-induced autophagy contributes to cell death and induces apoptosis partly in malignant glioma cells. Cancer Res Treat 47:221–241. https://doi.org/10.4143/crt.2013.159
Kerr JF, Wyllie AH, Currie AR (1972) Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer 26:239–257
Kerr JF, Winterford CM, Harmon BV (1994) Apoptosis. Its significance in cancer and cancer therapy. Cancer 73:2013–2026
Kim BM et al (2015) Therapeutic implications for overcoming radiation resistance in cancer therapy. Int J Mol Sci 16:26880–26913. https://doi.org/10.3390/ijms161125991
Kroemer G et al (2009) Classification of cell death: recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ 16:3–11. https://doi.org/10.1038/cdd.2008.150
Kuwahara Y et al (2009) Clinically relevant radioresistant cells efficiently repair DNA double-strand breaks induced by X-rays. Cancer Sci 100:747–752. https://doi.org/10.1111/j.1349-7006.2009.01082.x
Kuwahara Y et al (2011) Enhancement of autophagy is a potential modality for tumors refractory to radiotherapy. Cell Death Dis 2:e177. https://doi.org/10.1038/cddis.2011.56
Kuwahara Y et al (2017) Clinically relevant radioresistant cell line: a simple model to understand cancer radioresistance. Med Mol Morphol 50:195–204. https://doi.org/10.1007/s00795-017-0171-x
Liao EC et al (2014) Radiation induces senescence and a bystander effect through metabolic alterations. Cell Death Dis 5:e1255. https://doi.org/10.1038/cddis.2014.220
Lomax ME, Folkes LK, O’Neill P (2013) Biological consequences of radiation-induced DNA damage: relevance to radiotherapy. Clin Oncol (R Coll Radiol) 25:578–585. https://doi.org/10.1016/j.clon.2013.06.007
Mansilla S, Priebe W, Portugal J (2006) Mitotic catastrophe results in cell death by caspase-dependent and caspase-independent mechanisms. Cell Cycle 5:53–60. https://doi.org/10.4161/cc.5.1.2267
Meier P, Finch A, Evan G (2000) Apoptosis in development. Nature 407:796–801. https://doi.org/10.1038/35037734
Mirzaie-Joniani H, Eriksson D, Johansson A, Lofroth PO, Johansson L, Ahlstrom KR, Stigbrand T (2002a) Apoptosis in HeLa Hep2 cells is induced by low-dose, low-dose-rate radiation. Radiat Res 158:634–640
Mirzaie-Joniani H, Eriksson D, Sheikholvaezin A, Johansson A, Lofroth PO, Johansson L, Stigbrand T (2002b) Apoptosis induced by low-dose and low-dose-rate radiation. Cancer 94:1210–1214
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873. https://doi.org/10.1101/gad.1599207
Moretti L, Yang ES, Kim KW, Lu B (2007) Autophagy signaling in cancer and its potential as novel target to improve anticancer therapy. Drug Resist Updat 10:135–143. https://doi.org/10.1016/j.drup.2007.05.001
Motyl T, Gajewska M, Zarzynska J, Sobolewska A, Gajkowska B (2007) Regulation of autophagy in bovine mammary epithelial cells. Autophagy 3:484–486
Nehs MA et al (2011) Necroptosis is a novel mechanism of radiation-induced cell death in anaplastic thyroid and adrenocortical cancers. Surgery 150:1032–1039. https://doi.org/10.1016/j.surg.2011.09.012
Ogawa K, Murayama S, Mori M (2007) Predicting the tumor response to radiotherapy using microarray analysis. Oncol Rep 18:1243–1248
Ohnishi K, Inaba H, Yasumoto J, Yuki K, Takahashi A, Ohnishi T (2004) C-terminal peptides of p53 molecules enhance radiation-induced apoptosis in human mutant p53 cancer cells. Apoptosis 9:591–597. https://doi.org/10.1023/B:APPT.0000038044.40337.35
Ohyama H, Yamada T, Ohkawa A, Watanabe I (1985) Radiation-induced formation of apoptotic bodies in rat thymus. Radiat Res 101:123–130
Ouyang L, Shi Z, Zhao S, Wang FT, Zhou TT, Liu B, Bao JK (2012) Programmed cell death pathways in cancer: a review of apoptosis, autophagy and programmed necrosis. Cell Prolif 45:487–498. https://doi.org/10.1111/j.1365-2184.2012.00845.x
Pinar B et al (2010) Radiation induced apoptosis and initial DNA damage are inversely related in locally advanced breast cancer patients. Radiat Oncol 5:85. https://doi.org/10.1186/1748-717X-5-85
Potten CS, Grant HK (1998) The relationship between ionizing radiation-induced apoptosis and stem cells in the small and large intestine. Br J Cancer 78:993–1003
Radford IR, Murphy TK (1994) Radiation response of mouse lymphoid and myeloid cell lines. Part III. Different signals can lead to apoptosis and may influence sensitivity to killing by DNA double-strand breakage. Int J Radiat Biol 65:229–239
Rainaldi G, Ferrante A, Indovina PL, Santini MT (2003) Induction of apoptosis or necrosis by ionizing radiation is dose-dependent in MG-63 osteosarcoma multicellular spheroids. Anticancer Res 23:2505–2518
Roninson IB, Broude EV, Chang BD (2001) If not apoptosis, then what? Treatment-induced senescence and mitotic catastrophe in tumor cells. Drug Resist Updat 4:303–313. https://doi.org/10.1054/drup.2001.0213
Sabin RJ, Anderson RM (2011) Cellular Senescence—its role in cancer and the response to ionizing radiation. Genome Integr 2:7. https://doi.org/10.1186/2041-9414-2-7
Sato K et al (2007) Autophagy is activated in colorectal cancer cells and contributes to the tolerance to nutrient deprivation. Cancer Res 67:9677–9684. https://doi.org/10.1158/0008-5472.CAN-07-1462
Shimura T et al (2010) Acquired radioresistance of human tumor cells by DNA-PK/AKT/GSK3beta-mediated cyclin D1 overexpression. Oncogene 29:4826–4837. https://doi.org/10.1038/onc.2010.238
Shinohara K, Nakano H (1993) Interphase death and reproductive death in X-irradiated MOLT-4 cells. Radiat Res 135:197–205
Shinomiya N (2001) New concepts in radiation-induced apoptosis: ‘premitotic apoptosis’ and ‘postmitotic apoptosis’. J Cell Mol Med 5:240–253
Silke J, Rickard JA, Gerlic M (2015) The diverse role of RIP kinases in necroptosis and inflammation. Nat Immunol 16:689–697. https://doi.org/10.1038/ni.3206
Su Z, Yang Z, Xie L, DeWitt JP, Chen Y (2016) Cancer therapy in the necroptosis era. Cell Death Differ 23:748–756. https://doi.org/10.1038/cdd.2016.8
Sun H, Yang S, Li J, Zhang Y, Gao D, Zhao S (2016) Caspase-independent cell death mediated by apoptosis-inducing factor (AIF) nuclear translocation is involved in ionizing radiation induced HepG2 cell death. Biochem Biophys Res Commun 472:137–143. https://doi.org/10.1016/j.bbrc.2016.02.082
Tait SW, Green DR (2008) Caspase-independent cell death: leaving the set without the final cut. Oncogene 27:6452–6461. https://doi.org/10.1038/onc.2008.311
Tsuboi Y et al (2009) Induction of autophagic cell death and radiosensitization by the pharmacological inhibition of nuclear factor-kappa B activation in human glioma cell lines. J Neurosurg 110:594–604. https://doi.org/10.3171/2008.8.JNS17648
Tsujimoto Y, Shimizu S (2005) Another way to die: autophagic programmed cell death. Cell Death Differ 12 Suppl 2:1528–1534. https://doi.org/10.1038/sj.cdd.4401777
Vakkila J, Lotze MT (2004) Inflammation and necrosis promote tumour growth. Nat Rev Immunol 4:641–648. https://doi.org/10.1038/nri1415
Vandenabeele P, Galluzzi L, Vanden Berghe T, Kroemer G (2010) Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat Rev Mol Cell Biol 11:700–714. https://doi.org/10.1038/nrm2970
Walsh CM (2014) Grand challenges in cell death and survival: apoptosis vs. necroptosis. Front Cell Dev Biol 2:3. https://doi.org/10.3389/fcell.2014.00003
Wu W, Liu P, Li J (2012) Necroptosis: an emerging form of programmed cell death. Crit Rev Oncol Hematol 82:249–258. https://doi.org/10.1016/j.critrevonc.2011.08.004
Acknowledgements
This work is supported by JSPS KAKENHI Grant Numbers 24659174, 25340026, 26670184, 18K07727, 16K11513 and 16K15571.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuwahara, Y., Tomita, K., Urushihara, Y. et al. Association between radiation-induced cell death and clinically relevant radioresistance. Histochem Cell Biol 150, 649–659 (2018). https://doi.org/10.1007/s00418-018-1728-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1728-z