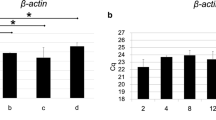
Abstract
Mammalian tooth development is regulated by paracrine signal molecules of several conserved family interactions between epithelium and mesenchyme. The expression patterns and regulative roles of FGF signaling have been extensively studied in the mouse odontogenesis; however, that is not well known in human tooth development. In order to unveil the molecular mechanisms that regulate human tooth morphogenesis, we examined the expression patterns of the critical molecules involved in FGF signaling pathway in the developing human tooth germ by in situ hybridization, immunohistochemistry, and real-time RT-PCR, including FGF ligands, receptors, and intracellular transducer. We found overlapping but distinct expression pattern of FGF ligands and receptors in the different stages and components. Expression of FGF4, FGF7, FGF8, and FGF9 persists widespread in human tooth mesenchyme, which is quite different to that of in mouse. FGFR1 may be the major receptor in regulate mechanisms of FGF signals in human tooth development. Real-time RT-PCR indeed confirmed the results of in situ hybridization. Results of K-Ras, p-ERK1/2, p-p38, p-JNK, and p-PDK1 expression reveal spatial and temporal patterns of FGF signaling during morphogenesis and organogenesis of human tooth germ. Activity of the FGF signaling transducer protein in human tooth germ was much higher than that of in mouse. Our results provided important FGF singling information in the developing process, pinpoint to the domains where the downstream target genes of FGF signaling can be sought, and enlightened our knowledge about the nature of FGF signaling in human tooth germ.








Similar content being viewed by others
References
Adachi-Yamada T et al (1999) p38 mitogen-activated protein kinase can be involved in transforming growth factor β superfamily signal transduction in Drosophila wing morphogenesis. Mol Cell Biol 19:2322–2329
Amand TRS et al (2000) Antagonistic signals between BMP4 and FGF8 define the expression of Pitx1 and Pitx2 in mouse tooth-forming anlage. Dev Biol 217:323–332
Bhowmick NA, Zent R, Ghiassi M, McDonnell M, Moses HL (2001) Integrin β1 signaling is necessary for transforming growth factor-β activation of p38MAPK and epithelial plasticity. J Biol Chem 276:46707–46713
Castellano E, Downward J (2011) RAS interaction with PI3K more than just another effector pathway. Genes Cancer 2:261–274
Chang L, Karin M (2001) Mammalian MAP kinase signalling cascades. Nature 410:37–40
Charles C, Lazzari V, Tafforeau P, Schimmang T, Tekin M, Klein O, Viriot L (2009) Modulation of Fgf3 dosage in mouse and men mirrors evolution of mammalian dentition. Proc Natl Acad Sci 106:22364–22368
Cho KW, Cho SW, Lee JM, Lee MJ, Gang HS, Jung HS (2008) Expression of phosphorylated forms of ERK, MEK, PTEN and PI3K in mouse oral development. Gene Expr Patterns 8(4):284–290
Corson LB, Yamanaka Y, Lai K-MV, Rossant J (2003) Spatial and temporal patterns of ERK signaling during mouse embryogenesis. Development 130:4527–4537
De Moerlooze L, Spencer-Dene B, Revest J, Hajihosseini M, Rosewell I, Dickson C (2000) An important role for the IIIb isoform of fibroblast growth factor receptor 2 (FGFR2) in mesenchymal-epithelial signalling during mouse organogenesis. Development 127:483–492
Dong X, Shen B, Ruan N, Guan Z, Zhang Y, Chen Y, Hu X (2014) Expression patterns of genes critical for BMP signaling pathway in developing human primary tooth germs. Histochem Cell Biol 142:657–665
Engel ME, McDonnell MA, Law BK, Moses HL (1999) Interdependent SMAD and JNK signaling in transforming growth factor-β-mediated transcription. J Biol Chem 274:37413–37420
Eswarakumar V, Lax I, Schlessinger J (2005) Cellular signaling by fibroblast growth factor receptors. Cytokine Growth Factor Rev 16:139–149
Goetz R, Mohammadi M (2013) Exploring mechanisms of FGF signalling through the lens of structural biology. Nat Rev Mol Cell Biol 14:166–180
Harada H et al (2002) FGF10 maintains stem cell compartment in developing mouse incisors. Development 129:1533–1541
Hartsough MT, Mulder KM (1995) Transforming growth factor β activation of p44mapk in proliferating cultures of epithelial cells. J Biol Chem 270:7117–7124
Hu X, Zhang S, Chen G, Lin C, Huang Z, Chen Y, Zhang Y (2013) Expression of SHH signaling molecules in the developing human primary dentition. BMC Dev Biol 13:11
Hu X, Xu S, Lin C, Zhang L, Chen Y, Zhang Y (2014) Precise chronology of differentiation of developing human primary dentition. Histochem Cell Biol 141:221–227
Huang Z, Hu X, Lin C, Chen S, Huang F, Zhang Y (2014) Genome-wide analysis of gene expression in human embryonic tooth germ. J Mol Histol 45:609–617
Itoh N (2007) The Fgf families in humans, mice, and zebrafish: their evolutional processes and roles in development, metabolism, and disease. Biol Pharm Bull 30:1819–1825
Itoh N, Ornitz DM (2004) Evolution of the Fgf and Fgfr gene families. Trends Genet 20:563–569
Itoh N, Ornitz DM (2008) Functional evolutionary history of the mouse Fgf gene family. Dev Dyn 237:18–27
Jernvall J, Kettunen P, Karavanova I, Martin L, Thesleff I (1994) Evidence for the role of the enamel knot as a control center in mammalian tooth cusp formation: non-dividing cells express growth stimulating Fgf-4 gene. Int J Dev Biol 38:463–469
Johnson GL, Lapadat R (2002) Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 298:1911–1912
Johnson L et al (1997) K-ras is an essential gene in the mouse with partial functional overlap with N-ras. Genes Dev 11:2468–2481
Kettunen P, Thesleff I (1998) Expression and function of FGFs-4,-8, and-9 suggest functional redundancy and repetitive use as epithelial signals during tooth morphogenesis. Dev Dyn 211:256–268
Kettunen P, Karavanova I, Thesleff I (1998) Responsiveness of developing dental tissues to fibroblast growth factors: expression of splicing alternatives of FGFR1,-2,-3, and of FGFR4; and stimulation of cell proliferation by FGF-2,-4,-8, and-9. Dev Genet 22:374–385
Kettunen P, Laurikkala J, Itäranta P, Vainio S, Itoh N, Thesleff I (2000) Associations of FGF-3 and FGF-10 with signaling networks regulating tooth morphogenesis. Dev Dyn 219:322–332
Klein OD et al (2006) Sprouty genes control diastema tooth development via bidirectional antagonism of epithelial-mesenchymal FGF signaling. Dev Cell 11:181–190
Koussoulakou DS, Margaritis LH, Koussoulakos SL (2009) A curriculum vitae of teeth: evolution, generation, regeneration. Int J Biol Sci 5:226
Li C-Y, Prochazka J, Goodwin AF, Klein OD (2014) Fibroblast growth factor signaling in mammalian tooth development. Odontology 102:1–13
Lin D, Huang Y, He F, Gu S, Zhang G, Chen Y, Zhang Y (2007) Expression survey of genes critical for tooth development in the human embryonic tooth germ. Dev Dyn 236:1307–1312
Liu C, Gu S, Sun C, Ye W, Song Z, Zhang Y, Chen Y (2013) FGF signaling sustains the odontogenic fate of dental mesenchyme by suppressing β-catenin signaling. Development 140:4375–4385
Nakamura T, Yamada Y, Fukumoto S (2012) Review: the regulation of tooth development and morphogenesis. In: Keiichi S (ed) Interface oral health science 2011, Springer, Japan, pp 14–21
Ohuchi H, Hori Y, Yamasaki M, Harada H, Sekine K, Kato S, Itoh N (2000) FGF10 acts as a major ligand for FGF receptor 2 IIIb in mouse multi-organ development. Biochem Biophys Res Commun 277:643–649
Pacold ME et al (2000) Crystal structure and functional analysis of Ras binding to its effector phosphoinositide 3-kinase γ. Cell 103:931–944
Schlessinger J (2000) Cell signaling by receptor tyrosine kinases. Cell 103:211–225
Thesleff I, Keranen S, Jernvall J (2001) Enamel knots as signaling centers linking tooth morphogenesis and odontoblast differentiation. Adv Dent Res 15:14–18
Thisse B, Thisse C (2005) Functions and regulations of fibroblast growth factor signaling during embryonic development. Dev Biol 287:390–402
Umanoff H, Edelmann W, Pellicer A, Kucherlapati R (1995) The murine N-ras gene is not essential for growth and development. Proc Natl Acad Sci 92:1709–1713
Vaahtokari A, Åberg T, Jernvall J, Keränen S, Thesleff I (1996) The enamel knot as a signaling center in the developing mouse tooth. Mech Dev 54:39–43
Vieira A et al (2013) Candidate gene studies in hypodontia suggest role for FGF3. Eur Arch Paediatr Dent 14:405–410
Wang X-P et al (2007) An integrated gene regulatory network controls stem cell proliferation in teeth. PLoS Biol 5:e159
Wang B et al (2014) Expression patterns of WNT/β-CATENIN signaling molecules during human tooth development. J Mol Histol 45:487–496
Yu L, Hébert MC, Zhang YE (2002) TGF-β receptor-activated p38 MAP kinase mediates Smad-independent TGF-β responses. EMBO J 21:3749–3759
Zhang YD, Chen Z, Song YQ, Chao L, Chen YP (2005) Making a tooth: growth factors, transcription factors, and stem cells. Cell Res 15:301–316
Zhang P, Wu Y, Dai Q, Fang B, Jiang L (2013) p38-MAPK signaling pathway is not involved in osteogenic differentiation during early response of mesenchymal stem cells to continuous mechanical strain. Mol Cell Biochem 378:19–28
Acknowledgments
This study was supported by National Natural Science Foundation of China (81100730, 30771132).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Huang, F., Hu, X., Fang, C. et al. Expression profile of critical genes involved in FGF signaling pathway in the developing human primary dentition. Histochem Cell Biol 144, 457–469 (2015). https://doi.org/10.1007/s00418-015-1358-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-015-1358-7