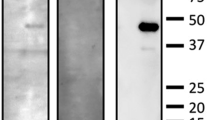
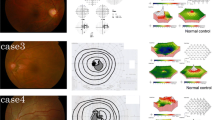
Abstract
Purpose
To evaluate the clinical characteristics of patients with acute zonal occult outer retinopathy (AZOOR), according to the presence or absence of anti-retinal antibodies (ARAs) that are frequently detected in autoimmune retinopathy.
Methods
Retrospective observational case series. This study included 33 patients with acute-stage AZOOR who had been followed up for more than 6 months after the initial visit. The median follow-up period was 26 months. Immunoblot analyses were used to detect autoantibodies for recoverin, carbonic anhydrase II, and α-enolase in serum from these patients. Main outcome measures comprised clinical factors at the initial and final visits, including best-corrected visual acuity, mean deviation on Humphrey perimetry, and retinal morphology, which were statistically compared between patients with AZOOR who exhibited ARAs and those who did not.
Results
At least one serum ARA was detected in 42% of patients with AZOOR. There were no significant differences in clinical factors between the two groups, including follow-up period, best-corrected visual acuity and mean deviation at the initial and final visits, a-wave amplitude on single-flash electroretinography at the initial visit, and frequencies of improvement of the macular ellipsoid zone and AZOOR recurrence.
Conclusions
Our findings suggest that the presence of ARAs did not influence visual outcomes or outer retinal morphology in patients with AZOOR.


Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Gass JD (1993) Acute zonal occult outer retinopathy. Donders Lecture: The Netherlands Ophthalmological Society, Maastricht, Holland, June 19, 1992. J Clin Neuroophthalmol 13:79–97
Gass JD, Agarwal A, Scott IU (2002) Acute zonal occult outer retinopathy: a long-term follow-up study. Am J Ophthalmol 134:329–339
Francis PJ, Marinescu A, Fitzke FW, Bird AC, Holder GE (2005) Acute zonal occult outer retinopathy: towards a set of diagnostic criteria. Br J Ophthalmol 89:70–73
Spaide RF, Koizumi H, Freund KB (2008) Photoreceptor outer segment abnormalities as a cause of blind spot enlargement in acute zonal occult outer retinopathy-complex diseases. Am J Ophthalmol 146:111–120
Saito S, Saito W, Saito M, Hashimoto Y, Mori S, Noda K, Namba K, Ishida S (2015) Acute zonal occult outer retinopathy in Japanese patients: clinical features, visual function, and factors affecting visual function. PLoS One 10:e0125133
Nakao S, Kaizu Y, Yoshida S, Iida T, Ishibashi T (2015) Spontaneous remission of acute zonal occult outer retinopathy: follow-up using adaptive optics scanning laser ophthalmoscopy. Graefes Arch Clin Exp Ophthalmol 253:839–843
Mrejen S, Khan S, Gallego-Pinazo R, Jampol LM, Yannuzzi LA (2014) Acute zonal occult outer retinopathy: a classification based on multimodal imaging. JAMA Ophthalmol 132:1089–1098
Saito M, Saito W, Hashimoto Y, Yoshizawa C, Shinmei Y, Noda K, Ishida S (2014) Correlation between decreased choroidal blood flow velocity and the pathogenesis of acute zonal occult outer retinopathy. Clin Experiment Ophthalmol 42:139–150
Kitakawa T, Hayashi T, Takashina H, Mitoola K, Gekka T, Tsuneoka H (2012) Improvement of central visual function following steroid pulse therapy in acute zonal occult outer retinopathy. Doc Ophthalmol 124:249–254
Adamus G (2009) Autoantibody targets and their cancer relationship in the pathogenicity of paraneoplastic retinopathy. Autoimmun Rev 8:410–414
Maeda T, Maeda A, Maruyama I, Ogawa KI, Kuroki Y, Sahara H, Sato N, Ohguro H (2001) Mechanisms of photoreceptor cell death in cancer-associated retinopathy. Invest Ophthalmol Vis Sci 42:705–712
Adamus G, Aptsiauri N, Guy J, Heckenlively J, Flannery J, Hargrave PA (1996) The occurrence of serum autoantibodies against enolase in cancer-associated retinopathy. Clin Immunol Immunopathol 78:120–129
Ren G, Adamus G (2004) Cellular targets of anti-a-enolase autoantibodies of patients with autoimmune retinopathy. J Autoimmun 23:161–167
Adamus G, Karren L (2009) Autoimmunity against carbonic anhydrase II affects retinal cell functions in autoimmune retinopathy. J Autoimmun 32:133–139
Braithwaite T, Vugler A, Tufail A (2012) Autoimmune retinopathy. Ophthalmologica 228:131–142
Suimon Y, Saito W, Hirooka K, Kanda A, Kitai H, Sakakibara-Konishi J, Ishida S (2017) Improvements of visual function and outer retinal morphology following spontaneous regression of cancer in anti-recoverin cancer-associated retinopathy. Am J Ophthalmol Case Rep 5:137–140
Weleber RG, Watzke RC, Shults WT, Trzupek KM, Heckenlively JR, Egan RA, Adamus G (2005) Clinical and electrophysiologic characterization of paraneoplastic and autoimmune retinopathies associated with antienolase antibodies. Am J Ophthalmol 139:780–794
Ando R, Saito W, Kanda A, Kase S, Fujinami K, Sugahawa M, Nakamura Y, Eguchi S, Mori S, Noda K, Shinoda K, Ishida S (2018) Clinical features of Japanese patients with anti-α-enolase antibody-positive autoimmune retinopathy: novel subtype of multiple drusen. Am J Ophthalmol 196:181–196
Heckenlively JR, Ferreyra HA (2008) Autoimmune retinopathy: a review and summary. Semin Immunopathol 30:127–134
Saito W, Ishida S (2019) Acute zonal occult ocular retinopathy. In: Yu HG (ed) Inflammatory and infectious ocular disorders. Springer Nature, Singapore, pp 45–50
Fagan XJ (2014) A new insight into an old mystery. Clin Exp Ophthalmol 42:103–104
Saito A, Saito W, Furudate N, Ohno S (2007) Indocyanine green angiography in a case of punctate inner choroidopathy associated with acute zonal occult outer retinopathy. Jpn J Ophthalmol 51:295–300
Monson DM, Smith JR (2011) Acute zonal occult outer retinopathy. Surv Ophthalmol 56:23–35
Hashimoto Y, Saito W, Saito M, Hasegawa Y, Takita A, Mori S, Noda K, Ishida S (2017) Relationship between choroidal thickness and visual field impairment in acute zonal occult outer retinopathy. J Ophthalmol 2017:2371032
Tagami M, Matsumiya W, Imai H, Kusuhara S, Honda S, Azumi A (2014) Autologous antibodies to outer retina in acute zonal occult outer retinopathy. Jpn J Ophthalmol 58:462–472
Qian CX, Wang A, DeMill DL, Jayasundera T, Branham K, Abalem MF, Khan N, Heckenlively JR (2017) Prevalence of anti-retinal antibodies in acute zonal occult outer retinopathy (AZOOR): a comprehensive review of 25 cases. Am J Ophthalmol 176:210–218
Adamus G (2011) Is Zonal occult outer retinopathy an autoimmune disease? J Clin Exp Ophthalmol 2:104e
Zeng HY, Liu Q, Peng XY, Cao K, Jin SS, Xu K (2019) Detection of serum anti-retinal antibodies in the Chinese patients with presumed autoimmune retinopathy. Graefes Arch Clin Exp Ophthalmol 257:1759–1764
Forooghian F (2017) Prevalence of antiretinal antibodies in acute zonal occult outer retinopathy: a comprehensive review of 25 cases. Am J Ophthalmol 179:210–211
Saito M, Saito W, Kanda A, Ohguro H, Ishida S (2014) A case of paraneoplastic optic neuropathy and outer retinitis positive for autoantibodies against collapsin response mediator protein-5, recoverin, and α-enolase. BMC Ophthalmol 14:5
Hashimoto Y, Saito W, Namba K, Mizuuchi K, Iwata D, Noda K, Ishida S (2019) Comparison of clinical characteristics in patients with Vogt-Koyanagi-Harada disease with and without anti-retinal antibodies. Graefes Arch Clin Exp Ophthalmol 257:1751–1758
Lavezzo MM, Sakata VM, Morita C, Rodriguez EE, Abdallah SF, da Silva FT, Hirata CE, Yamamoto JH (2016) Vogt-Koyanagi-Harada disease: review of a rare autoimmune disease targeting antigens of melanocytes. Orphanet J Rare Dis 11:29
Adamus G, Amundson D, Seigel GM, Machnicki M (1998) Anti-enolase-alpha autoantibodies in cancer-associated retinopathy: epitope mapping and cytotoxicity on retinal cells. J Autoimmun 11:671–677
Neri P, Ricci F, Giovannini A, Arapi I, De Felici C, Cusumano A, Mariotti C (2014) Successful treatment of an overlapping choriocapillaritis between multifocal choroiditis and acute zonal occult outer retinopathy (AZOOR) with adalimumab (HumiraTM). Int Ophthalmol 34:359–364
Heckenlively JR, Jordan BL, Aptsiauri N (1999) Association of antiretinal antibodies and cystoid macular edema in patients with retinitis pigmentosa. Am J Ophthalmol 127:565–573
Kubicka-Trząska A, Wilańska J, Romanowska-Dixon B, Sanak M (2012) Circulating antiretinal antibodies predict the outcome of anti-VEGF therapy in patients with exudative age-related macular degeneration. Acta Ophthalmol 90:e21–e24
Ten Berge JC, Schreurs MW, Vermeer J, Meester-Smoor MA, Rothova A (2016) Prevalence and clinical impact of antiretinal antibodies in uveitis. Acta Ophthalmol 94:282–288
Ten Berge JC, van Rosmalen J, Vermeer J, Hellström C, Lindskog C, Nilsson P, Qundos U, Rothova A, Schreurs MW (2016) Serum autoantibody profiling of patients with paraneoplastic and non-paraneoplastic autoimmune retinopathy. PLoS One 11:e0167909
Yoshitake S, Murakami T, Suzuma K, Yoshitake T, Uji A, Morooka S, Dodo Y, Fujimoto M, Shan Y, Fort PE, Ito S, Tsujikawa A, Yoshimura N (2019) Anti-fumarase antibody promotes the dropout of photoreceptor inner and outer segments in diabetic macular oedema. Diabetologia 62:504–516
Quintero OL, Amador-Patarroyo MJ, Montoya-Ortiz G, Rojas-Villarraga A, Anaya JM (2012) Autoimmune disease and gender: plausible mechanisms for the female predominance of autoimmunity. J Autoimmun 38:J109-119
Acknowledgements
We would like to thank Mrs. Ikuyo Hirose (Hokkaido University) for her technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The current study was approved by the ethics committee of Hokkaido University Hospital (#020–0236).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Hashimoto, Y., Saito, W., Kanaizumi, S. et al. Comparison of clinical characteristics in patients with acute zonal occult outer retinopathy according to anti-retinal antibody status. Graefes Arch Clin Exp Ophthalmol 259, 2967–2976 (2021). https://doi.org/10.1007/s00417-021-05198-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-021-05198-w