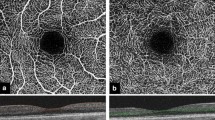
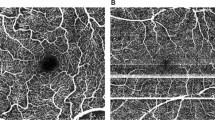
Abstract
Purpose
To assess the prevalences of segmentation errors and motion artifacts in optical coherence tomography angiography (OCT-A) in different retinal diseases
Methods
In a retrospective analysis, multimodal retinal imaging including OCT-A was performed in one eye of 57 healthy controls (50.96 ± 22.4 years) and 149 patients (66.42 ± 14.1 years) affected by different chorioretinal diseases: early/intermediate age-related macular degeneration (AMD; n = 26), neovascular AMD (nAMD; n = 22), geographic atrophy due to AMD (GA; n = 6), glaucoma (n = 28), central serous chorioretinopathy (CSC; n = 14), epiretinal membrane (EM; n = 26), retinal vein occlusion (RVO; n = 11), and retinitis pigmentosa (RP; n = 16). Central 3 × 3 mm2 OCT-A imaging was performed with active eye-tracking (AngioVue, Optovue). Best-corrected visual acuity (BCVA) and signal strength index (SSI) were recorded. Images were independently evaluated by two graders using the OCT-A motion artifact score (MAS; scores I–IV) as well as a newly introduced segmentation accuracy score (SAS; score I–IIB).
Results
Mean SSI was 63.67 ± 9.2 showing a negative correlation with increasing age (rSp = − 0.42, p < 0.001, n = 206). In the healthy cohort, mean MAS was 1.45 ± 0.8 and segmentation was accurate (SAS I) in all eyes. In eyes with retinal pathologies, mean MAS was 2.1 ± 0.9 (p < 0.001). Lowest MAS was observed in GA (2.67 ± 0.5) and RVO (2.45 ± 1.1). Compared to an accurate segmentation in 100% in healthy subjects, 34.2% (n = 51) of all patients showed highest segmentation quality (p < 0.001). 63.8% showed segmentation errors in more than 5% of all single b-scans in one (SAS IIA, n = 58) or at least two (SAS IIB, n = 40) segmentation boundaries. Highest percentages of inaccurate segmentation (SAS IIA or IIB) were observed in the nAMD group (90.1%). The inner plexiform layer was the segmentation boundary most prone to inaccurate segmentation in all pathologies compared to the inner limiting membrane (ILM) and retinal pigment epithelium (RPE) segmentation layer. Incorrect ILM segmentation was only seen in patients with EM.
Conclusions
Prior to both qualitative and quantitative analysis, OCT-A images must be carefully reviewed as motion artifacts and segmentation errors in current OCT-A technology are frequent particularly in pathologically altered maculae.




Similar content being viewed by others
References
Jia Y, Bailey ST, Hwang TS et al (2015) Quantitative optical coherence tomography angiography of vascular abnormalities in the living human eye. Proc Natl Acad Sci U S A 112:E2395–E2402
Cole ED, Ferrara D, Novais EA, Louzada RN, Waheed NK (2016) Clinical trial endpoints for optical coherence tomography angiography in neovascular age-related macular degeneration. Retina 36(Suppl 1):S83–S92
Say EA, Ferenczy S, Magrath GN, Samara WA, Khoo CT, Shields CL (2017) Image quality and artifacts on optical coherence tomography angiography: comparison of pathologic and paired fellow eyes in 65 patients with unilateral choroidal melanoma treated with plaque radiotherapy. Retina 37(9):1660–1673
Sadda SR, Wu Z, Walsh AC et al (2006) Errors in retinal thickness measurements obtained by optical coherence tomography. Ophthalmology 113:285–293
Al-Sheikh M, Ghasemi Falavarjani K, Akil H, Sadda SR (2017) Impact of image quality on OCT angiography based quantitative measurements. Int J Retina Vitreous 3:13
Lauermann JL, Treder M, Heiduschka P, Clemens CR, Eter N, Alten F (2017) Impact of eye-tracking technology on OCT-angiography imaging quality in age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 255:1535–1542
Spaide RF, Fujimoto JG, Waheed NK (2015) Image artifacts in optical coherence tomography angiography. Retina 35:2163–2180
Lumbroso B, Huang D, Jia Y, Rispoli M, Romano A, Waheed NK (2015) Clinical OCT angiography atlas. Jaypee Brothers Medical Publishers, New Delhi
Alten F, Lauermann JL, Clemens CR, Heiduschka P, Eter N (2017) Signal reduction in choriocapillaris and segmentation errors in spectral domain OCT angiography caused by soft drusen. Graefes Arch Clin Exp Ophthalmol 255(12):2347–2355
Ghasemi Falavarjani K, Al-Sheikh M, Akil H, Sadda SR (2017) Image artefacts in swept-source optical coherence tomography angiography. Br J Ophthalmol 101(5):564–568
Han IC, Jaffe GJ (2010) Evaluation of artifacts associated with macular spectral-domain optical coherence tomography. Ophthalmology 117:1177–89.e4
Kraus MF, Potsaid B, Mayer MA et al (2012) Motion correction in optical coherence tomography volumes on a per A-scan basis using orthogonal scan patterns. Biomed Opt Express 3(6):1182–1199
Uji A, Balasubramanian S, Lei J, Baghdasaryan E, Al-Sheikh M, Sadda SR (2017) Choriocapillaris imaging using multiple en face optical coherence tomography angiography image averaging. JAMA Ophthalmol 135:1197–1204
Decroos FC, Stinnett SS, Heydary CS, Burns RE, Jaffe GJ (2013) Reading center characterization of central retinal vein occlusion using optical coherence tomography during the COPERNICUS trial. Transl Vis Sci Technol 2(7):7
Spaide RF, Curcio CA (2017) Evaluation of segmentation of the superficial and deep vascular layers of the retina by optical coherence tomography angiography instruments in normal eyes. JAMA Ophthalmol 135:259–262
Linderman R, Salmon AE, Strampe M, Russillo M, Khan J, Carroll J (2017) Assessing the accuracy of foveal avascular zone measurements using optical coherence tomography angiography: segmentation and scaling. Transl Vis Sci Technol 6(3):16
Zhang M, Hwang TS, Campbell JP, Bailey ST, Wilson DJ, Huang D, Jia Y (2016) Projection-resolved optical coherence tomographic angiography. Biomed Opt Express 7(3):816–828
Wang J, Zhang M, Hwang TS, Bailey ST, Huang D, Wilson DJ, Jia Y (2017) Reflectance-based projection-resolved optical coherence tomography angiography [invited]. Biomed Opt Express 8(3):1536–1548
Onishi AC, Ashraf M, Soetikno BT, Fawzi AA (2018) Multilevel ischemia in disorganization of the retinal inner layers on projection-resolved optical coherence tomography angiography. Retina. https://doi.org/10.1097/IAE.0000000000002179
Fenner BJ, Tan GS, Tan AC, Yeo IY, Wong TY, Cheung GC (2017) Identification of imaging features that determine quality and repeatability of retinal capillary plexus density measurements in OCT angiography. Br J Ophthalmol 102(4):509–514
Shields CL, Say EA, Samara WA, Khoo CT, Mashayekhi A, Shields JA (2016) Optical coherence tomography angiography of the macula after plaque radiotherapy of choroidal melanoma: comparison of irradiated versus nonirradiated eyes in 65 patients. Retina 36(8):1493e1505
Jeoung JW, Choi YJ, Park KH, Kim DM (2013) Macular ganglion cell imaging study: glaucoma diagnostic accuracy of spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci 54:4422–4429
Mwanza JC, Durbin MK, Budenz DL et al (2012) Glaucoma diagnostic accuracy of ganglion cell-inner plexiform layer thickness: comparison with nerve fiber layer and optic nerve head. Ophthalmology 119:1151–1158
Kim HJ, Lee SY, Park KH, Kim DM, Jeoung JW (2016) Glaucoma diagnostic ability of layer-by-layer segmented ganglion cell complex by spectral-domain optical coherence tomography. Invest Ophthalmol Vis Sci 57:4799–4805
Nelis P, Alten F, Clemens CR, Heiduschka P, Eter N (2017) Quantification of changes in foveal capillary architecture caused by idiopathic epiretinal membrane using OCT angiography. Graefes Arch Clin Exp Ophthalmol 255:1319–1324
Rogala J, Zangerl B, Assaad N, Fletcher EL, Kalloniatis M, Nivison-Smith L (2015) In vivo quantification of retinal changes associated with drusen in age-related macular degeneration. Invest Ophthalmol Vis Sci 56:1689–1700
Muftuoglu IK, Ramkumar HL, Bartsch DU, Meshi A, Gaber R, Freeman WR (2017) Quantitative analysis of the inner retinal layer thickness in age-related macular degeneration using corrected optical coherence tomography segmentation. Retina. https://doi.org/10.1097/IAE.0000000000001759
Louzada RN, de Carlo TE, Adhi M et al (2017) Optical coherence tomography angiography artifacts in retinal pigment epithelial detachment. Can J Ophthalmol 52(4):419–424
Lang A, Carass A, Bittner AK, Ying HS, Prince JL (2017) Improving graph-based OCT segmentation for severe pathology in retinitis pigmentosa patients. Proc SPIE Int Soc Opt Eng 10137
Spaide RF (2016) Volume-rendered optical coherence tomography of retinal vein occlusion pilot study. Am J Ophthalmol 165:133–144
Iida Y, Muraoka Y, Ooto S, Suzuma K, Murakami T, Iida-Miwa Y, Ghashut R, Tsujikawa A (2017) Morphologic and functional retinal vessel changes in branch retinal vein occlusion: an optical coherence tomography angiography study. Am J Ophthalmol 182:168–179
Niu S, Chen Q, de Sisternes L, Rubin DL, Zhang W, Liu Q (2014) Automated retinal layers segmentation in SD-OCT images using dual gradient and spatial correlation smoothness constraint. Comput Biol Med 54:116–128
Treder M, Lauermann JL, Eter N (2018) Automated detection of exudative age-related macular degeneration in spectral domain optical coherence tomography using deep learning. Graefes Arch Clin Exp Ophthalmol 256(2):259–265
Bogunovic H, Waldstein S, Schlegl T, Langs G, Sadeghipour A, Liu X, Gerendas B, Osborne A, Schmidt-Erfurth U (2017) Prediction of anti-VEGF treatment requirements in neovascular AMD using a machine learning approach. Invest Ophthalmol Vis Sci 58:3240–4248
de Sisternes L, Jonna G, Moss J, Marmor MF, Leng T, Rubin DL (2017) Automated intraretinal segmentation of SD-OCT images in normal and age-related macular degeneration eyes. Biomed Opt Express 8(3):1926–1949
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Lauermann, J.L., Woetzel, A.K., Treder, M. et al. Prevalences of segmentation errors and motion artifacts in OCT-angiography differ among retinal diseases. Graefes Arch Clin Exp Ophthalmol 256, 1807–1816 (2018). https://doi.org/10.1007/s00417-018-4053-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-018-4053-2