Abstract
Purpose
To describe the treatment response to aflibercept in patients with exudative age-related macular degeneration that showed insufficient or diminishing treatment effects under ranibizumab.
Methods
From December 2012 till June 2013 all patients receiving intravitreal injections of aflibercept after previous treatment with ranibizumab were collected in a database and retrospectively reviewed. Clinical data such as visual acuity or central subfield retinal thickness on optical coherence tomography (OCT) scans were analyzed for the time frame before, during, and shortly after the aflibercept injections. Of particular interest was the comparison of clinical features under ongoing ranibizumab treatment to the time during aflibercept treatment.
Results
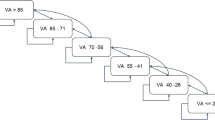
Seventy-one eyes of 65 patients were included in the study. All eyes had previous ranibizumab injections in their medical history, the average number of which was nine (range 3-43). For the total group the mean visual acuity (VA) before the first ranibizumab injection was 0.54 logMAR, and after the last ranibizumab injection was 0.57 logMAR. Mean VA changed from 0.47 logMAR before the first aflibercept injection to 0.25 logMAR after the last aflibercept injection. Central subfield retinal thickness (CSRT) on OCT changed from a mean of 417.28 μm to 349.52 μm under ranibizumab treatment and from 338.76 μm to 272.00 μm under aflibercept treatment. Interestingly, 33 % of cases that did not show a functional improvement under ranibizumab therapy gained visual acuity after aflibercept treatment.
Conclusion
Aflibercept appears to be an effective choice for patients with neovascular age-related macular degeneration who were resistant to previous therapy of ranibizumab. The longevity of this effect still remains questionable.



Similar content being viewed by others
Abbreviations
- OCT:
-
Optical coherence tomography
- SD-OCT:
-
Spectral-domain OCT
- FA:
-
Fluorescein angiography
- VA:
-
Visual acuity
- AMD:
-
Age-related macular degeneration
- CSRT:
-
Central subfield retinal thickness
References
Boyer DS, Heier JS, Brown DM, Francom SF, Ianchulev T, Rubio RG (2009) A phase IIIb study to evaluate the safety of ranibizumab in subjects with neovascular age-related macular degeneration. Ophthalmology 116:1731–1739
Brown DM, Kaiser PK, Michels M, Heier JS, Sy JP, Ianchulev T; ANCHOR Study Group (2006) Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 355:1432–1444
Holz FG, Amoaku W, Donate J, Guymer RH, Kellner U, Schlingemann RO, Weichselberger A, Staurenghi G; SUSTAIN Study Group (2011) Safety and efficacy of a flexible dosing regimen of ranibizumab in neovascular agerelated macular degeneration: the SUSTAIN study. Ophthalmology 118:663–671
Lally DR, Gerstenblith AT, Regillo CD (2012) Preferred therapies for neovascular age-related macular degeneration. Curr Opin Ophthalmol 23:182–188
Lim LS, Mitchell P, Seddon JM, Holz FG, Wong TY (2012) Age-related macular degeneration. Lancet 379:1728–1738
Singer MA, Awh CC, Sadda S, Freeman WR, Antoszyk AN, Wong P, Tuomi L (2012) HORIZON: an open-label extension trial of ranibizumab for choroidal neovascularization secondary to age-related macular degeneration. Ophthalmology 119:1175–1183
Keane PA, Liakopoulos S, Ongchin SC, Heussen FM, Msutta S, Chang KT, Walsh AC, Sadda SR (2008) Quantitative subanalysis of optical coherence tomography after treatment with ranibizumab for neovascular age-related macular degeneration. Invest Ophthalmol Vis Sci 49:3115–3120
Binder S (2012) Loss of reactivity in intravitreal anti-VEGF therapy: tachyphylaxis or tolerance? Br J Ophthalmol 96:1–2
Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD, Kirchhof B, Ho A, Ogura Y, Yancopoulos GD, Stahl N, Vitti R, Berliner AJ, Soo Y, Anderesi M, Groetzbach G, Sommerauer B, Sandbrink R, Simader C, Schmidt-Erfurth U; VIEW 1 and VIEW 2 Study Groups (2012) Intravitreal aflibercept (VEGF Trap-Eye) in wet age-related macular degeneration. Ophthalmology 119:2537–2548
Browning DJ, Kaiser PK, Rosenfeld PJ, Stewart MW (2012) Aflibercept for age-related macular degeneration: a game-changer or quiet addition? Am J Ophthalmol 154:222–226
Gasperini JL, Fawzi AA, Khondkaryan A, Lam L, Chong LP, Eliott D, Walsh AC, Hwang J, Sadda SR (2012) Bevacizumab and ranibizumab tachyphylaxis in the treatment of choroidal neovascularisation. Br J Ophthalmol 96:14–20
Holash J, Davis S, Papadopoulos N, Croll SD, Ho L, Russell M, Boland P, Leidich R, Hylton D, Burova E, Ioffe E, Huang T, Radziejewski C, Bailey K, Fandl JP, Daly T, Wiegand SJ, Yancopoulos GD, Rudge JS (2002) VEGF-Trap: a VEGF blocker with potent antitumor effects. Proc Natl Acad Sci U S A 99:11393–11398
Papadopoulos N, Martin J, Run Q, Rafique A, Rosconi MP, Shi E, Pyles EA, Yancopoulos GD, Stahl N, Wiegand SJ (2012) Binding and neutralization of vascular endothelial growth factor (VEGF) and related ligands by VEGF Trap, ranibizumab and bevacizumab. Angiogenesis 15:171–185
Stewart MW, Rosenfeld PJ (2008) Predicted biological activity of intravitreal VEGF Trap. Br J Ophthalmol 92:667–668
Dixon JA, Oliver SC, Olson JL, Mandava N (2009) VEGF Trap-Eye for the treatment of neovascular age-related macular degeneration. Expert Opin Investig Drugs 18:1573–1580
Schaal S, Kaplan HG, Tezel TH (2008) Is there tachyphylaxis to intravitreal anti-vascular endothelial growth factor pharmacotherapy in age-related macular degeneration? Ophthalmology 115:2199–2205
Grossniklaus HE, Ling JX, Wallace TM, Dithmar S, Lawson DH, Cohen C, Elner VM, Elner SG, Sternberg P Jr (2002) Macrophage and retinal pigment epithelium expression of angiogenic cytokines in choroidal neovascularization. Mol Vis 8:119–126
Espinosa-Heidmann DG, Suner IJ, Hernandez P, Monroy D, Csaky KG, Cousins SW (2003) Macrophage depletion diminishes lesion size and severity in experimental choroidal neovascularization. Invest Ophthalmol Vis Sci 44:3586–3592
Sakurai E, Anand A, Ambati BK, van Rooijen N, Ambati J (2003) Macrophage depletion inhibits experimental choroidal neovascularization. Invest Ophthalmol Vis Sci 44:3578–3585
Muether PS, Hoerster R, Hermann MM, Kirchhof B, Fauser S (2013) Long-term effects of ranibizumab treatment delay in neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 251(2):453–458
Disclosure
Dr. Florian Heussen receives research support from Novartis and Heidelberg Engineering. He also serves as a consultant for Allergan Inc. Dr. Antonia Joussen receives financial compensation for lectures from Novartis Inc., Allergan Inc., and Bayer Inc.
Conflict of interest
The authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heussen, F.M., Shao, Q., Ouyang, Y. et al. Clinical outcomes after switching treatment from intravitreal ranibizumab to aflibercept in neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 252, 909–915 (2014). https://doi.org/10.1007/s00417-013-2553-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-013-2553-7