Abstract
Background
To investigate the role of CYR61 as a retinal angiogenic factor in proliferative diabetic retinopathy (PDR).
Methods
Effects of CYR61 on RF/6A cell proliferation, migration and angiogenesis were observed by MTT assay, Transwell assay, and tube formation assay. The expression and distribution of CYR61 on retina layers of diabetic mouse were demonstrated by immunohistochemistry. The expression of Cyr61 mRNA in diabetic mouse retina was evaluated by reverse transcription–polymerase chain reaction (RT-PCR). Vitreous CYR61 levels of PDR and non-diabetic patients were measured by enzyme-linked immunosorbent assay (ELISA). Expression and distribution of CYR61 on epiretinal membrane of PDR, proliferative vitreoretinopathy (PVR) and idiopathic epiretinal membrane were evaluated by immunohistochemistry.
Results
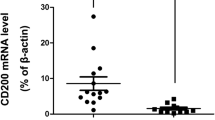
RF/6A cell proliferation, migration and tube formation capacity increased with increased concentration of CYR61 (p = 0.000). Anti-CYR61 antibody could inhibit cell migration and tube formation promoted by CYR61. In diabetic mouse, CYR61 was expressed in retina layers just as normal mouse, but the staining was stronger than normal in ganglion cell layer and inner plexiform layer. The Cyr61 mRNA expression in retina of diabetic mouse was more than that in normal mouse (p = 0.009). Vitreous CYR61 level was higher in patients with PDR than non-diabetic patients (p = 0.000). PDR patients with plenty of neovasculature on retina and epiretinal membranes had higher level of vitreous CYR61 than patients with little neovasculature (p = 0.001). CYR61 expressed in the cytoplasm of epiretinal membranes in PDR, especially in the wall cells of the tube-like structure.
Conclusions
CYR61 are likely to be involved in the pathogenesis of diabetic retinopathy, and may play a role in the course of neovasculation.






Similar content being viewed by others
References
Wang X, Wang G, Wang Y (2009) Intravitreous vascular endothelial growth factor and hypoxia-inducible factor 1a in patients with proliferative diabetic retinopathy. Am J Ophthalmol 148:883–889
Erdol H, Turk A, Akyol N, Imamoglu HI (2010) The results of intravitreal bevacizumab injections for persistent neovascularizations in proliferative diabetic retinopathy after photocoagulation therapy. Retina 30:570–577
Soheilian M, Ramezani A, Obudi A, Bijanzadeh B, Salehipour M, Yaseri M, Ahmadieh H, Dehghan MH, Azarmina M, Moradian S, Peyman GA (2009) Randomized trial of intravitreal bevacizumab alone or combined with triamcinolone versus macular photocoagulation in diabetic macular edema. Ophthalmology 116:1142–1150
Chen Y, Du XY (2007) Functional properties and intracellular signaling of CCN1/Cyr61. J Cell Biochem 100:1337–1345
Leu SJ, Lam SC, Lau LF (2002) Pro-angiogenic activities of CYR61 (CCN1) mediated through integrins αvβ3 and α6β1 in human umbilical vein endothelial cells. J Biol Chem 277:46248–46255
Chen CC, Chen N, Lau LF (2001) The angiogenic factors Cyr61 and connective tissue growth factor induce adhesive signaling in primary human skin fibroblasts. J Biol Chem 276:10443–10452
Hughes JM, Kuiper EJ, Klaassen I, Canning P, Stitt AW, Van Bezu J, Schalkwijk CG, Van Noorden CJ, Schlingemann RO (2007) Advanced glycation end products cause increased CCN family and extracellular matrix gene expression in the diabetic rodent retina. Diabetologia 50:1089–1098
You JJ, Yang CH, Chen MS, Yang CM (2009) Cysteine-rich 61, a member of the CCN family, is involved in the pathogenesis of proliferative diabetic retinopathy. Invest Ophthalmol Vis Sci 50:3447–3455
Liu H, Yang R, Tinner B, Choudhry A, Schutze N, Chaqour B (2008) Cysteine-rich protein 61 and connective tissue growth factor induce deadhesion and anoikis of retinal pericytes. Endocrinology 149:1666–1677
Kuiper EJ, Hughes JM, Van Geest RJ, Vogels IM, Goldschmeding R, Van Noorden CJ, Schlingemann RO, Klaassen I (2007) Effect of VEGF-A on expression of profibrotic growth factor and extracellular matrix genes in the retina. Invest Ophthalmol Vis Sci 48:4267–4276
Rachfal AW, Brigstock DR (2005) Structural and functional properties of CCN proteins. Vitam Horm 70:69–103
Chen CC, Lau LF (2009) Functions and mechanisms of action of CCN matricellular proteins. Int J Biochem Cell Biol 41:771–783
Babic AM, Kireeva ML, Kolesnikova TV, Lau LF (1998) CYR61, a product of a growth factor-inducible immediate early gene, promotes angiogenesis and tumor growth. Proc Natl Acad Sci USA 95:6355–6360
Dhar A, Ray A (2010) The CCN family proteins in carcinogenesis. Exp Oncol 32:2–9
Lou DA, Hu FN (1987) Co-distribution of von Willebrand factor and fibronectin in cultured rhesus endothelial cells. Histochem J 19:431–438
Lou DA, Hu FN (1987) Specific antigen and organelle expression of a long-term rhesus endothelial cell line. In Vitro Cell Dev Biol 23:75–85
Chen Y, Li XX, Xing NZ, Cao XG (2008) Quercetin inhibits choroidal and retinal angiogenesis in vitro. Graefes Arch Clin Exp Ophthalmol 246:373–378
Lin MT, Kuo IH, Chang CC, Chu CY, Chen HY, Lin BR, Sureshbabu M, Shih HJ, Kuo ML (2008) Involvement of hypoxia-inducing factor-1alpha-dependent plasminogen activator inhibitor-1 up-regulation in Cyr61/CCN1-induced gastric cancer cell invasion. J Biol Chem 283:15807–15815
Hirschfeld M, zur Hausen A, Bettendorf H, Jäger M, Stickeler E (2009) Alternative splicing of Cyr61 is regulated by hypoxia and significantly changed in breast cancer. Cancer Res 69:2082–2090
Wolf N, Yang W, Dunk CE, Gashaw I, Lye SJ, Ring T, Schmidt M, Winterhager E, Gellhaus A (2010) Regulation of the matricellular proteins CYR61 (CCN1) and NOV (CCN3) by hypoxia-inducible factor-1{alpha} and transforming-growth factor-{beta}3 in the human trophoblast. Endocrinology 151:2835–2845
Hilfiker-Kleiner D, Kaminski K, Kaminska A, Fuchs M, Klein G, Podewski E, Grote K, Kiian I, Wollert KC, Hilfiker A, Drexler H (2004) Regulation of proangiogenic factor CCN1 in cardiac muscle: impact of ischemia, pressure overload, and neurohumoral activation. Circulation 109:2227–2233
Muramatsu Y, Tsujie M, Kohda Y, Pham B, Perantoni AO, Zhao H, Jo SK, Yuen PS, Craig L, Hu X, Star RA (2002) Early detection of cysteine rich protein 61 (CYR61, CCN1) in urine following renal ischemic reperfusion injury. Kidney Int 62:1601–1610
Mendez JD, Ramos HG (1994) Animal models in diabetes research. Arch Med Res 25:367–375
Takeo-Goto S, Doi M, Ma N, Goto R, Semba R, Uji Y (2002) Immunohistochemical localization of amino acids in the diabetic retina of Goto-Kakizaki rats. Ophthalmic Res 34:139–145
Conflict of Interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial relationship
None.
Presentation at a conference
None.
Clinical Trial Registration number
None.
The authors have full control of all primary data, and agree to allow Graefe's Archive for Clinical and Experimental Ophthalmology to review the data upon request.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
Immunohistochemistry staining of CYR61 in epiretinal membranes (scale: 50 μm). A. epiretinal membrane of PDR, CYR61 expression in the wall cells of the tube-like structure; B. Negative control of the same sample in A; C. CYR61 expression in epiretinal membrane of PDR; D. CYR61 expression in epiretinal membrane of PVR (JPEG 150 kb)
Rights and permissions
About this article
Cite this article
Zhang, X., Yu, W. & Dong, F. Cysteine-rich 61 (CYR61) is up-regulated in proliferative diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 250, 661–668 (2012). https://doi.org/10.1007/s00417-011-1882-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-011-1882-7