Abstract
Background
Mesial temporal lobe epilepsy with hippocampal sclerosis (MTLE-HS) is usually associated with a poor response to antiseizure medications. We focused on MTLE-HS patients who were seizure free on medication to: (1) determine the clinical factors associated with seizure freedom and (2) develop a machine-learning classifier to better earlier identify those patients.
Methods
We performed a retrospective, multicentric study comparing 64 medically treated seizure-free MTLE-HS patients with 200 surgically treated drug-resistant MTLE-HS patients. First, we collected medical history and seizure semiology data. Then, we developed a machine-learning classifier based on clinical data.
Results
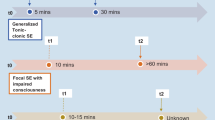
Medically treated seizure-free MTLE-HS patients were seizure-free for at least 2 years, and for a median time of 7 years at last follow-up. Compared to drug-resistant MTLE-HS patients, they exhibited: an older age at epilepsy onset (22.5 vs 8.0 years, p < 0.001), a lesser rate of: febrile seizures (39.0% vs 57.5%, p = 0.035), focal aware seizures (previously referred to as aura)(56.7% vs 90.0%, p < 0.001), autonomic focal aware seizures in presence of focal aware seizure (17.6% vs 59.4%, p < 0.001), dystonic posturing of the limbs (9.8% vs 47.0%, p < 0.001), gestural (27.4% vs 94.0%, p < 0.001), oro-alimentary (32.3% vs 75.5%, p < 0.001) or verbal automatisms (12.9% vs 36.0%, p = 0.001). The classifier had a positive predictive value of 0.889, a sensitivity of 0.727, a specificity of 0.962, a negative predictive value of 0.893.
Conclusions
Medically treated seizure-free MTLE-HS patients exhibit a distinct clinical profile. A classifier built with readily available clinical data can identify them accurately with excellent positive predictive value. This may help to individualize the management of MTLE-HS patients according to their expected pharmacosensitivity.
Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Abbreviations
- MTLE-HS:
-
Mesial temporal lobe epilepsy with hippocampal sclerosis
- MTLE:
-
Mesial temporal lobe epilepsy
- TLE:
-
Temporal lobe epilepsy
- ASM:
-
Antiseizure medications
- HS:
-
Hippocampal sclerosis
- FTBS:
-
Focal to bilateral tonic–clonic seizures
- SEEG:
-
Stereo-electroencephalography
References
Kim WJ, Park SC, Lee SJ et al (1999) The prognosis for control of seizures with medications in patients with MRI evidence for mesial temporal sclerosis. Epilepsia 40(3):290–293. https://doi.org/10.1111/j.1528-1157.1999.tb00706.x
Aguglia U, Gambardella A, Le Piane E et al (1998) Mild non-lesional temporal lobe epilepsy. a common, unrecognized disorder with onset in adulthood. Can J Neurol Sci. 25(4):282–286. https://doi.org/10.1017/s0317167100034284.
Coan AC, Campos BM, Bergo FPG et al (2015) Patterns of seizure control in patients with mesial temporal lobe epilepsy with and without hippocampus sclerosis. Arq Neuropsiquiatr 73(2):79–82. https://doi.org/10.1590/0004-282X20140199
Rambeck B, Jürgens UH, May TW et al (2006) Comparison of brain extracellular fluid, brain tissue, cerebrospinal fluid, and serum concentrations of antiepileptic drugs measured intraoperatively in patients with intractable epilepsy. Epilepsia 47(4):681–694. https://doi.org/10.1111/j.1528-1167.2006.00504.x
Löscher W, Klotz U, Zimprich F, Schmidt D (2009) The clinical impact of pharmacogenetics on the treatment of epilepsy. Epilepsia 50(1):1–23. https://doi.org/10.1111/j.1528-1167.2008.01716.x
Semah F, Picot MC, Adam C et al (1998) Is the underlying cause of epilepsy a major prognostic factor for recurrence? Neurology 51(5):1256–1262. https://doi.org/10.1212/wnl.51.5.1256
Stephen LJ, Kwan P, Brodie MJ (2001) Does the cause of localisation-related epilepsy influence the response to antiepileptic drug treatment? Epilepsia 42(3):357–362. https://doi.org/10.1046/j.1528-1157.2001.29000.x
Labate A, Ventura P, Gambardella A et al (2006) MRI evidence of mesial temporal sclerosis in sporadic “benign” temporal lobe epilepsy. Neurology 66(4):562–565. https://doi.org/10.1212/01.wnl.0000198208.59347.96
Brodie MJ, Barry SJE, Bamagous GA, Norrie JD, Kwan P (2012) Patterns of treatment response in newly diagnosed epilepsy. Neurology 78(20):1548–1554. https://doi.org/10.1212/WNL.0b013e3182563b19
Lee SK (2014) Treatment strategy for the patient with hippocampal sclerosis who failed to the first antiepileptic drug. J Epilepsy Res 4(1):1–6. https://doi.org/10.14581/jer.14001
Kwan P, Brodie MJ (2000) Early identification of refractory epilepsy. N Engl J Med 342(5):314–319. https://doi.org/10.1056/NEJM200002033420503
Kwan P, Arzimanoglou A, Berg AT et al (2009) Definition of drug resistant epilepsy: consensus proposal by the ad hoc task force of the ILAE commission on therapeutic strategies: definition of drug resistant epilepsy. Epilepsia 51(6):1069–1077. https://doi.org/10.1111/j.1528-1167.2009.02397.x
Mathon B, Bielle F, Samson S et al (2017) Predictive factors of long-term outcomes of surgery for mesial temporal lobe epilepsy associated with hippocampal sclerosis. Epilepsia 58(8):1473–1485. https://doi.org/10.1111/epi.13831
Aguglia U, Beghi E, Labate A et al (2011) Age at onset predicts good seizure outcome in sporadic non-lesional and mesial temporal sclerosis based temporal lobe epilepsy. J Neurol Neurosurg Psychiatry 82(5):555–559. https://doi.org/10.1136/jnnp.2010.217620
Steinhoff BJ, Scholly J, Dentel C, Staack AM (2013) Is routine electroencephalography (EEG) a useful biomarker for pharmacoresistant epilepsy? Epilepsia 54:63–66
Stefanatou M, Gatzonis S, Peskostas A, Paraskevas G, Koutroumanidis M (2019) Drug-responsive versus drug-refractory mesial temporal lobe epilepsy: a single-center prospective outcome study. Postgrad Med 131(7):479–485. https://doi.org/10.1080/00325481.2019.1663126
Andrade-Valença LP, Valença MM, Ribeiro LT et al (2003) Clinical and neuroimaging features of good and poor seizure control patients with mesial temporal lobe epilepsy and hippocampal atrophy. Epilepsia 44(6):807–814. https://doi.org/10.1046/j.1528-1157.2003.58002.x
Labate A, Gambardella A, Andermann E et al (2011) Benign mesial temporal lobe epilepsy. Nat Rev Neurol 7(4):237–240. https://doi.org/10.1038/nrneurol.2010.212
Silva-Alves MS, Secolin R, Carvalho BS et al (2017) A prediction algorithm for drug response in patients with mesial temporal lobe epilepsy based on clinical and genetic information. PLoS One 12(1):e0169214. https://doi.org/10.1371/journal.pone.0169214
Labate A, Cerasa A, Gambardella A, Aguglia U, Quattrone A (2008) Hippocampal and thalamic atrophy in mild temporal lobe epilepsy A VBM study. Neurology 71(14):1094–1101. https://doi.org/10.1212/01.wnl.0000326898.05099.04
Labate A, Caligiuri ME, Fortunato F, Ferlazzo E, Aguglia U, Gambardella A (2020) Late drug-resistance in mild MTLE: Can it be influenced by preexisting white matter alterations? Epilepsia 61(5):924–934. https://doi.org/10.1111/epi.16503
Asadi-Pooya AA, Rostamihosseinkhani M, Farazdaghi M (2021) Seizure and social outcomes in patients with non-surgically treated temporal lobe epilepsy. Epilepsy Behav 122:108227. https://doi.org/10.1016/j.yebeh.2021.108227
Hitiris N, Mohanraj R, Norrie J, Sills GJ, Brodie MJ (2007) Predictors of pharmacoresistant epilepsy. Epilepsy Res 75(2–3):192–196. https://doi.org/10.1016/j.eplepsyres.2007.06.003
Brodie MJ (2013) Road to refractory epilepsy: the Glasgow story. Epilepsia 54:5–8. https://doi.org/10.1111/epi.12175
Josephson CB, Pohlmann-Eden B (2012) The, “natural” history of medically treated temporal lobe epilepsy: What can an evidence-based approach tell us? Epilepsy Res Treatment 2012:1–8. https://doi.org/10.1155/2012/216510
Kobayashi E, Li LM, Lopes-Cendes I, Cendes F (2002) Magnetic resonance imaging evidence of hippocampal sclerosis in asymptomatic, first-degree relatives of patients with familial mesial temporal lobe epilepsy. Arch Neurol 59(12):1891. https://doi.org/10.1001/archneur.59.12.1891
No YJ, Zavanone C, Bielle F et al (2017) Medial temporal lobe epilepsy associated with hippocampal sclerosis is a distinctive syndrome. J Neurol 264(5):875–881. https://doi.org/10.1007/s00415-017-8441-z
Heuser K, Cvancarova M, Gjerstad L, Taubøll E (2011) Is Temporal Lobe Epilepsy with childhood febrile seizures a distinctive entity? A comparative study Seizure 20(2):163–166. https://doi.org/10.1016/j.seizure.2010.11.015
Russo A, Arbune AA, Bansal L, Mindruta I, Gobbi G, Duchowny M (2019) The localizing value of epileptic auras: pitfalls in semiology and involved networks. Epileptic Disord 21(6):519–528. https://doi.org/10.1684/epd.2019.1106
Vercueil L, Hirsch E (2002) Seizures and the basal ganglia: a review of the clinical data. Epileptic Disord 4(Suppl 3):S47-54
Calleja J, Carpizo R, Berciano J (1988) Orgasmic epilepsy. Epilepsia 29(5):635–639. https://doi.org/10.1111/j.1528-1157.1988.tb03774.x
Fish DR, Gloor P, Quesney FL, Olivier A (1993) Clinical responses to electrical brain stimulation of the temporal and frontal lobes in patients with epilepsy. Pathophysiological implications Brain 116(Pt 2):397–414. https://doi.org/10.1093/brain/116.2.397
Bouilleret V, Dupont S, Spelle L, Baulac M, Samson Y, Semah F (2002) Insular cortex involvement in mesiotemporal lobe epilepsy: a positron emission tomography study. Ann Neurol 51(2):202–208. https://doi.org/10.1002/ana.10087
Li H, Meng Q, Liu Y, Dong S, Zhang H (2021) Oral and manual automatisms elicited by electrical stimulation of the pars opercularis cortex in a patient with frontal lobe epilepsy. Clin Neurol Neurosurg 208:106784. https://doi.org/10.1016/j.clineuro.2021.106784
Aupy J, Noviawaty I, Krishnan B et al (2018) Insulo-opercular cortex generates oroalimentary automatisms in temporal seizures. Epilepsia 59(3):583–594. https://doi.org/10.1111/epi.14011
Maillard L, Vignal JP, Gavaret M et al (2004) Semiologic and electrophysiologic correlations in temporal lobe seizure subtypes. Epilepsia 45(12):1590–1599. https://doi.org/10.1111/j.0013-9580.2004.09704.x
Munari C, Soncini M, Brunet P et al (1985) Electro-clinical semiology of subintrant temporal lobe seizures. Rev Electroencephalogr Neurophysiol Clin 15(3):289–298. https://doi.org/10.1016/s0370-4475(85)80011-3
Talairach J, Bancaud J, Geier S et al (1973) The cingulate gyrus and human behaviour. Electroencephalogr Clin Neurophysiol 34(1):45–52. https://doi.org/10.1016/0013-4694(73)90149-1
Dupont S, Semah F, Baulac M, Samson Y (1998) The underlying pathophysiology of ictal dystonia in temporal lobe epilepsy: An FDG-PET study. Neurology 51(5):1289–1292. https://doi.org/10.1212/WNL.51.5.1289
Newton MR, Berkovic SF, Austin MC, Reutens DC, McKay WJ, Bladin PF (1992) Dystonia, clinical lateralization, and regional blood flow changes in temporal lobe seizures. Neurology 42(2):371–377. https://doi.org/10.1212/wnl.42.2.371
Tang F, Hartz AMS, Bauer B (2017) Drug-Resistant Epilepsy: Multiple Hypotheses. Few Answers Front Neurol 8:301. https://doi.org/10.3389/fneur.2017.00301
Fang M, Xi ZQ, Wu Y, Wang XF (2011) A new hypothesis of drug refractory epilepsy: neural network hypothesis. Med Hypotheses 76(6):871–876. https://doi.org/10.1016/j.mehy.2011.02.039
Bone B, Fogarasi A, Schulz R et al (2012) Secondarily generalized seizures in temporal lobe epilepsy: Secondarily Generalized Seizures. Epilepsia 53(5):817–824. https://doi.org/10.1111/j.1528-1167.2012.03435.x
von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP et al (2014) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg 12(12):1495–1499
Acknowledgements
The authors thank Drs. J.P.Vignal, A.Gales, V.Frazzini, C.Adam and Pr P.Thomas for their contribution to the recruitment of this study
Funding
No funding to disclose.
Author information
Authors and Affiliations
Contributions
MC and SD had the idea for the study, included patients and wrote the initial protocol and analysis plan. MC collected the data. MC, MH and SD did the data analysis. MH did the statistical analysis and developed the machine-learning classifier. MC produced the table and wrote the first draft of the manuscript. MH and SD further reviewed the manuscript and contributed to the final version of the manuscript. All other authors (NC, WS, CM, HC, LV, MG, BH, AB, SL, L.Maz, L.Min, L.Mai) included in the study patients from their epilepsy centres and contributed to editing and commenting on the final version.
Corresponding authors
Ethics declarations
Conflicts of interest
None of the authors has any conflict of interest to disclose.
Ethical publication statement
We confirm that we have read the Journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines. We used the STROBE reporting guidelines [44].
IRB statement
The study was approved by the local ethics committee of Sorbonne University (no CER-2021-089). The patients signed an informed consent form for the use of medical reports and data.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cheval, M., Houot, M., Chastan, N. et al. Early identification of seizure freedom with medical treatment in patients with mesial temporal lobe epilepsy and hippocampal sclerosis. J Neurol 270, 2715–2723 (2023). https://doi.org/10.1007/s00415-023-11603-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11603-7